-

Figure 1.
Maximum likelihood strict consensus tree illustrating the phylogeny of the species of Russulales based on ITS + nLSU + mtSSU + rpb2 + tef1- α sequences. Branches are labeled with maximum likelihood bootstrap values higher than 70%, and Bayesian posterior probabilities more than 0.95, respectively.
-
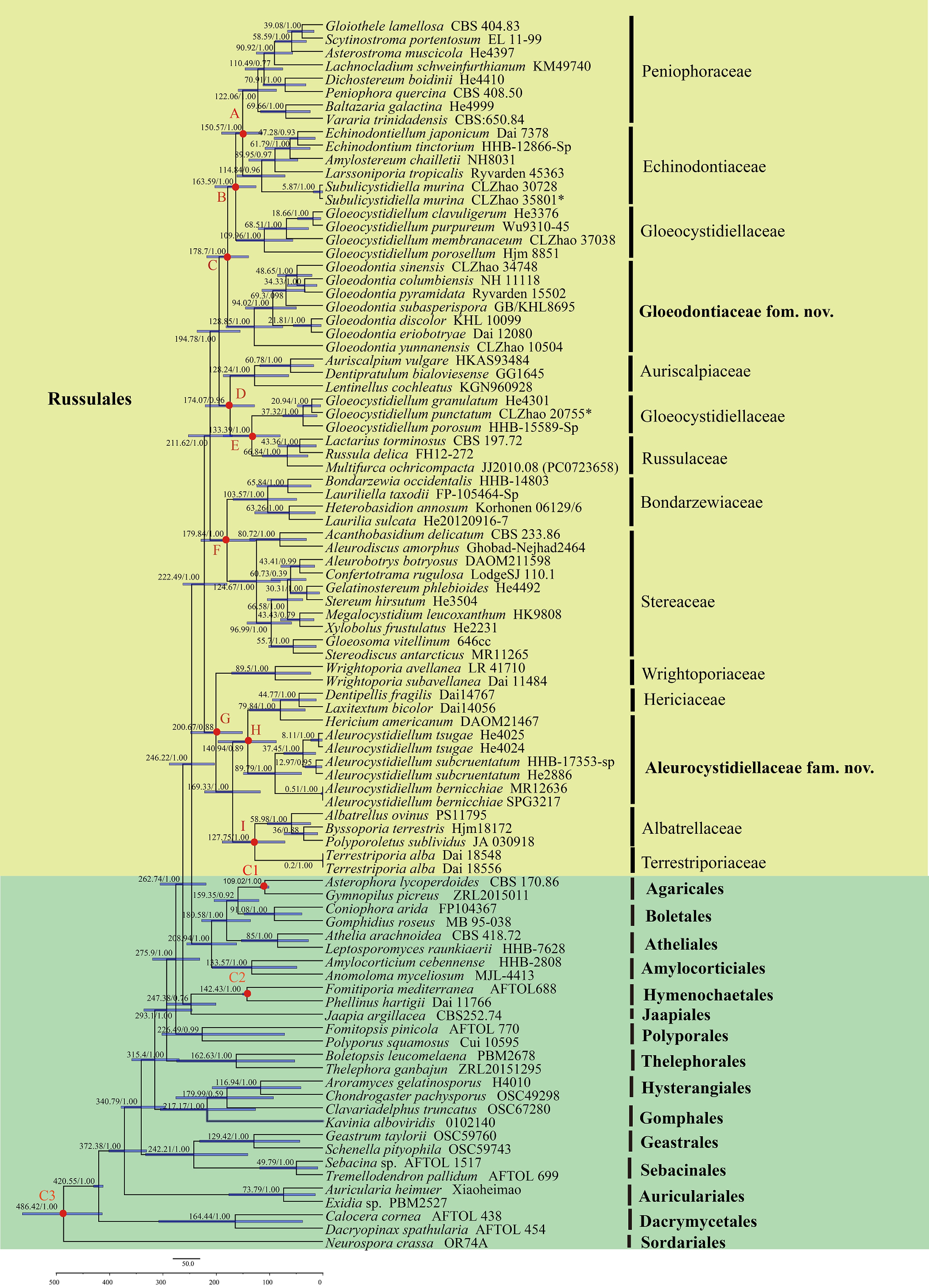
Figure 2.
Divergence time estimation of families within Russulales from molecular clock analysis sampling tree based on the combined sequence dataset of ITS, nLSU, rpb2, and tef1- α. Posterior probabilities not less than 0.80, and the mean ages of each node are annotated. The 90% highest posterior densities of divergence time estimation are marked by horizontal bars.
-
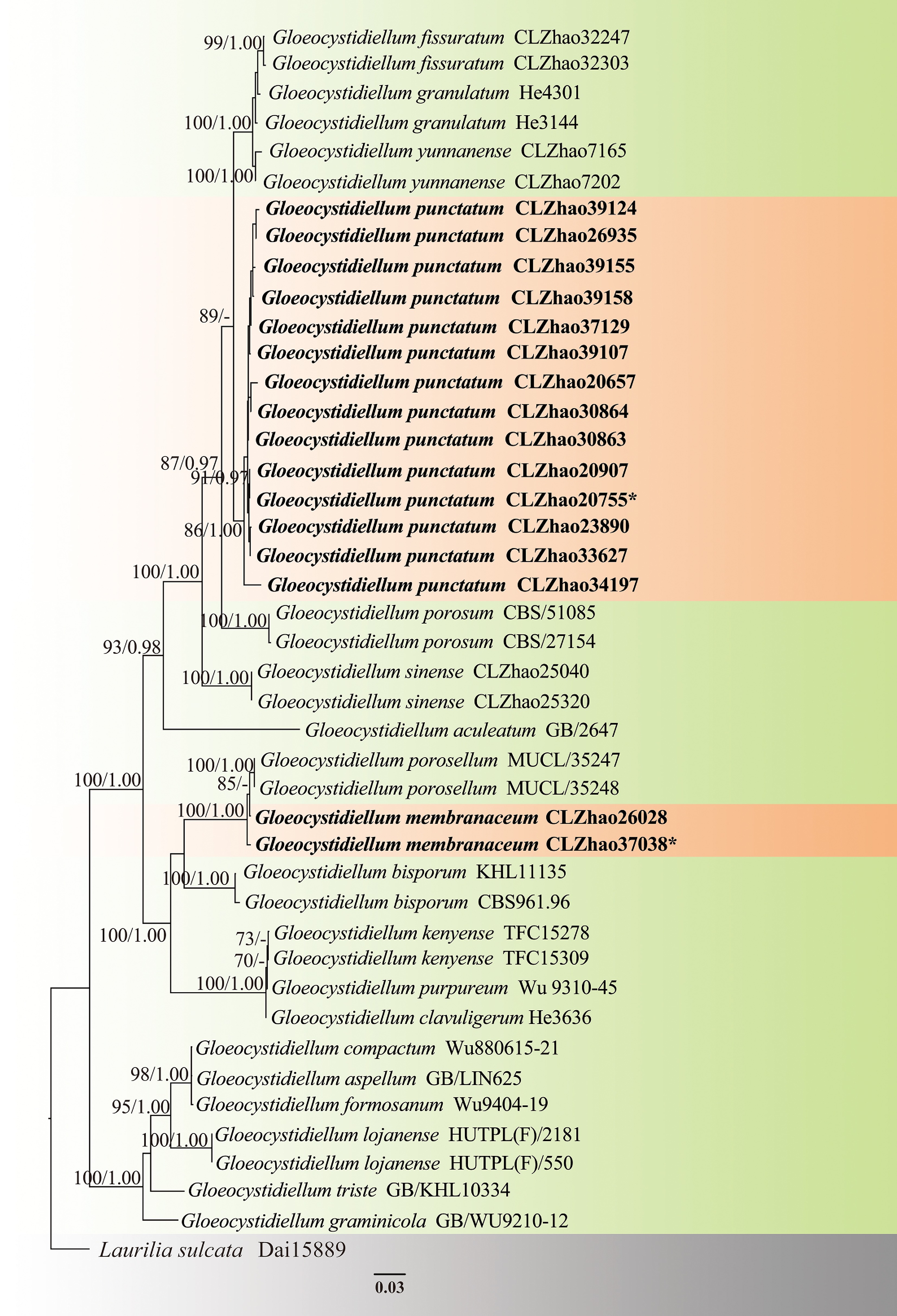
Figure 3.
Maximum likelihood strict consensus tree illustrating the phylogeny of two new species of Gloeocystidiellum based on ITS and nLSU sequences. Branches are labeled with maximum likelihood bootstrap values higher than 70%, and Bayesian posterior probabilities more than 0.95, respectively. The new species are in bold,, and type specimens are indicated with an asterisk (*).
-

Figure 4.
Maximum likelihood strict consensus tree illustrating the phylogeny of species of family Hericiaceae, and related families based on ITS and nLSU sequences. Branches are labeled with maximum likelihood bootstrap values higher than 70%, and Bayesian posterior probabilities more than 0.95, respectively. The new species are in bold, and type specimens are indicated with an asterisk (*).
-
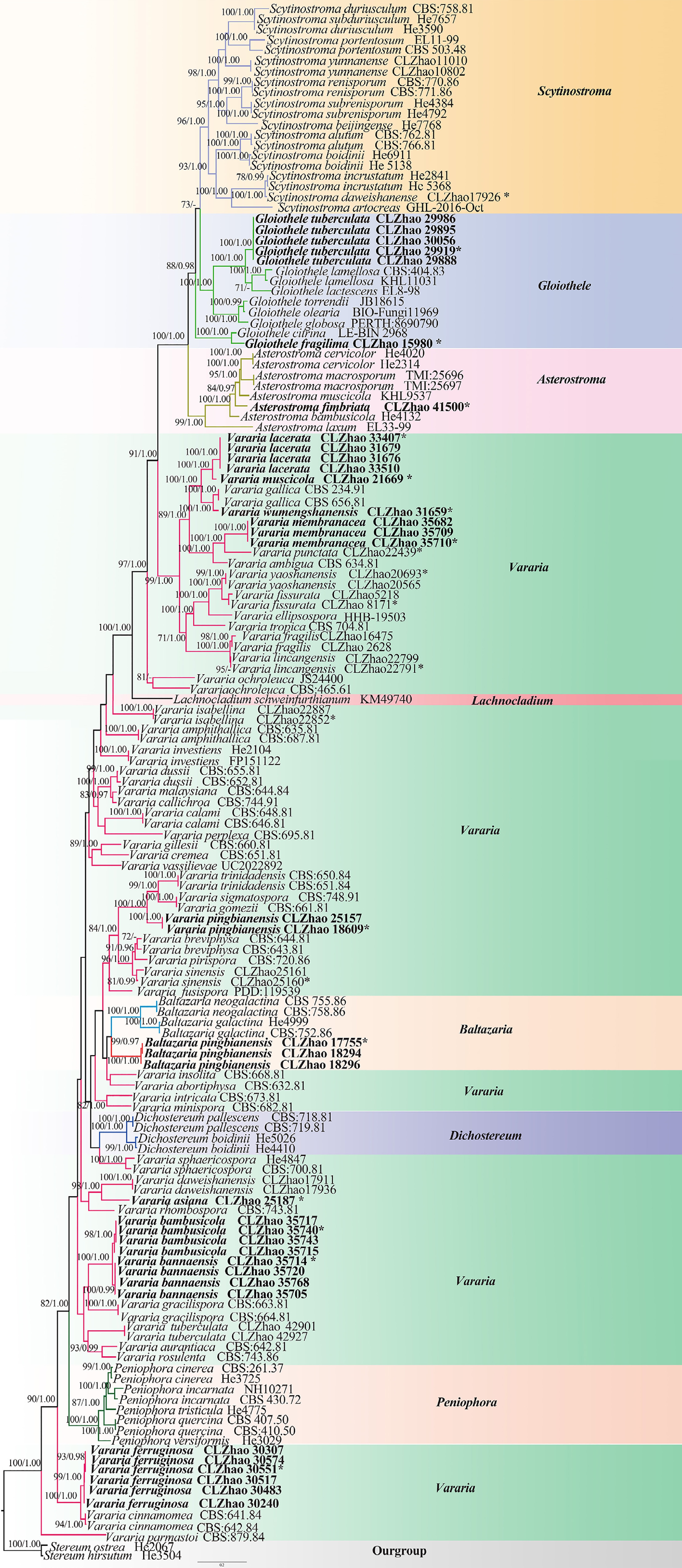
Figure 5.
Maximum likelihood strict consensus tree illustrating the phylogeny of species of family Peniophoraceae based on ITS and nLSU sequences. Branches are labeled with maximum likelihood bootstrap values higher than 70%, and Bayesian posterior probabilities more than 0.95, respectively. The new species are in bold, and type specimens are indicated with an asterisk (*).
-
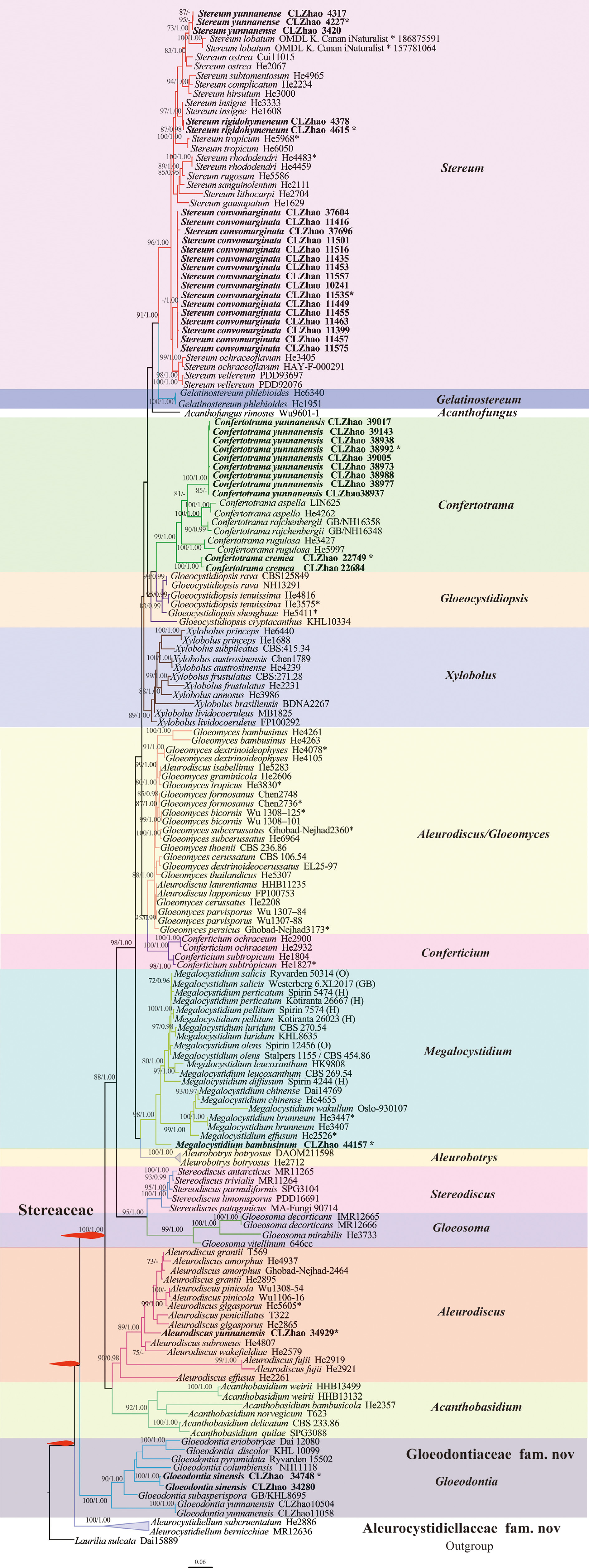
Figure 6.
Maximum likelihood strict consensus tree illustrating phylogeny of species of family Stereaceae and related families based on ITS and nLSU sequences. Branches are labeled with maximum likelihood bootstrap values higher than 70%, and Bayesian posterior probabilities more than 0.95, respectively. The new species are in bold, and type specimens are indicated with an asterisk (*).
-

Figure 7.
Basidiomata and microscopic structures of Subulicystidiella murina (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidia and basidioles. (e) Barrelled gloeocystidia. (f) Subulate cystidia (g) Section of hymenium. Scale bars: (a) = 1 cm, (b) = 1 mm, (c) = 5 µm, (d)–(g) = 10 µm.
-
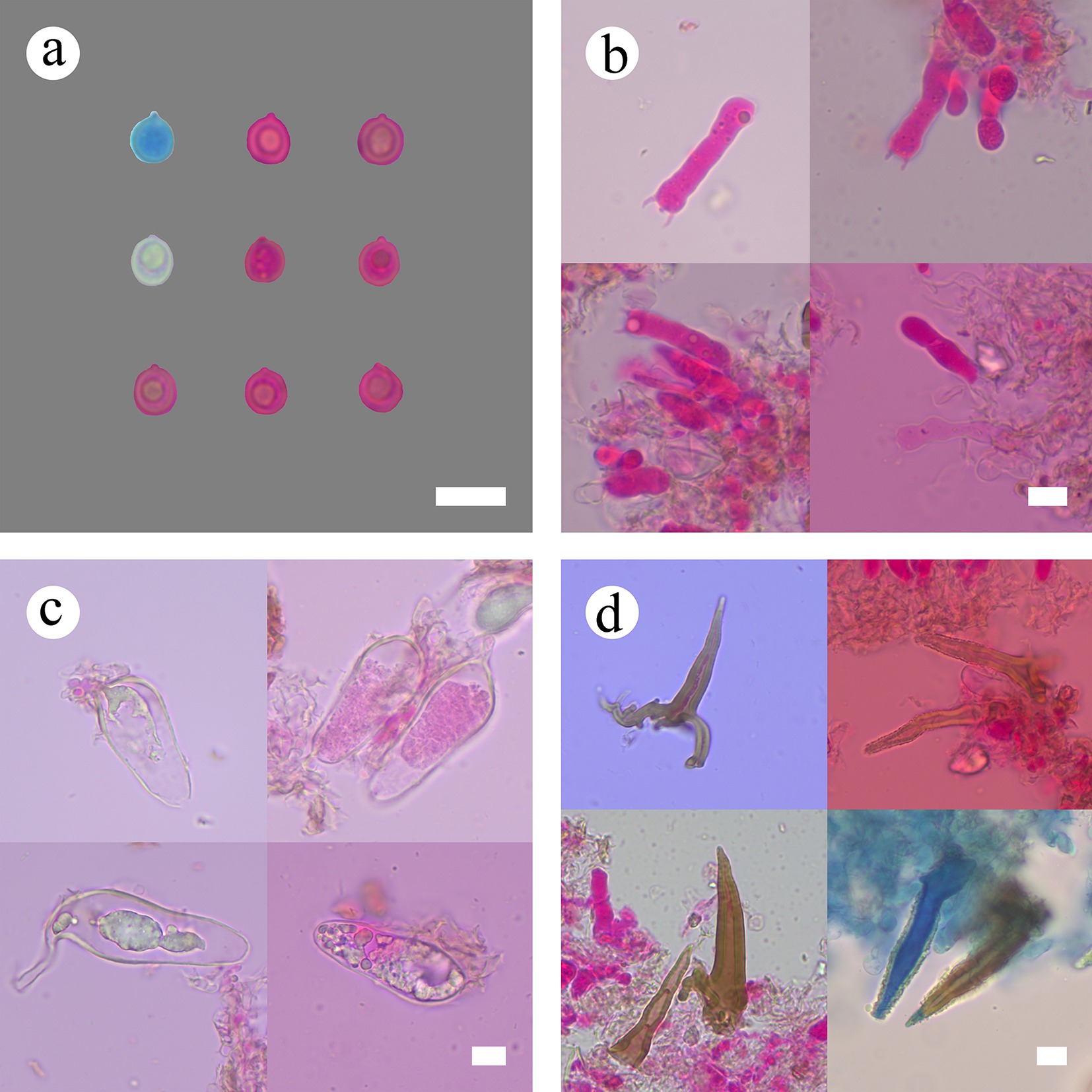
Figure 8.
Sections of hymenium of Subulicystidiella murina (holotype). (a) Basidiospores. (b) Basidia and basidioles. (c) Barrelled cystidia. (d) Subulate cystidia. Scale bars: (a)–(d) = 10 µm.
-

Figure 9.
Basidiomata and microscopic structures of Gloeocystidiellum membranaceum (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Gloeocystidia. (e) Basidioles. (f) Basidia. (g) Section of hymenium. Scale bars: (a) = 1 cm, (b) = 1 mm, (c) = 5 µm, (d)–(g) = 10 µm.
-
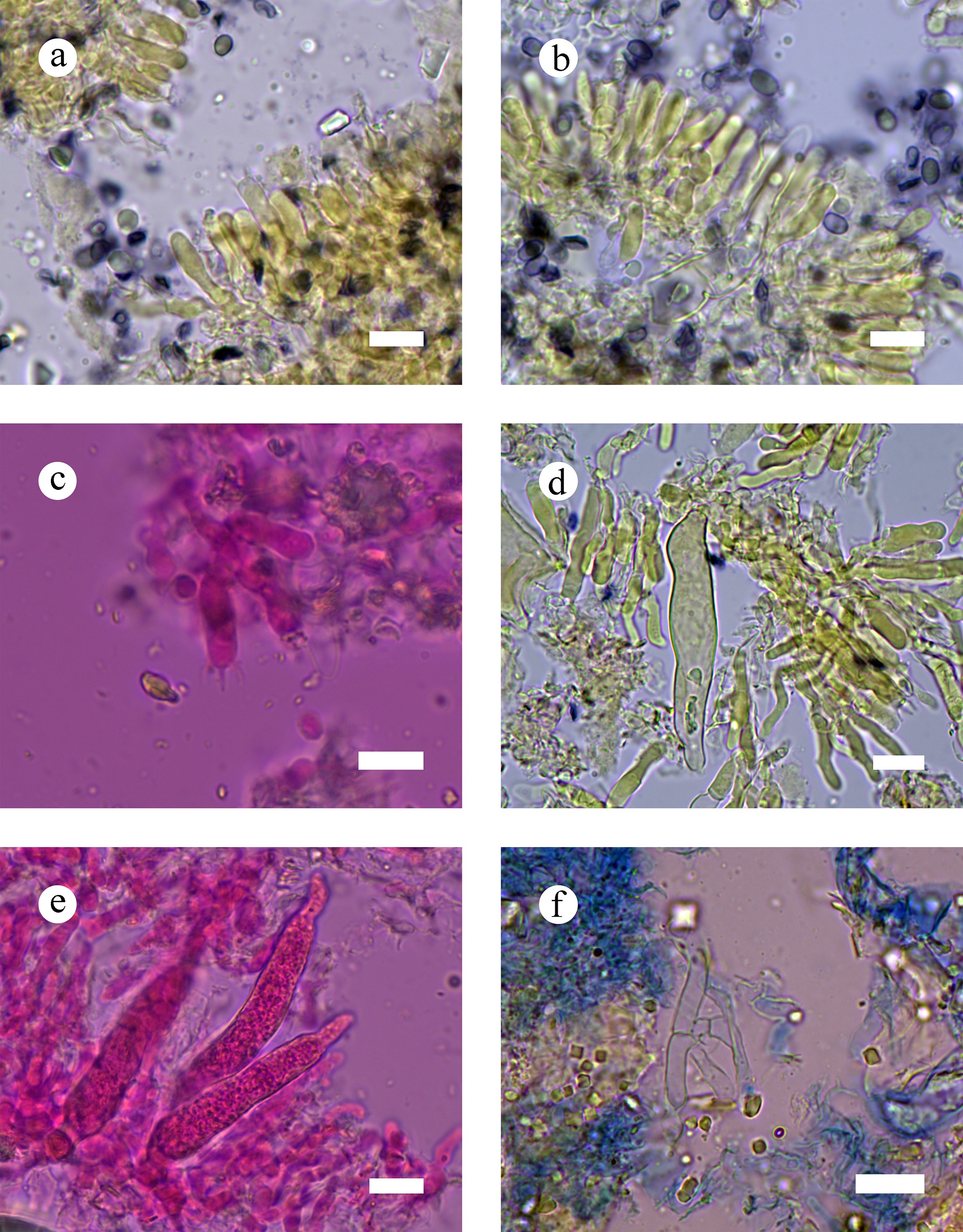
Figure 10.
Sections of hymenia of Gloeocystidiellum membranaceum (holotype). (a)–(c) Basidiospores and basidia. (d) Gloeocystidia. (e) Hyphae from context. (f) Section of hymenium. Scale bars: (a)–(f) = 10 µm.
-

Figure 11.
Basidiomata and microscopic structures of Gloeocystidiellum punctatum (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidioles. (e) Basidia. (f) Section of hymenium. Scale bars: (a) = 1 cm, (b) = 1 mm, (c) = 5 µm, (d)–(f) = 10 µm.
-

Figure 12.
Basidiomata and microscopic structures of Gloeodontia sinensis (holotype). (a) Basidiomata on the substrate. (b) Characteristics of hymenophore. (c) Basidiospores. (d) Basidioles. (e) Basidia. (f) Cystidia. (g) Section of the hymenium. Scale bars: (a) = 1 cm, (b) = 1 mm, (c) = 5 μm, (d)–(g) = 10 μm.
-
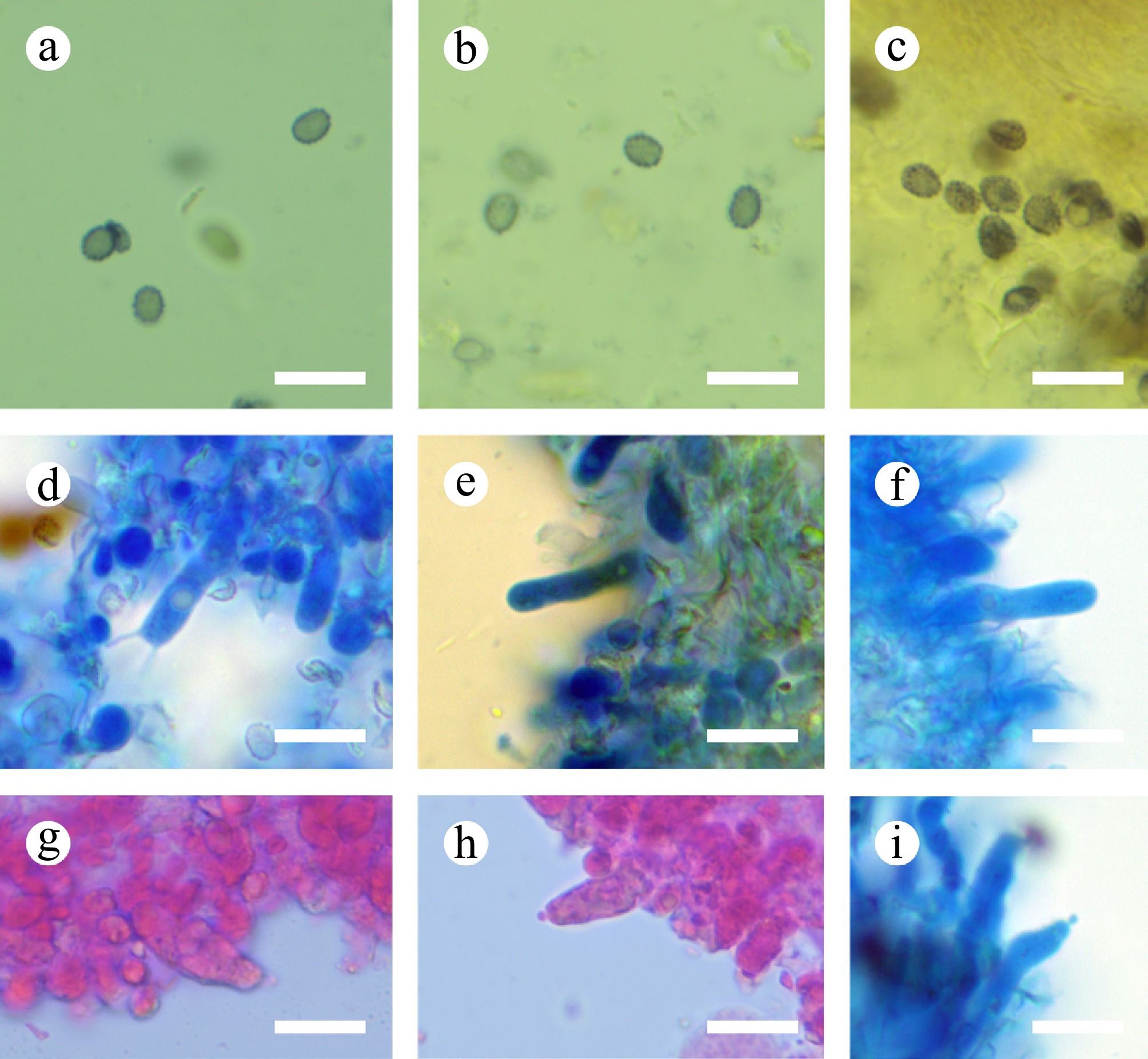
Figure 13.
Sections of hymenium of Gloeodontia sinensis (holotype). (a)–(c) Basidiospores. (d)–(f) Basidia and basidioles. (g)–(i) Cystidia. Scale bars: (a)–(i) = 10 µm.
-

Figure 14.
Basidiomata and microscopic structures of Dentipellis yingjiangensis (holotype). (a) Basidiomata on the substrate. (b) Characteristics of hymenophore. (c) Basidiospores. (d) Basidia and basidioles. (e) Fusiform gloeocystidia. (f) Mushroom-shaped gloeocystidia. (g) Long cylindrical gloeocystidia. (h) Section of hymenium. Scale bars: (a) = 1 cm; (b) = 1 mm, (c) = 5 µm, (d)–(h) = 10 µm.
-
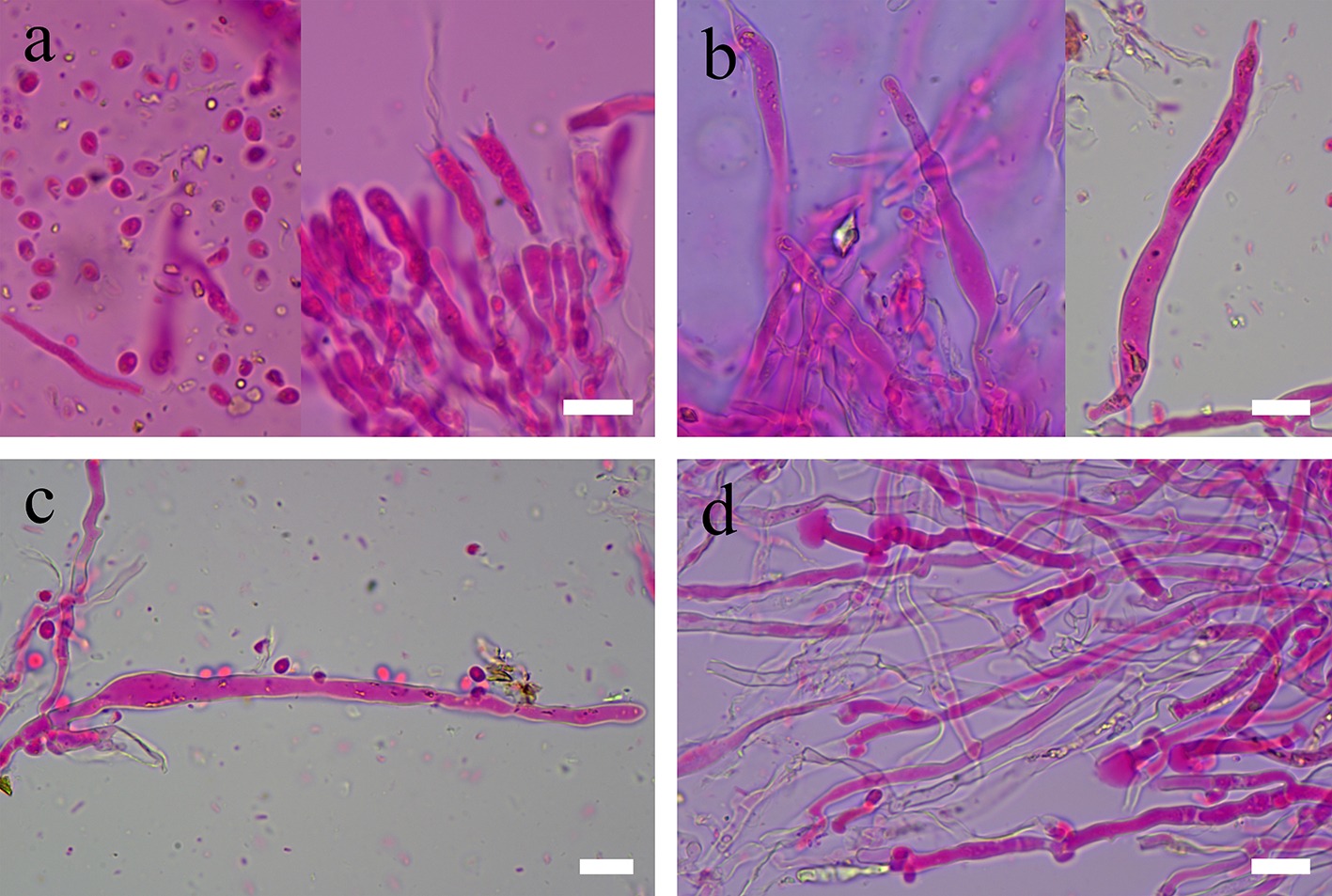
Figure 15.
Sections of hymenium of Dentipellis yingjiangensis (holotype). (a) Basidiospores, basidia, and basidioles. (b), (c) Fusiform gloeocystidia. (d) Mushroom-shaped gloeocystidia. Scale bars: (a)–(d) = 10 µm.
-

Figure 16.
Basidiomata and microscopic structures of Laxitextum cremeum (holotype). (a) Basidiomata on the substrate. (b) Characteristics of hymenophore. (c) Basidiospores. (d) Basidia. (e) Basidioles. (f) Subcylindrical gloeocystidia. (g) Narrowly subcylindrical gloeocystidia. (h) Bottled gloeocystidia. (i) Piriform gloeocystidia. (g) Section of hymenium. Scale bars: (a) = 1 cm; (b) = 1 mm, (c) = 5 µm, (d)–(j) = 10 µm.
-
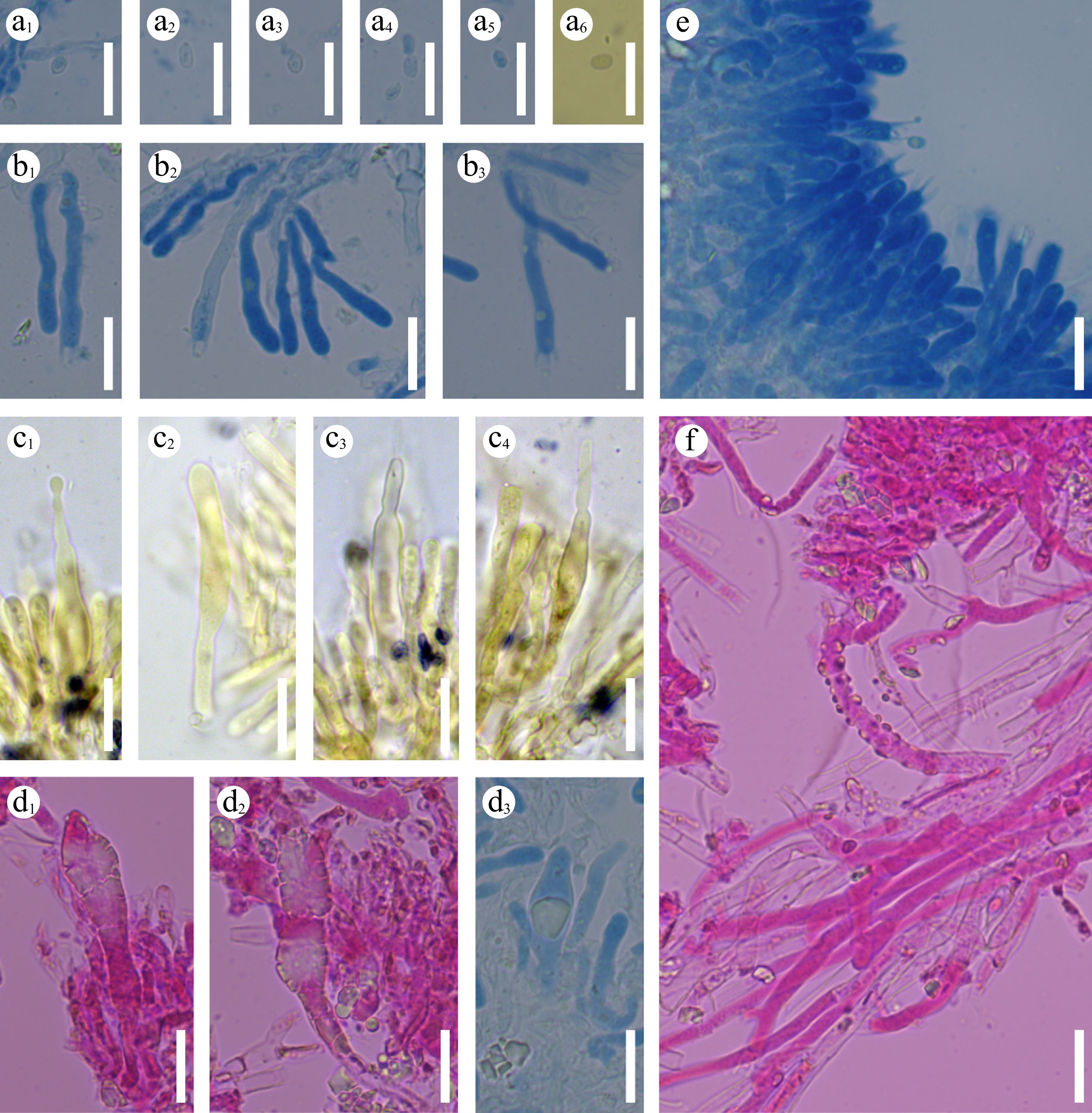
Figure 17.
Sections of hymenium of Laxitextum cremeum (holotype). a 1–a 6 Basidiospores. b 1–b 3 Basidia and basidioles. c 1–c 4 subcylindrical gloeocystidia. d 1–d 3 Pyriform gloeocystidia. (e) Section of hymenium. (f) Generative hyphae. Scale bars: (a)–(f) = 10 µm.
-
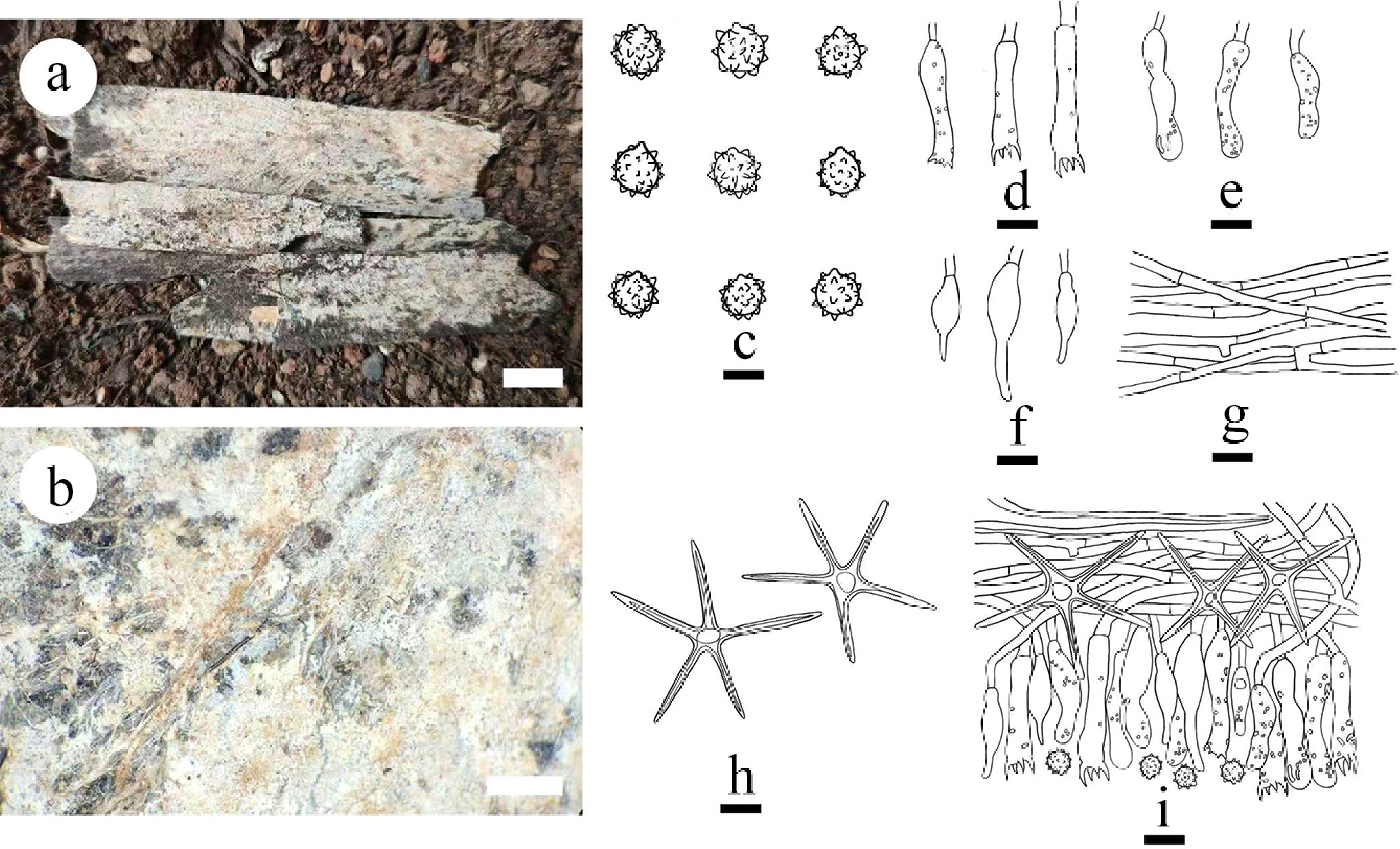
Figure 18.
Basidiomata and microscopic structures of Asterostroma fimbriatum (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidia. (e) Basidioles. (f) Gloeocystidia. (g) Hyphae from context. (h) Asterosetae. (i) Section of hymenium. Scale bars: (a) = 1 cm, (b) = 1 mm, (c) = 5 µm, (d)–(i) = 10 µm.
-
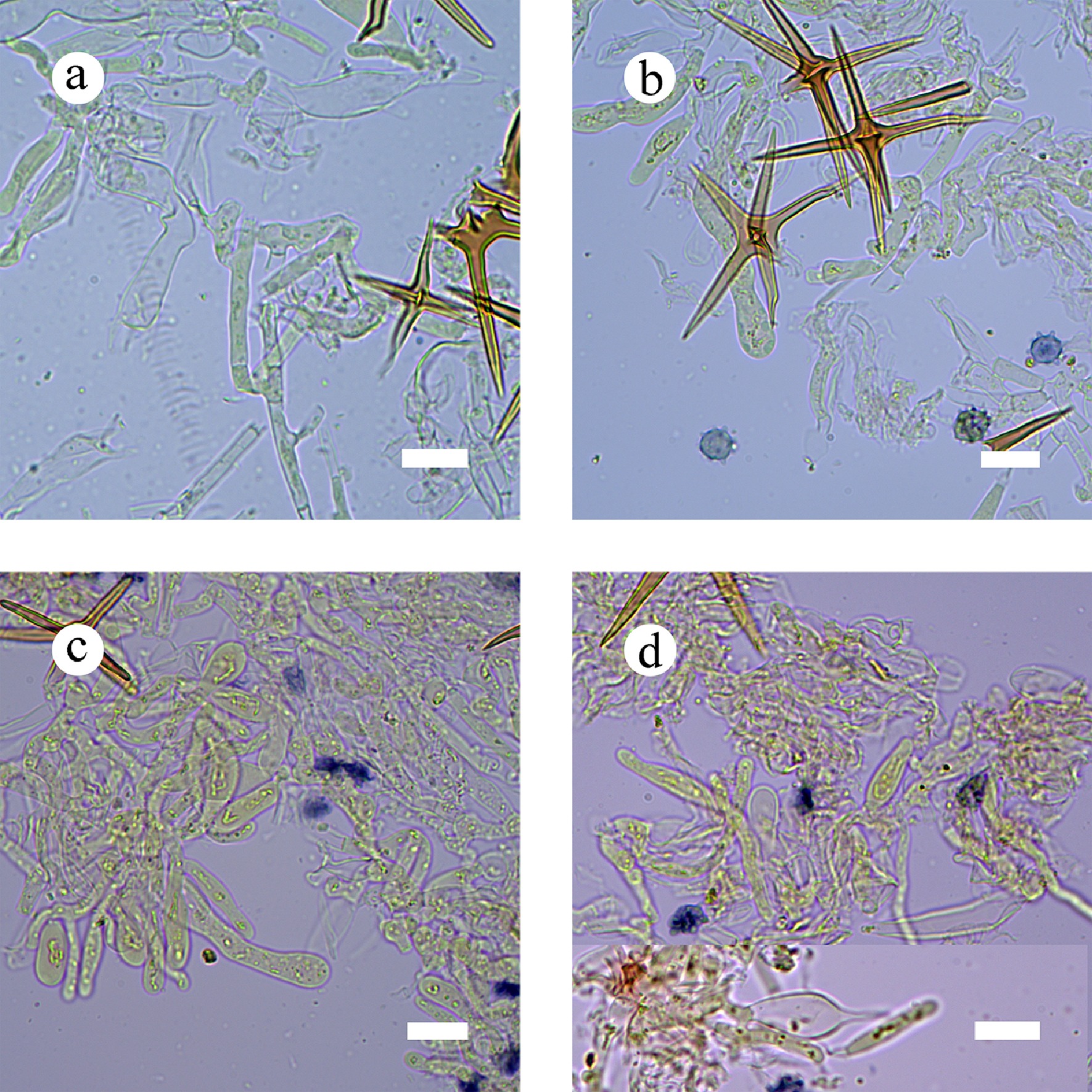
Figure 19.
Sections of hymenium of Asterostroma fimbriatum (holotype). (a) Basidia. (b) Basidiospores. (c) Basidioles. (d) Gloeocystidia. Scale bars: (a)–(d) = 10 µm.
-

Figure 20.
Basidiomata and microscopic structures of Baltazaria pingbianensis (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidioles. (e) Basidia. (f) Skeletal hyphae. (g) Section of hymenium. Scale bars: (a) = 1 cm, (b) = 1 mm, (c) = 5 µm, (d)–(g) = 10 µm.
-

Figure 21.
Basidiomata and microscopic structures of Gloiothele fragilima (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidioles. (e) Basidia. (f) Gloeocystidia. (g) Section of hymenium. Scale bars: (a) = 1 cm; (b) = 1 mm, (c) = 5 µm, (d)–(g) = 10 µm.
-
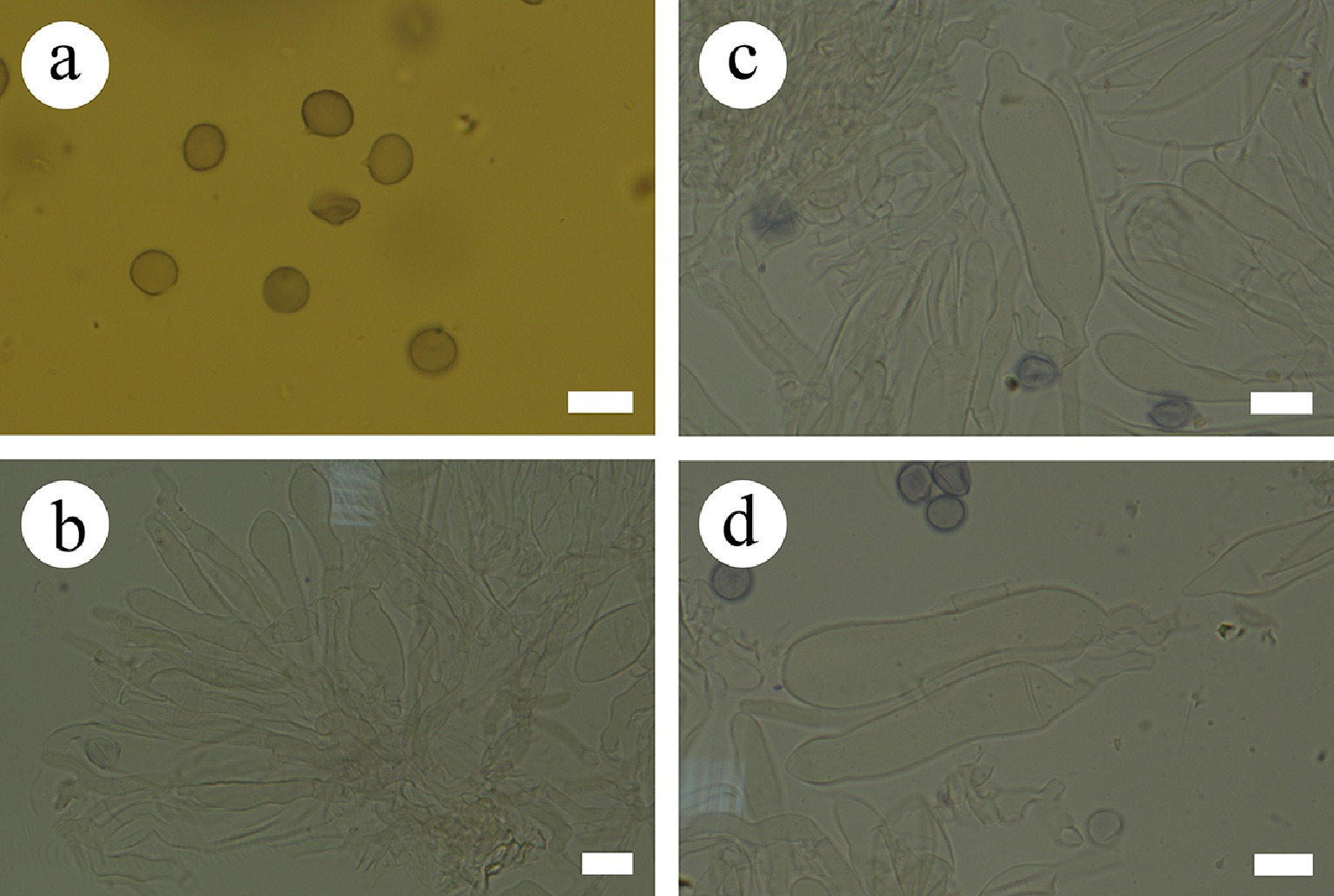
Figure 22.
Sections of hymenium of Gloiothele fragilima (holotype). (a) Basidiospores. (b) Section of hymenium. (c), (d) Gloeocystidia. Scale bars: (a)–(d) = 10 µm.
-

Figure 23.
Basidiomata and microscopic structures of Gloiothele tuberculata (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidia and basidioles. (e) Barrelled gloeocystidia. (f) Long cylindrical gloeocystidia. (g) Section of hymenium. Scale bars: (a) = 1 cm; (b) = 1 mm, (c) = 5 µm, (d)–(g) = 10 µm.
-
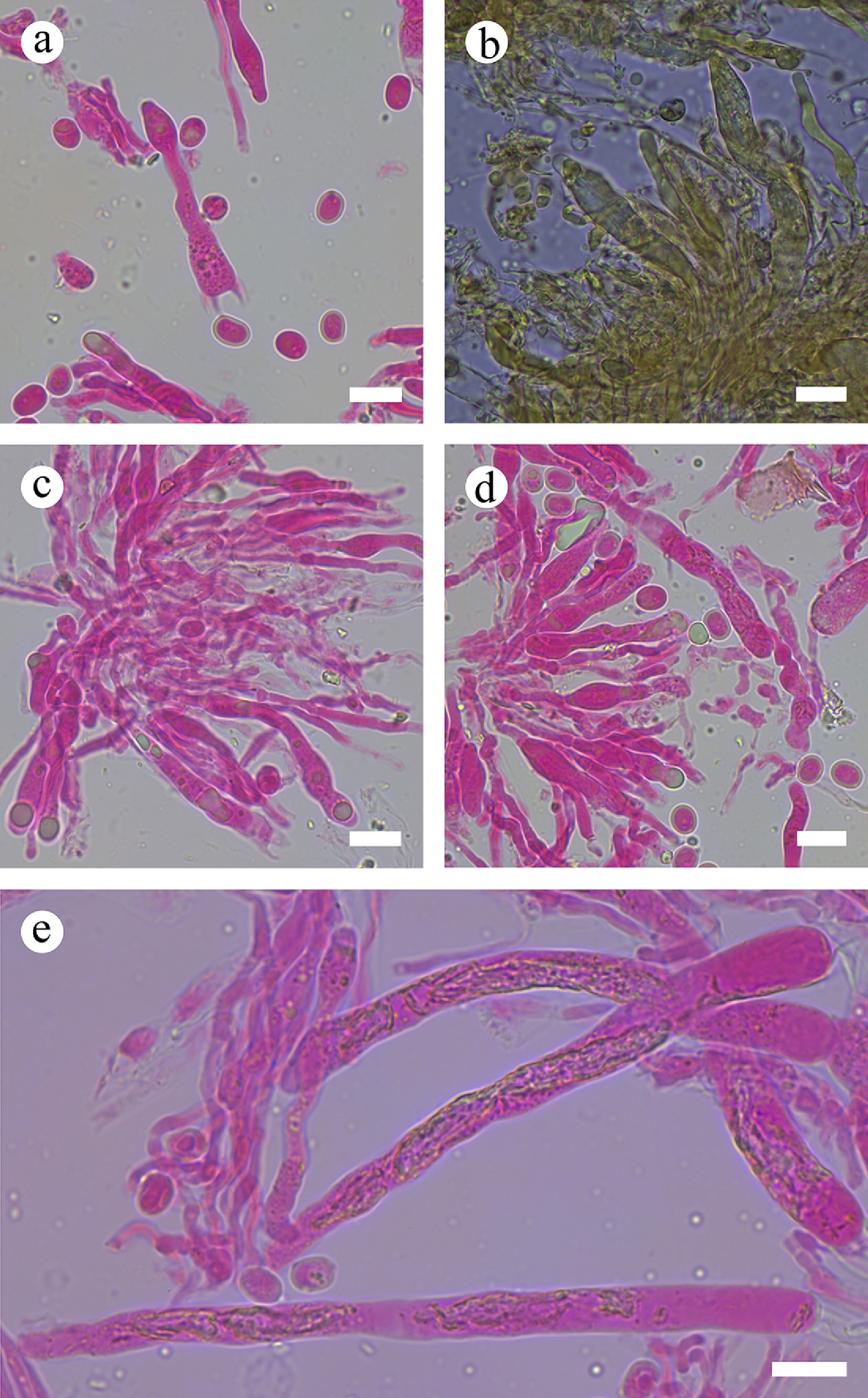
Figure 24.
Sections of hymenium of Gloiothele tuberculata (holotype). (a) Basidiospores and basidia. (b), (e) Gloeocystidia. (c), (d) Section of hymenium. Scale bars: (a)–(e) = 10 µm.
-

Figure 25.
Basidiomata and microscopic structures of Vararia asiana (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidia and basidioles. e Gloeocystidia. (f) Dichohyphae. (g) Section of the hymenium. Scale bars: a = 1 cm, b = 1 mm, c = 5µm, d–g = 10 µm.
-

Figure 26.
Sections of the hymenium of Vararia asiana (holotype). (a) Basidiospores. (b) Basidia. (c) Dichohyphae. (d) Basidioles. (e) Gloeocystidia. (f) Skeletal hyphae. Scale bars: (a)–(f) = 10 µm.
-
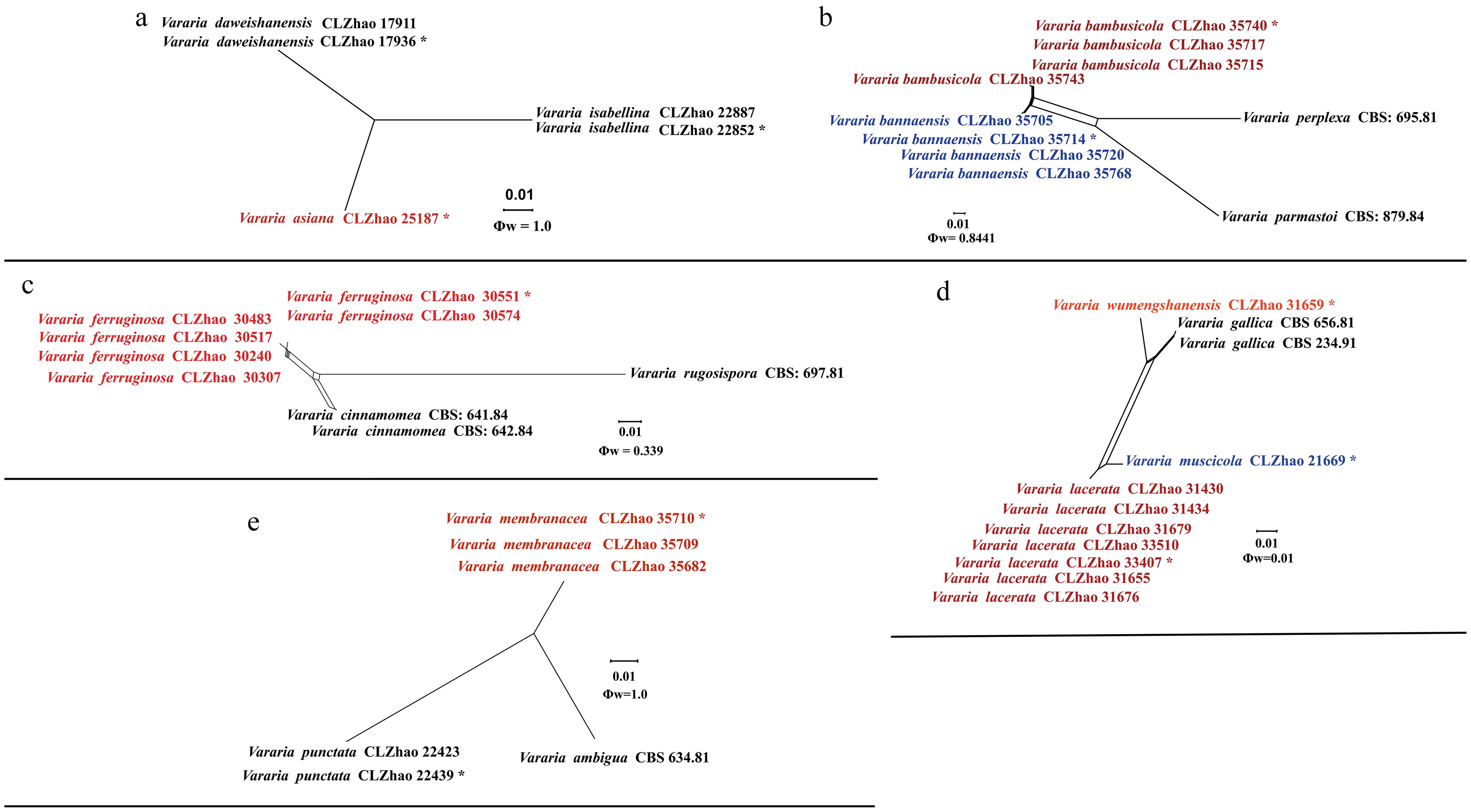
Figure 27.
Pairwise homoplasy index (PHI) test of the new species of Vararia and closely related species using both LogDet transformation and splits decomposition. PHI test results Φw ≤ 0.05 indicate that there is significant recombination within the dataset. The new taxa are in bold red type.
-

Figure 28.
Basidiomata and microscopic structures of Vararia bambusicola (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidia and basidioles. (e) Urniform gloeocystidia. (f) Mushroom-shaped gloeocystidia. (g) Fusiform gloeocystidia. (h) Dichohyphae. (i) Section of the hymenium. Scale bars: (a) = 1 cm, (b) = 1 mm, (c)–(i) = 10 μm.
-
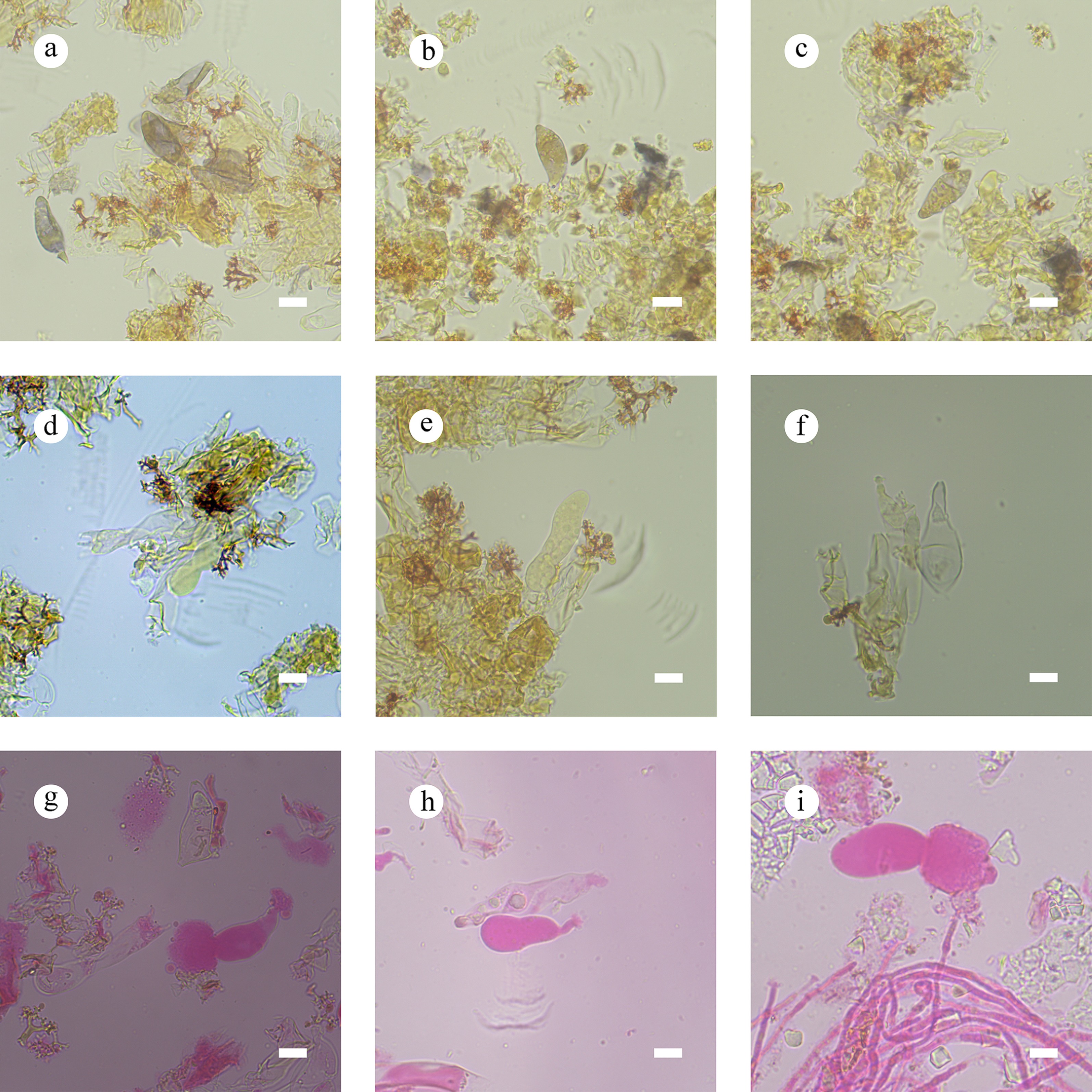
Figure 29.
Sections of the hymenium of Vararia bambusicola (holotype). (a)–(c) Basidiospores. (d) Basidia. (e) Basidioles. (f)–(i) Gloeocystidia. Scale bars: (a)–(i) = 10 µm.
-

Figure 30.
Basidiomata and microscopic structures of Vararia bannaensis (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (d) Basidia and basidioles. (e), (f) Gloeocystidia. (g) Section of the hymenium. Scale bars: (a) = 1 cm, (b) = 1 mm, (c) Basidiospores. Scale bars: (c)–(g) = 10 μm.
-

Figure 31.
Sections of hymenium of Vararia bannaensis (holotype). (a) Basidiospores. (b) Basidia. (c)–(e) Basidioles and gloeocystidia. (f) Dichohyphae. Scale bars: (a)–(f) = 10 µm.
-
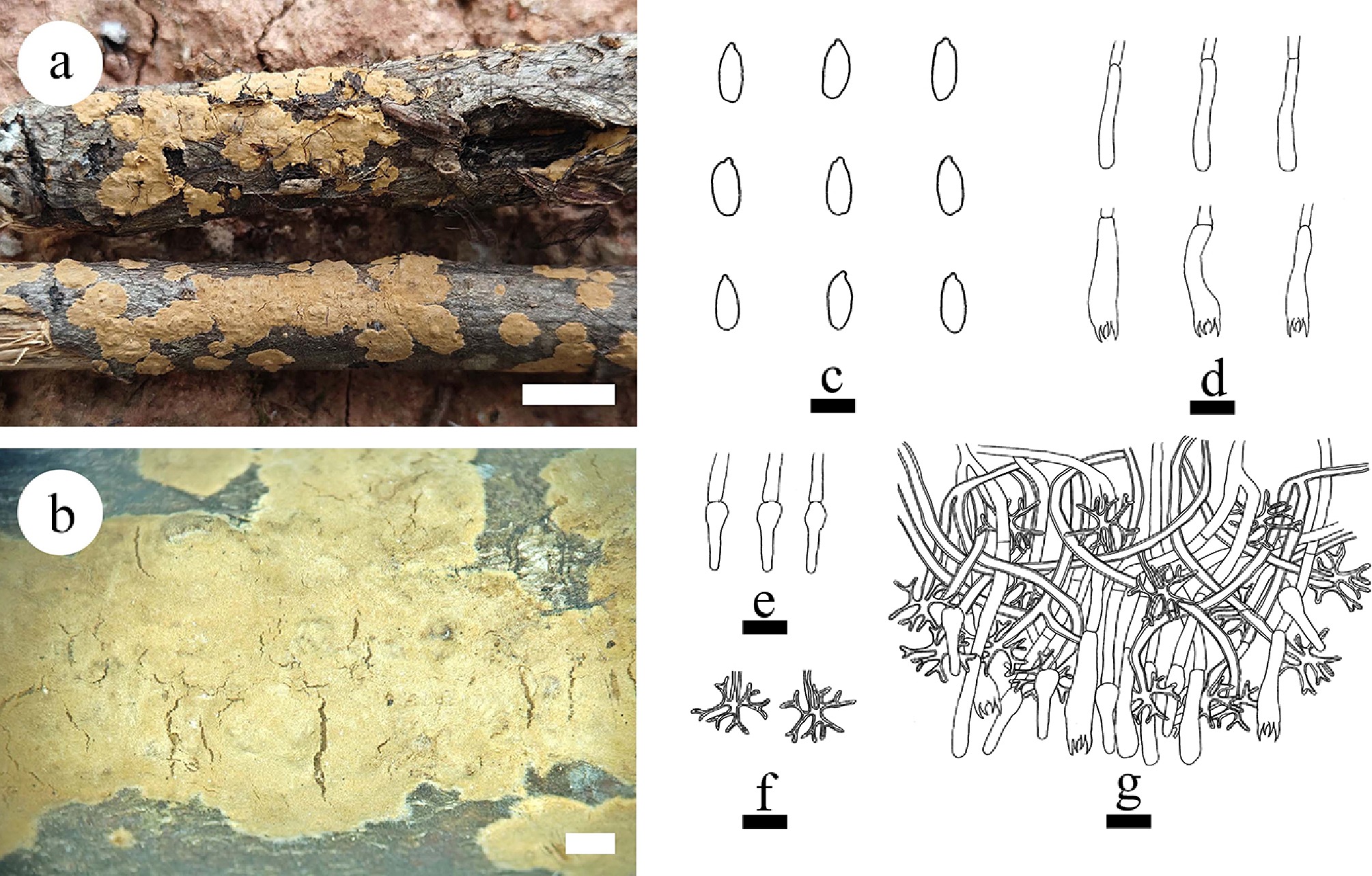
Figure 32.
Basidiomata and microscopic structures of Vararia ferruginosa (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidia and basidioles. (e) Gloeocystidia. (f) Dichohyphae. (g) Section of the hymenium. Scale bars: (a) = 1 cm, (b) = 1 mm, (c) = 5 µm; (d)–(g) = 10 µm.
-
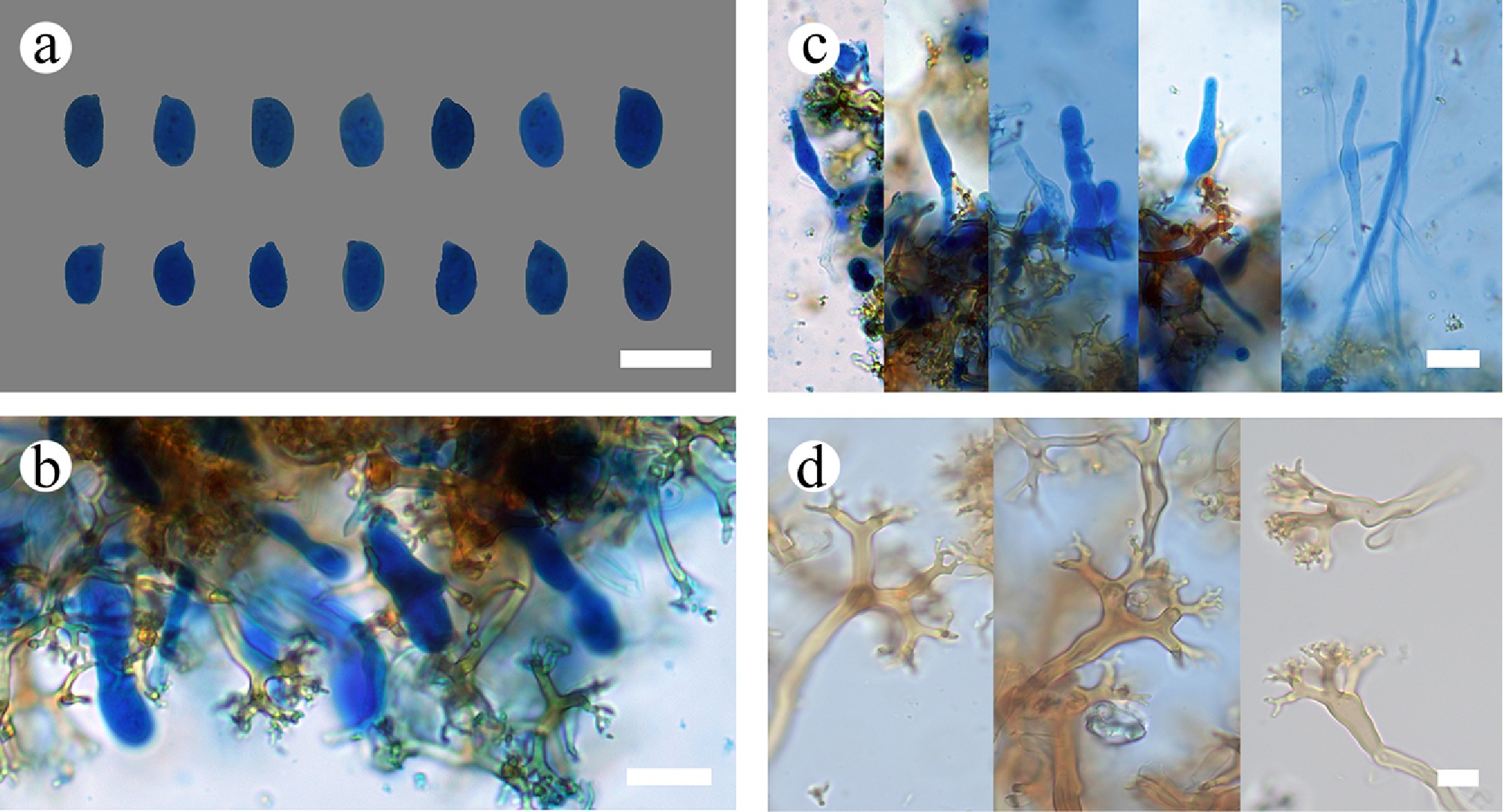
Figure 33.
Sections of the hymenium of Vararia ferruginosa (holotype). (a) Basidiospores. (b) Bbasidioles. (c) Gloeocystidia. (d) Dichohyphae. Scale bars: (a)–(d) = 10 µm.
-
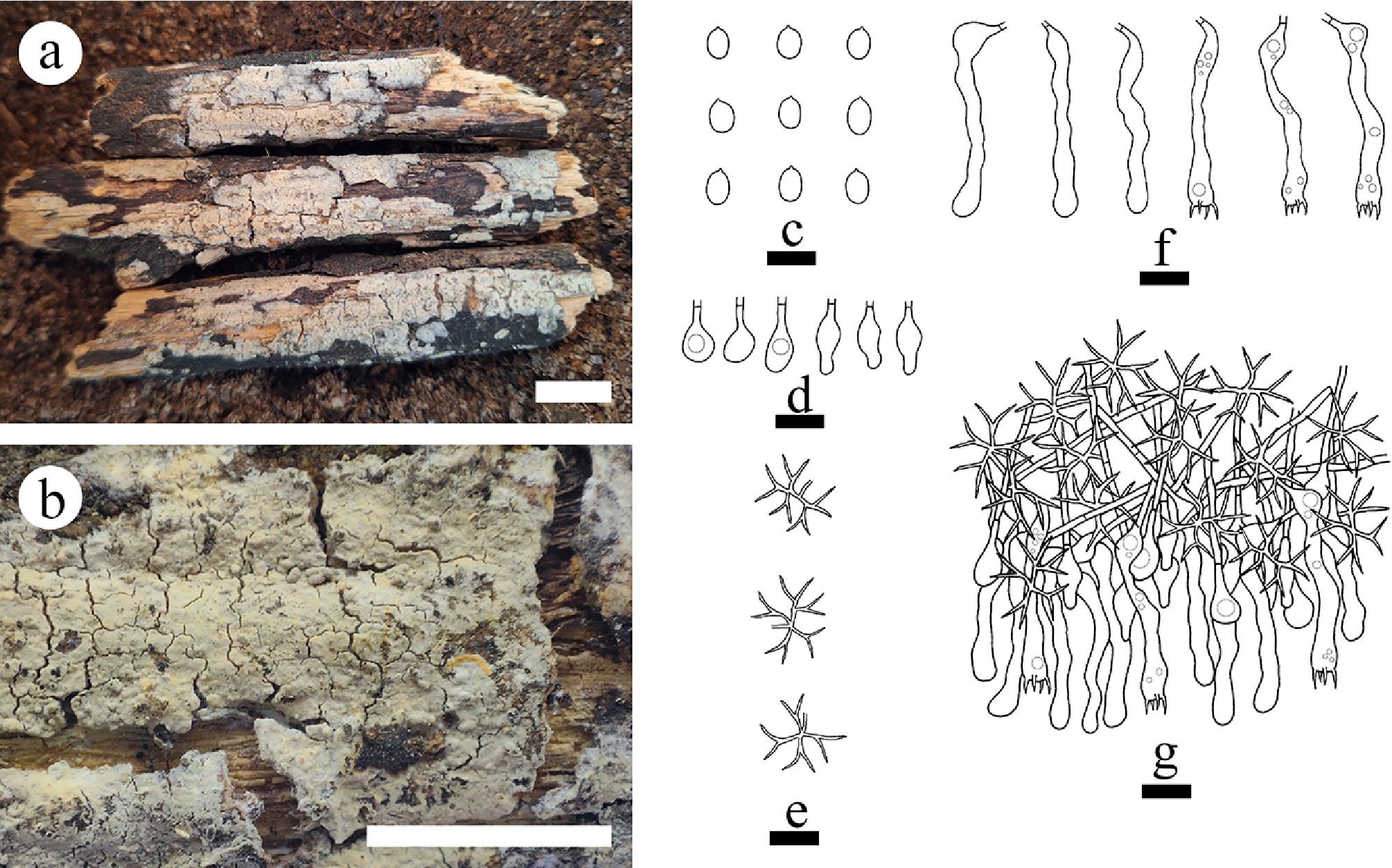
Figure 34.
Basidiomata and microscopic structures of Vararia lacerata (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidia and basidioles. (e) Gloeocystidia. (f) Section of the hymenium. (g) Dichohyphae. Scale bars: (a) = 1 cm, (b) = 1 mm, (c)–(g) = 10 µm.
-
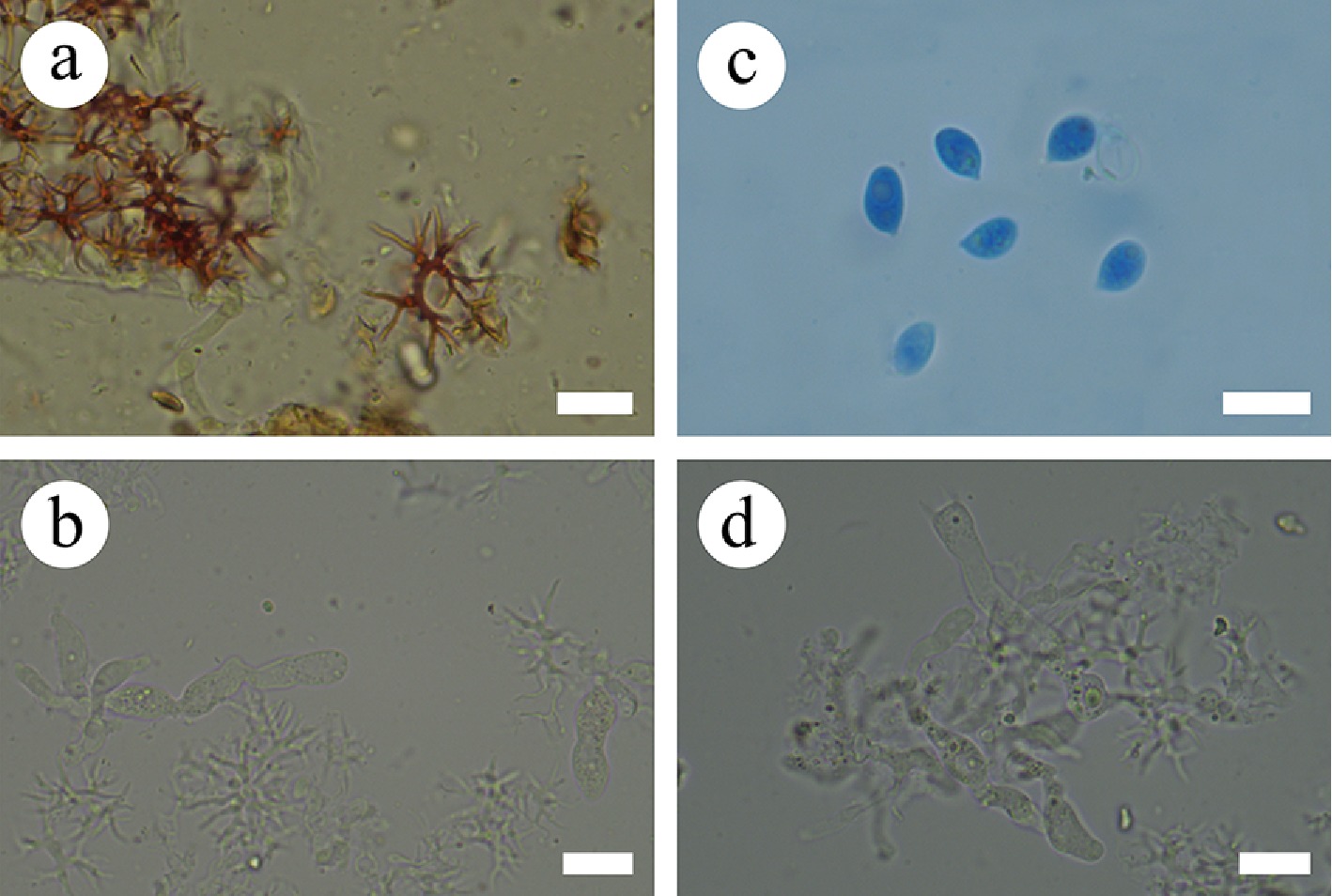
Figure 35.
Sections of the hymenium of Vararia lacerata (holotype). (a) Dichohyphae. (b) Gloeocystidia. (c) Basidiospores. (d) Basidia. Scale bars: (a)–(d) = 10 µm.
-

Figure 36.
Basidiomata and microscopic structures of Vararia membranacea (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidioles. (e) Gloeocystidia. (f) Dichohyphae. (g) Section of the hymenium. Scale bars: (a) = 1 cm, (b) = 1 mm, (c)–(g) = 10 μm.
-
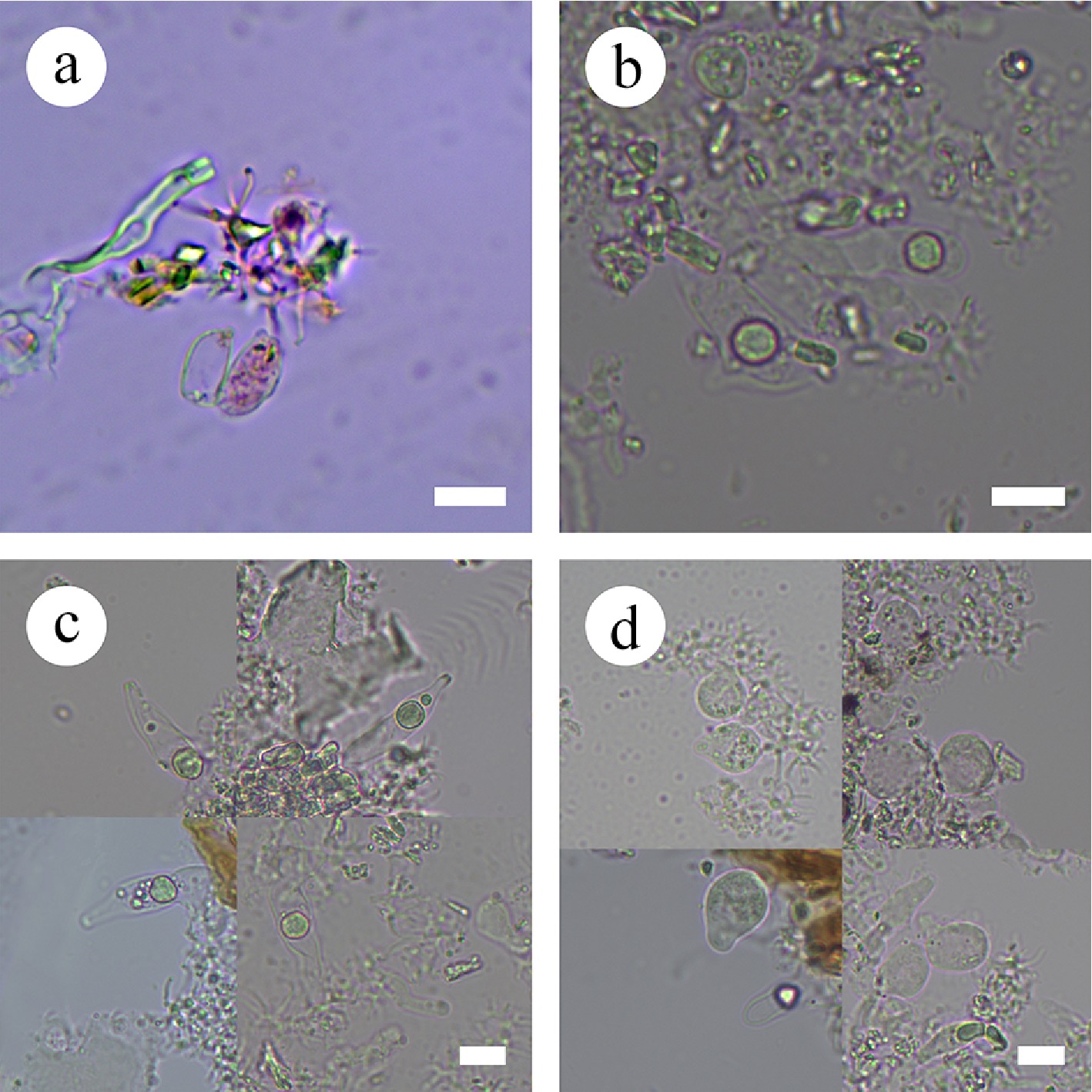
Figure 37.
Sections of the hymenium of Vararia membranacea (holotype). (a) Basidiospores. (b) Basidioles. (c), (d) Gloeocystidia. Scale bars: (a)–(d) = 10 µm.
-

Figure 38.
Basidiomata of and microscopic structures Vararia muscicola (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidioles. (e) Basidia. (f) Gloeocystidia. (g) Dichohyphae. (h) Section of the hymenium. Scale bars: (a) = 1 cm, (b) = 1 mm, (c) = 5 μm, (d)–(h) = 10 µm.
-
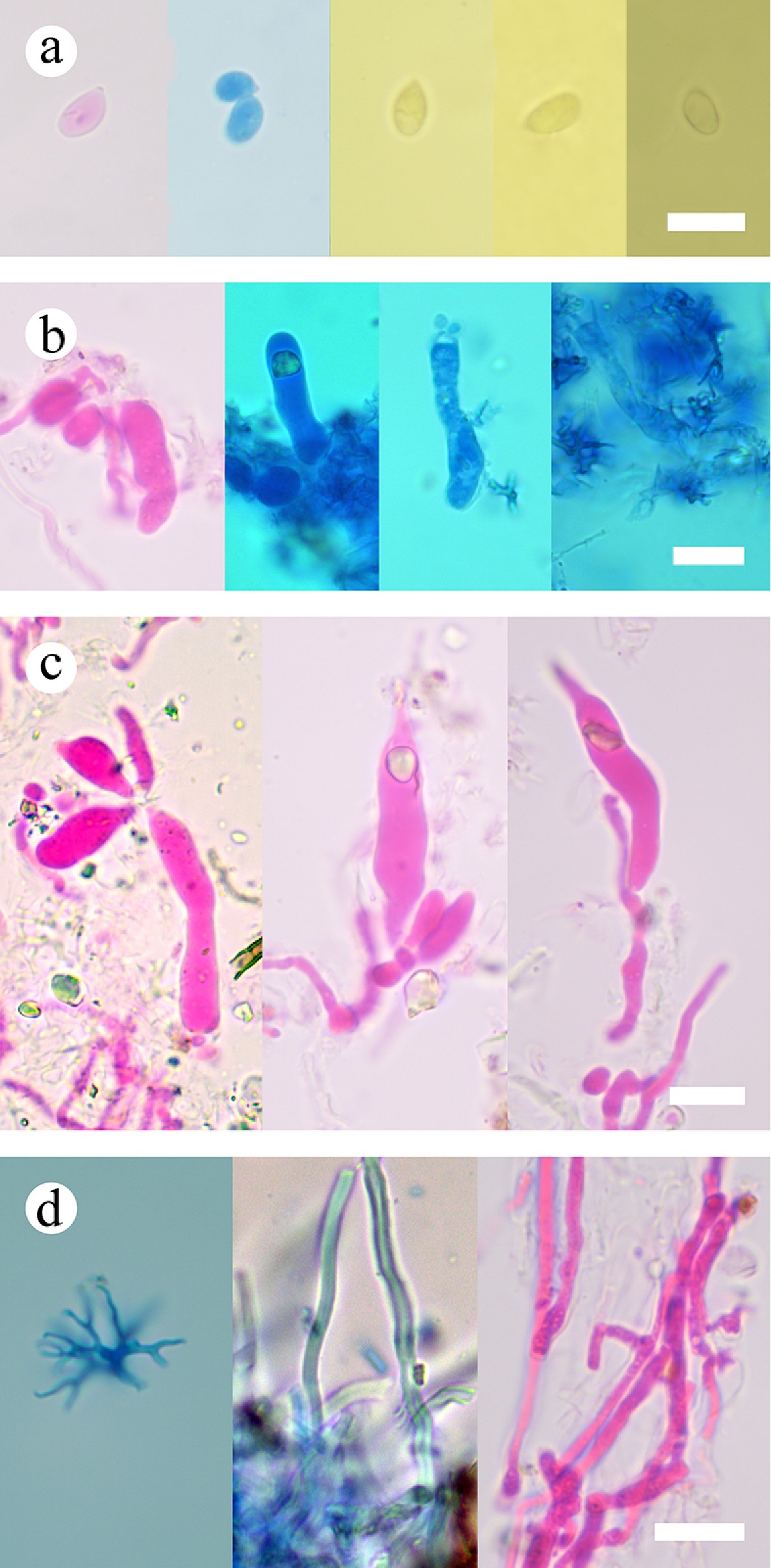
Figure 39.
Sections of the hymenium of Vararia muscicola (holotype). (a) Basidiospores. (b) Basidia and basidioles. (c) Gloeocystidia. (d) Hyphae. Scale bars: (a)–(d) = 10 µm.
-

Figure 40.
Basidiomata of and microscopic structures Vararia pingbianensis (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidia and basidioles. (e) Cystidia. (f) Dichohyphae hyphae. (g) Section of hymenium. Scale bars: (a) = 1 cm, (b) = 1 mm. Bars: (a) = 1 cm, (b) = 1 mm, (c) = 5 µm, (d)–(g) = 10 µm.
-

Figure 41.
Basidiomata and microscopic structures of Vararia wumengshanensis (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidia and basidioles. (e) Gloeocystidia. (f) Section of the hymenium. (g) Dichohyphae. Scale bars: (a) = 1 cm, (b) = 1 mm, (c)–(g) = 10 µm.
-
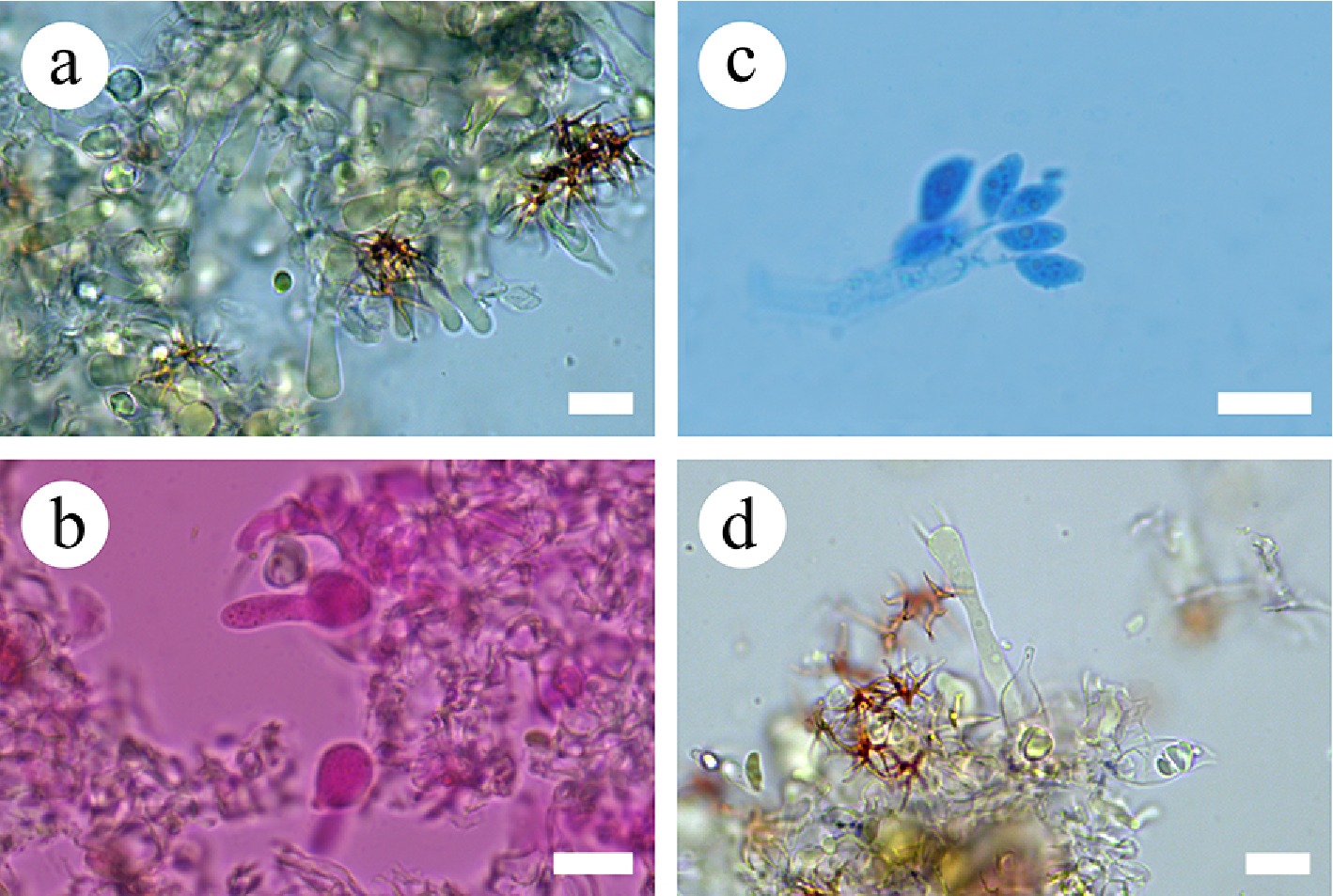
Figure 42.
Sections of the hymenium of Vararia wumengshanensis (holotype). (a) Section of the hymenium. (b) Gloeocystidia. (c) Basidiospores. (d) Basidia. Scale bars: (a)–(d) = 10 µm.
-

Figure 43.
Basidiomata and microscopic structures of Aleurodiscus yunnanensis (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidia and basidioles. (e) Clavate gloeocystidia. (f) Acanthocystidia. (g) Moniliform cystidia. (h) Section of hymenium. Scale bars: (a) = 1 cm, (b) = 1 mm, (c)–(h) = 10 µm.
-
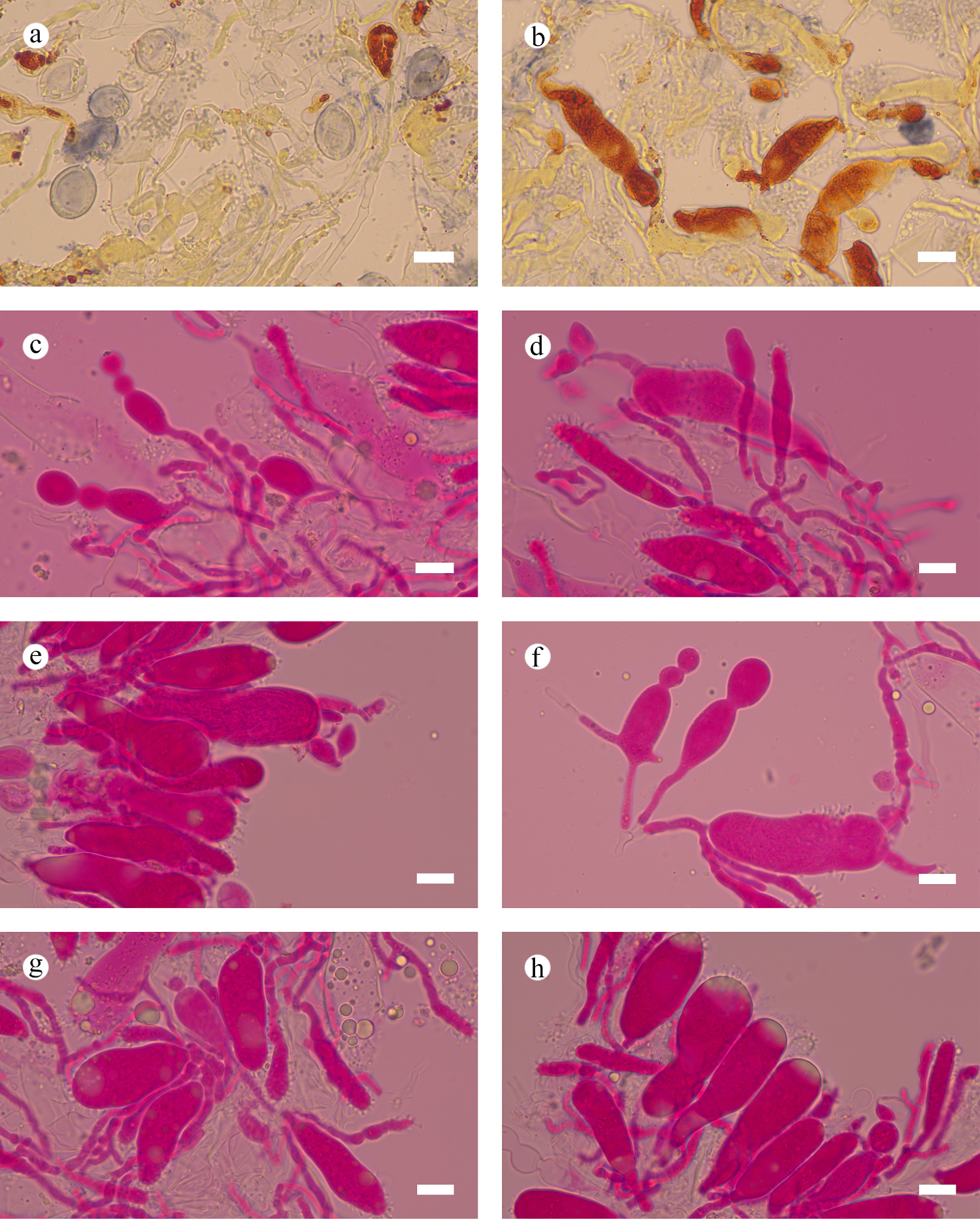
Figure 44.
Sections of hymenium of Aleurodiscus yunnanensis (holotype). (a) Basidiospores. (b), (g), (h) Clavate gloeocystidia and acanthocystidia. (c) Moniliform cystidia. (d)–(f) Basidia and basidioles. Scale bars: (a)–(h) = 10 µm.
-
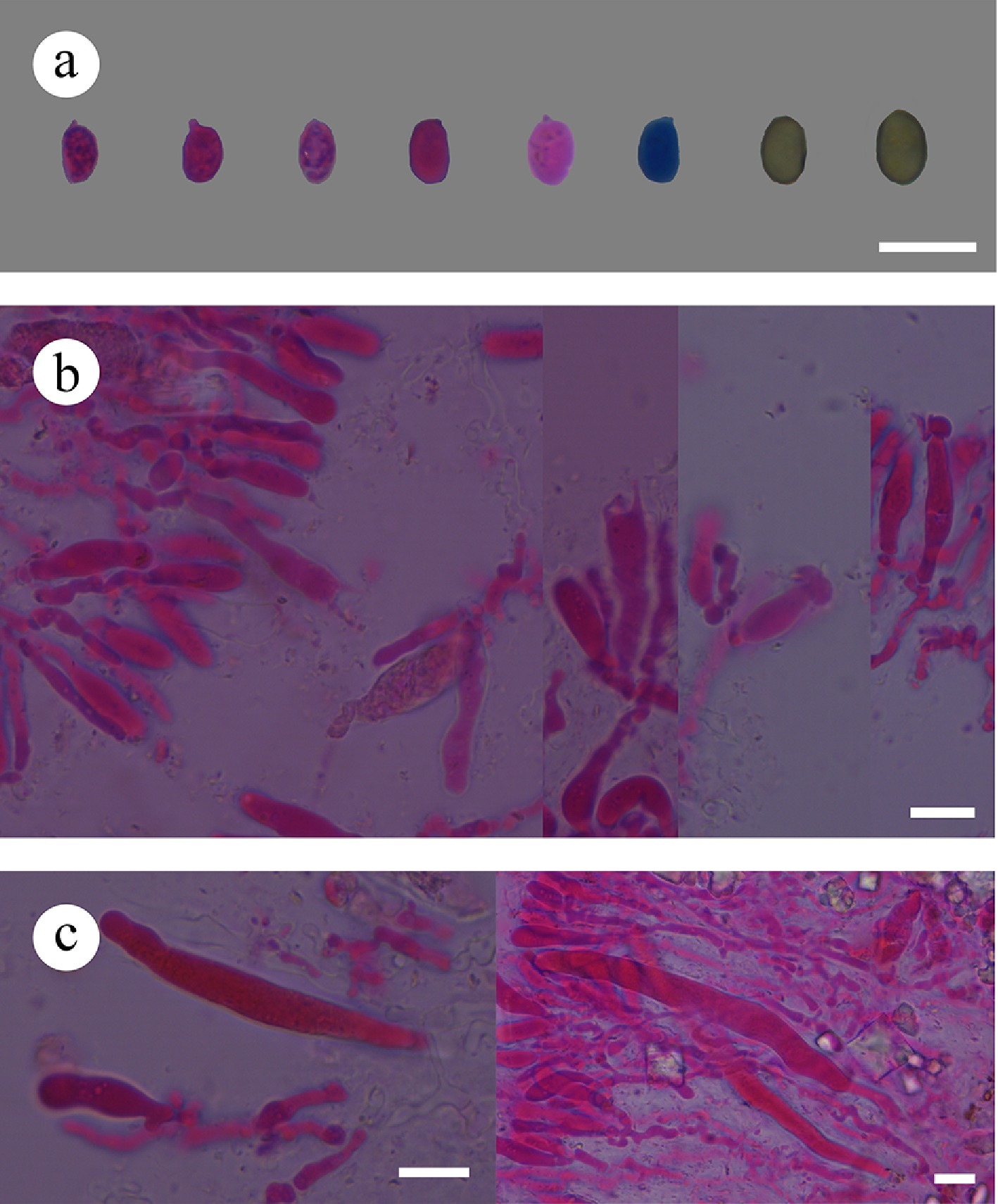
Figure 46.
Sections of hymenium of Confertotrama cremea (holotype). (a) Basidiospores. (b) Basidia and basidioles. (c) Cystidia. Scale bars: (a)–(c) = 10 µm.
-

Figure 45.
Basidiomata and microscopic structures of Confertotrama cremea (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidia and basidioles. (e) Halocystidia cystidia. (f) Cylindrical cystidia. (g) Tubular cystidia. (h) Section of the hymenium. Scale bars: (a) = 1 cm, (b) = 1 mm, (c) = 5 μm, (d)–(h) = 10 μm.
-

Figure 47.
Basidiomata and microscopic structures of Confertotrama yunnanensis (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidioles and basidia. (e), (f) Cystidia. (g) Hyphidia. (h) Section of hymenium. Scale bars: (a) = 1 cm, (b) = 2 mm, (c)–(h) = 10 µm.
-
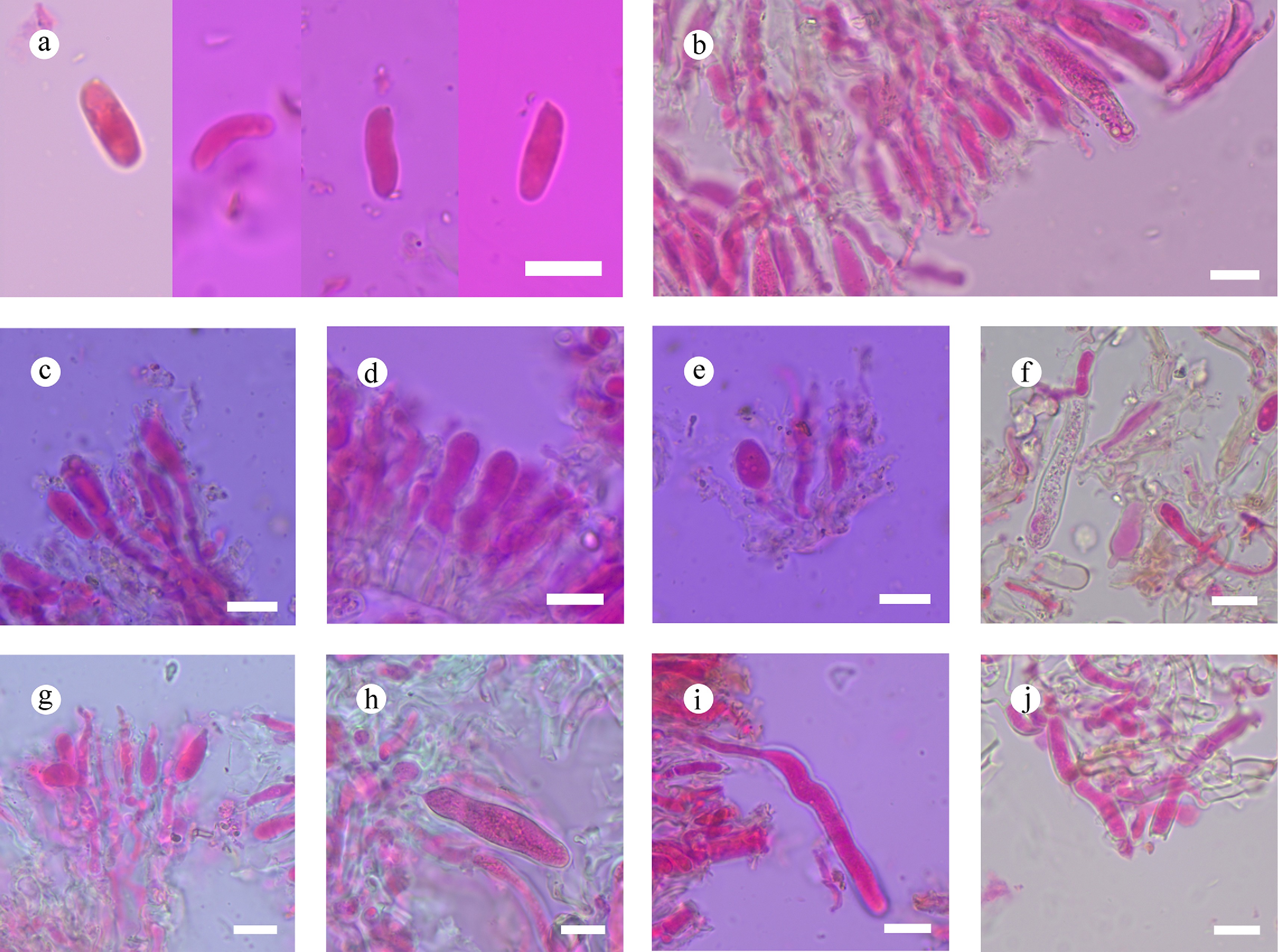
Figure 48.
Sections of hymenium of Confertotrama yunnanensis (holotype). (a) Basidiospores. (b) Section of hymenium. (c) Basidia and basidioles. (e)–(i) Cystidia. (j) Tramal hyphae. Scale bars: (a)–(j) = 10 µm.
-
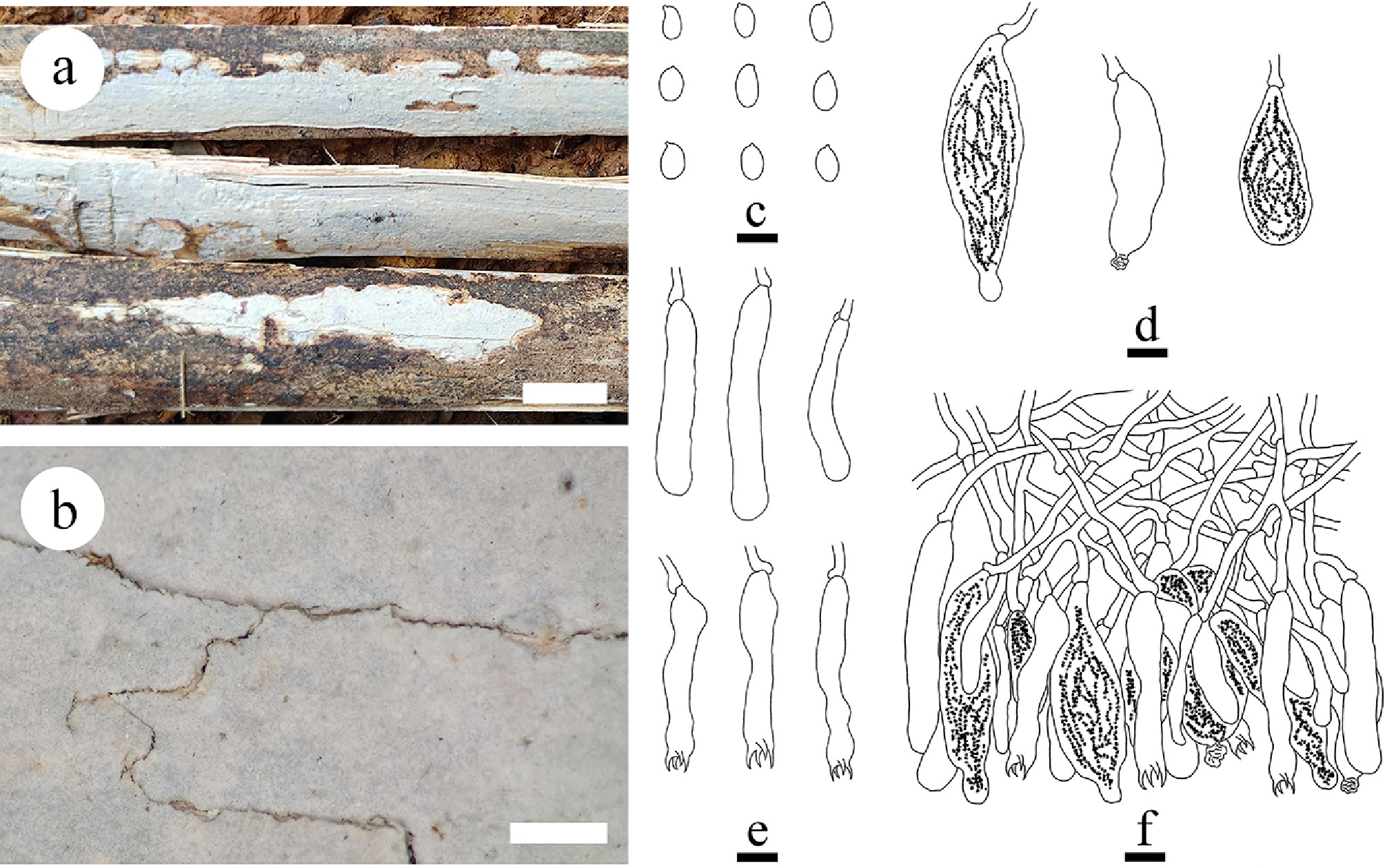
Figure 49.
Basidiomata and microscopic structures of Megalocystidium bambusinum (holotype). (a) Basidiomata on the substrate. (b) Section of hymenophore. (c) Basidiospores. (d) Basidioles and basidia. (e) Gloeocystidia. (f) Section of hymenium. Scale bars: (a) = 1 cm, (b) = 1 mm, (c)–(f) = 10 µm.
-
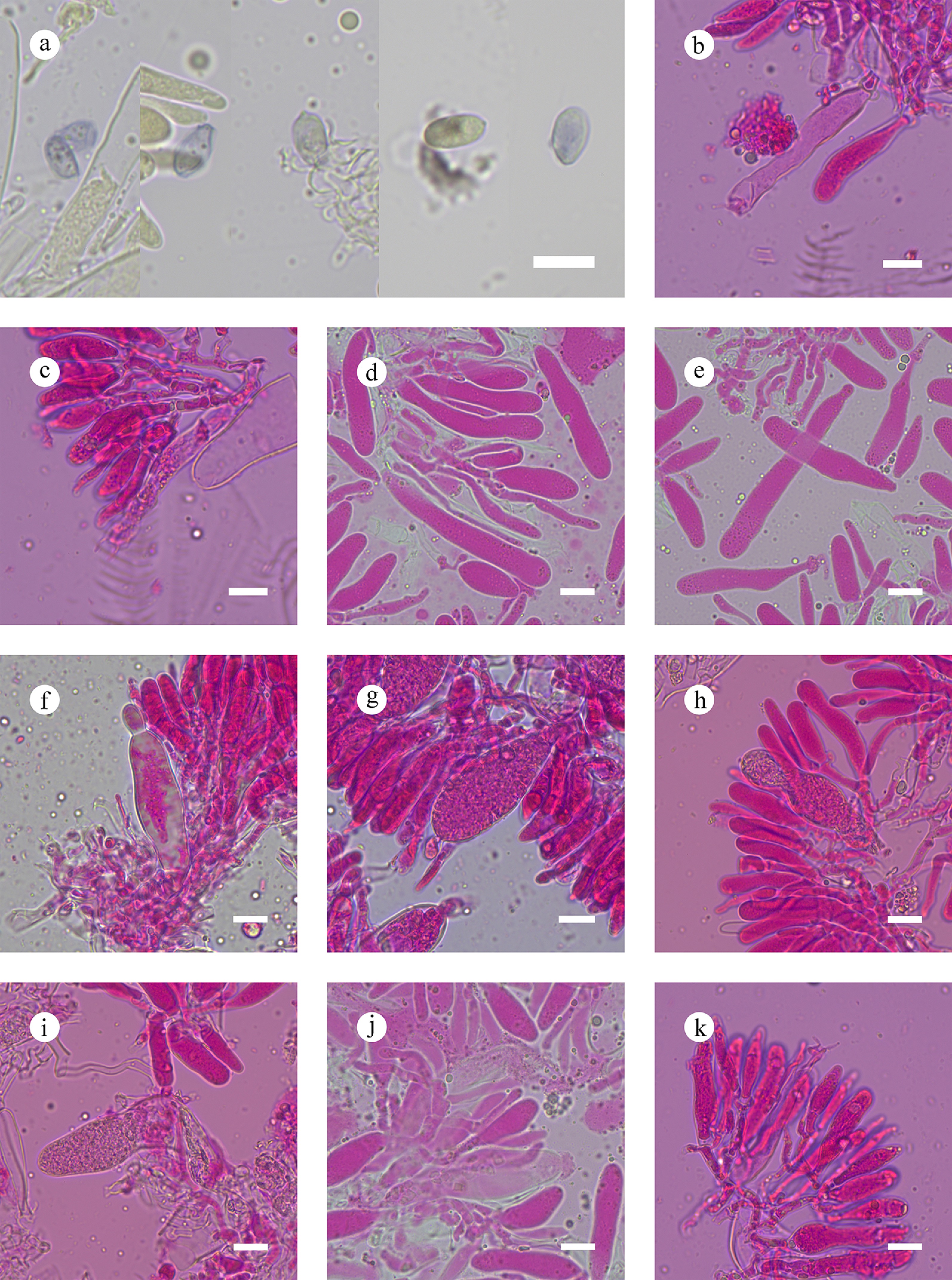
Figure 50.
Sections of hymenium of Megalocystidium bambusinum (holotype). (a) Basidiospores. (b), (c) Basidia. (d), (e) Basidioles. (f)–(j) Gloeocystidia. (k) Section of hymenium. Scale bars: (a)–(k) = 10 µm.
-

Figure 51.
Basidiomata and microscopic structures of Stereum convolutomarginatum (holotype). (a), (c) Basidiomata on the natural substrates. (b), (d) A character hymenophore. (e) Basidiospores. (f) Acanthocystidia. (g) Basidia and basidioles. (h) Section of hymenium. Scale bars: (a), (c) = 1 cm, (b), (d) = 1 mm, (e)–(h) = 10 µm.
-
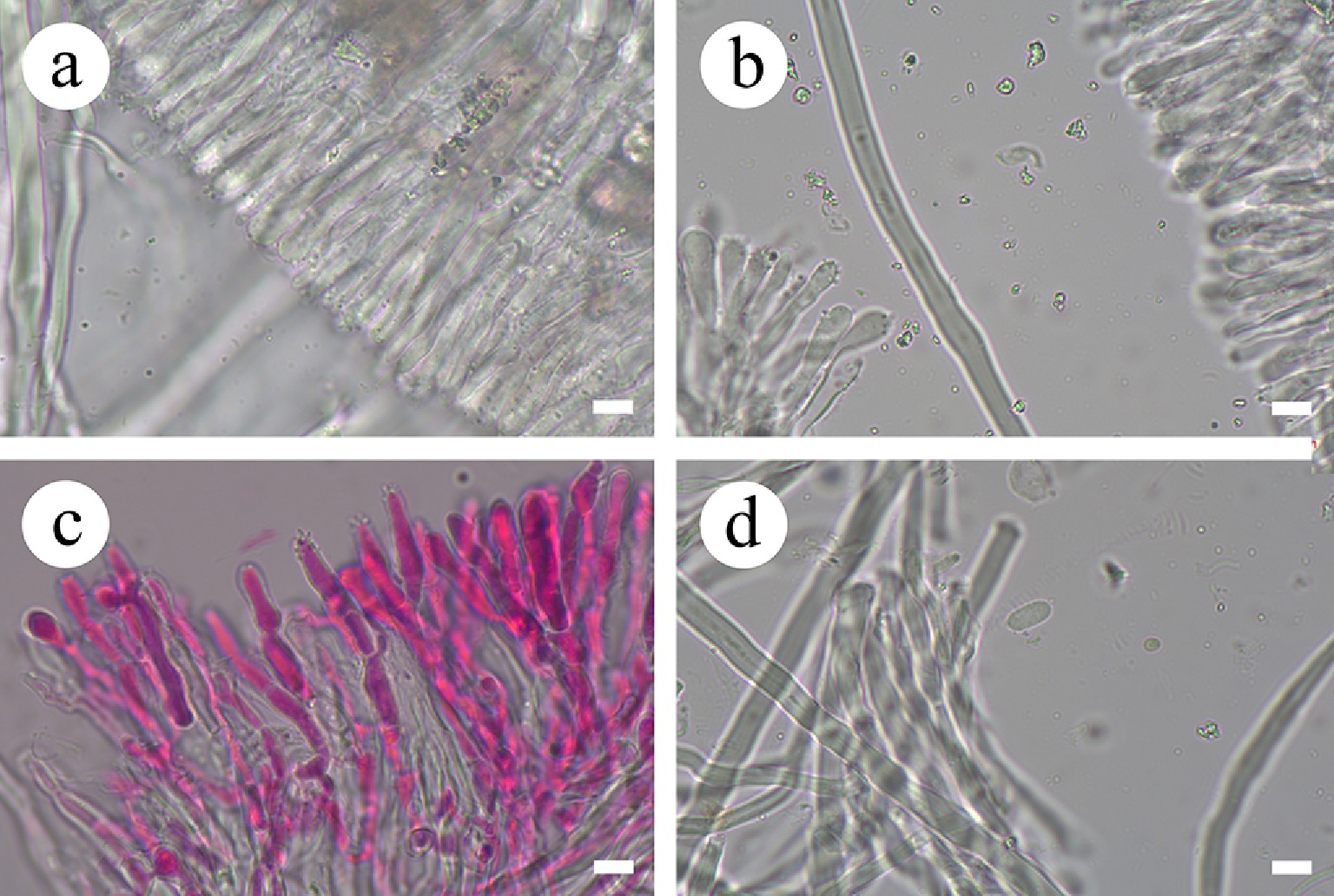
Figure 52.
Sections of hymenium of Stereum convolutomarginatum (holotype). (a), (c) Section of hymenium. (b) Skeletal hyphae. (d) Basidiospores. Scale bars: (a)–(d) = 10 µm.
-

Figure 53.
Basidiomata and microscopic structures of Stereum rigidohymeneum (holotype). (a), (b) Section of hymenophore. (c), (d) The back of the basidiomata. (e) Basidiospores. (f) Basidia and basidioles. (g) Acanthocystidia. (h) Section of hymenium. Scale bars: (a), (c) = 1 cm, (b), (d) = 1 mm, (e) = 5 µm, (f)–(h) = 10 µm.
-
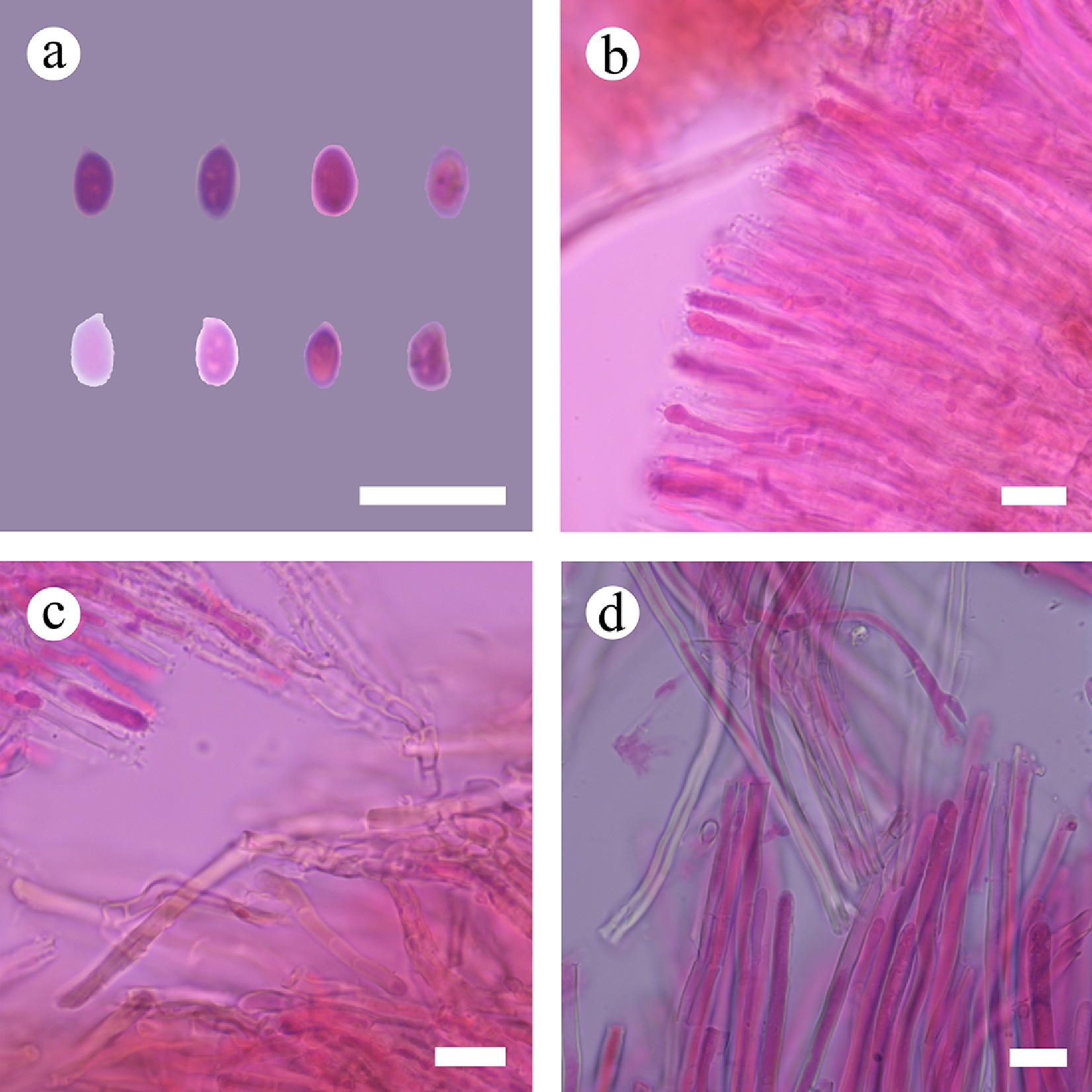
Figure 54.
Sections of hymenium of Stereum rigidohymeneum (holotype). (a) Basidiospores. (b), (c) Acanthocystidia. (d) Skeletal hyphae. Scale bars: (a)–(d) = 10 µm.
-

Figure 55.
Basidiomata and microscopic structures of Stereum yunnanense (holotype). (a), (b) Section of hymenophore. (c), (d) The back of the basidiomata. (e) Basidiospores. (r) Basidia and basidioles. (g) Cystidia. (h) Section of hymenium. Scale bars: (a), (c) = 1 cm, (b), (d) = 1 mm, (e)–(h) = 10 µm.
-
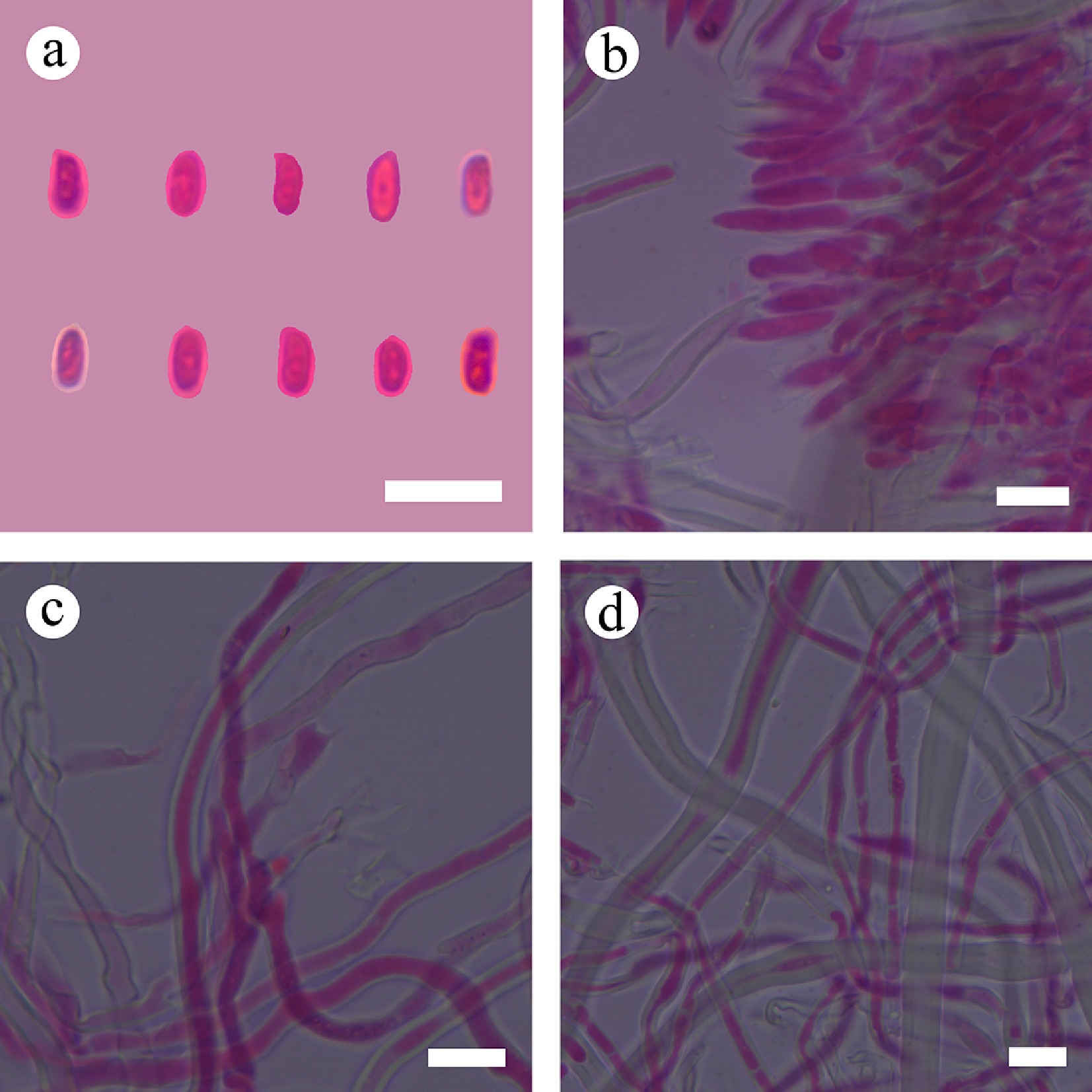
Figure 56.
Sections of hymenium of Stereum yunnanense (holotype). (a) Basidiospores. (b) Section of hymenium. (c) Basidia. (d) Skeletal hyphae. Scale bars: (a)–(d) = 10 µm.
-
Outline of the order Russulales Order Russulales Kreisel ex P.M. Kirk, P.F. Cannon & J.C. David 2001 Family Albatrellaceae Nuss 1980 Albatrellopsis Teixeira 1993 Albatrellus Gray 1821 (= Ovinus (Lloyd) Torrend 1920, Polyporus sect. Ovinus Lloyd 1911) (Note 1) Byssoporia M.J. Larsen & Zak 1978 (Note 2) Leucogaster R. Hesse 1882 Leucophleps Harkn. 1899 (= Cremeogaster Mattir. 1924, Leucophleps Harkn. 1899) Mycolevis A.H. Sm. 1965 Polyporoletus Snell 1936 (Note 3) Scutiger Paulet 1808 Family Aleurocystidiellaceae Y.L. Deng & C.L. Zhao, fam. nov. (Note 4) Aleurocystidiellum P.A. Lemke 1964 (Note 5) Family Auriscalpiaceae Maas Geest. 1963 Artomyces Jülich 1982 (Note 6) Auriscalpium Gray 1821(= Pleurodon Quél. ex P. Karst. 1881) (Note 7) Dentipratulum Domański 1965 (Note 8) Gloiodon P. Karst. 1879 (= Leaia Banker 1906, Sclerodon P. Karst. 1889) (Note 9) Lentinellus P. Karst. 1879 ( =Hemicybe P. Karst. 1879) Stalpersia Parmasto 2001 Family Bondarzewiaceae Kotl. & Pouzar 1957 (= Hybogasteraceae Jülich 1982) (Note 10) Amylaria Corner 1955 (Note 11) Amylonotus Ryvarden 1975 (Note 12) Amylosporus Ryvarden 1973 (= Rigidoporopsis I. Johans. & Ryvarden 1979) (Note 13) Bondarzewia Singer 1940 (= Hybogaster Singer 1964) (Note 14) Heterobasidion Bref. 1888 (= Spiniger Stalpers 1974, Spongioides Lázaro Ibiza 1916) (Note 15) Laurilia Pouzar 1959 (Note 16) Lauriliella Nakasone & S.H. He 2017 (Note 17) Stecchericium D.A. Reid 1963 (Note 18) Family Echinodontiaceae Donk 1961 (= Amylostereaceae Boidin, Mugnier & Canales 1998) Amylostereum Boidin 1958 (= Lloydellopsis Pouzar 1959, Trichocarpus P. Karst. 1889) (Note 19) Echinodontiellum S.H. He & Nakasone 2017 (Note 20) Echinodontium Ellis & Everh. 1900 (= Hydnofomes Henn. 1900, Hydnophysa Clem. 1909) (Note 21) Larssoniporia Y.C. Dai, Jia J. Chen & B.K. Cui 2015 (Note 22) Subulicystidiella Y.L. Deng & C.L. Zhao gen. nov. (Note 23) Family Gloeocystidiellaceae Jülich 1982 Gloeocystidiellum Donk 1931 (Note 24) Family Gloeodontiaceae Y.L. Deng & C.L. Zhao fam. nov. (Note 25) Gloeodontia Boidin 1966 (Note 26) Family Hericiaceae Donk 1964 Dentipellicula Y.C. Dai & L.W. Zhou 2013 (Note 27) Dentipellis Donk 1962 (= Amylodontia Nikol. 1967) (Note 28) Hericium Pers. 1794 (= Creolophus P. Karst. 1879, Dryodon Quél. ex P. Karst. 1881, Friesites P. Karst. 1879, Hericium Schrank 1786, Hericius Juss. 1789, Manina Banker 1912, Martela Adans. 1763, Martella Endl. 1836, Medusina Chevall. 1826) (Note 29) Laxitextum Lentz 1956 (Note 30) Pseudowrightoporia Y.C. Dai, Jia J. Chen & B.K. Cui 2015 (Note 31) Wrightoporiopsis Y.C. Dai, Jia J. Chen & B.K. Cui 2015 (Note 32) Family Peniophoraceae Lotsy 1907 (= Lachnocladiaceae D.A. Reid 1965) (Note 33) Amylofungus Sheng H. Wu 1996 (Note 34) Asterostroma Massee 1889 (Note 35) Asterostromella Höhn. & Litsch. 1907 Baltazaria Leal-Dutra, Dentinger & G.W. Grif 2018 (Note 36) Dendrophora (Parmasto) Chamuris 1987 (Note 37) Dichostereum Pilát 1926 (Note 38) Duportella Pat. 1915 (Note 39) Entomocorticium H.S. Whitney, Bandoni & Oberw. 1987 (Note 40) Gloiothele Bres. 1920 (Note 41) Lachnocladium Lév. 1846 (= Eriocladus Lév. 1846, Stelligera R. Heim 1938, Stelligera R. Heim ex Doty 1948) (Note 42) Metulodontia Parmasto 1968 Parapterulicium Corner 1952 (Note 43) Peniophora Cooke 1879 (= Cryptochaete P. Karst. 1889, Gloeopeniophora Höhn. & Litsch. 1907, Sterellum P. Karst. 1889) (Note 44) Sceptrulum K.H. Larss. 2014 Scytinostroma Donk 1956 (= Licrostroma P.A. Lemke 1964 = Michenera Berk. & M.A. Curtis 1868) (Note 45) Vararia P. Karst. 1898 (= Denrophysellum Parmasto 1968) (Note 46) Vesiculomyces E. Hagstr. 1977 Family Russulaceae Lotsy 1907 Boidinia Stalpers & Hjortstam 1982 (Note 47) Gloeopeniophorella Rick 1934 (Note 48) Lactarius Pers. 1797 (= Agaricus subdiv. Galorrheus Fr. 1818, Arcangeliella Cavara 1900, Galorrheus (Fr.) Fr. 1825, Gastrolactarius R. Heim 1971, Gastrolactarius R. Heim ex J.M. Vidal 2005, Gloeocybe Earle 1909, Hypophyllum Earle 1909, Lactariella J. Schröt. 1889, Zelleromyces Singer & A.H. Sm. 1960) (Note 49) Lactifluus (Pers.) Roussel 1806 (= Lactariopsis Henn. 1901, Pleurogala Redhead & Norvell 1993) (Note 50) Multifurca Buyck & V. Hofst. 2008 (Note 51) Pseudoxenasma K.H. Larss. & Hjortstam 1976 (Note 52) Russula Pers. 1796 (= Bucholtzia Lohwag 1924, Cystangium Singer & A.H. Sm. 1960, Dixophyllum Earle 1909, Elasmomyces Cavara 1897, Gymnomyces Massee & Rodway 1898, Hypochanum Kalchbr. 1876, Lactarelis Earle 1909, Macowanites Kalchbr. 1882, Martellia Mattir. 1900, Omphalomyces Battarra ex Earle 1909, Phaeohygrocybe Henn. 1901, Russulina J. Schröt. 1889) Family Stereaceae Pilát 1930 (Note 53) Acanthobasidium Oberw. 1965 (Note 54) Acanthofungus Sheng H. Wu, Boidin & C.Y. Chien 2000 Acanthophysellum Parmasto 1967 (Note 55) Acanthophysium (Pilát) G. Cunn. 1963 Aleurobotrys Boidin 1986 Aleurodiscus Rabenh. ex J. Schröt. 1888 ( =Aleurobotrys Boidin 1985, Aleurodiscus Cooke 1885, Aleurodiscus Rabenh. 1874, Gloeomyces Sheng H. Wu 1996, Gloeosoma Bres. 1920, Nodularia Peck 1871) (Note 56) Aleuromyces Boidin & Gilles 2002 Amylohyphus Ryvarden 1978 Amylosporomyces S.S. Rattan 1977 (Note 57) Conferticium Hallenb. 1980 (Note 58) Confertotrama Nakasone & S.H. He 2025 (Note 59) Coniophorafomes Rick 1934 Dextrinocystidium Sheng H. Wu 1996 Gelatinostereum S.H. He, S.L. Liu & Y.C. Dai 2025 (Note 60) Gloeocystidiopsis Jülich 1982 (Note 61) Gloeomyces Sheng H. Wu 1996 (Note 62) Gloeosoma Bres. 1920 (Note 63) Megalocystidium Jülich 1978 (Note 64) Neoaleurodiscus Sheng H. Wu 2010 (Note 65) Scotoderma Jülich 1974 Stereodiscus Rajchenb. & Pildain 2021 (Note 66) Stereum Hill ex Pers. 1794 (= Haematostereum Pouzar 1959) (Note 67) Xylobolus P. Karst. 1881 (Note 68) Family Terrestriporiaceae Y.C. Dai, B.K. Cui, F. Wu, Y. Yuan & Jia J. Chen 2020 (Note 69) Terrestriporia Y.C. Dai, B.K. Cui, F. Wu, Y. Yuan & Jia J. Chen 2020 (Note 70) Family Wrightoporiaceae Jülich 1982 Wrightoporia Pouzar 1966 (Note 71) Russulales genera incertae sedis Dentipellopsis Y.C. Dai & L.W. Zhou 2013 (Note 72) Dichantharellus Corner 1966 Dichopleuropus D.A. Reid 1965 Gloeoasterostroma Rick 1938 Gloeohypochnicium (Parmasto) Hjortstam 1987 (Note 73) Haloaleurodiscus N. Maek., Suhara & K. Kinjo 2005 (Note 74) Laeticutis Audet 2010 Neoalbatrellus Audet 2010 (Note 75) Perplexostereum Ryvarden & S. Tutka 2014 (Note 76) Polypus Audet 2010 Scopulodontia Hjortstam 1998 Scytinostromella Parmasto 1968 (= Confertobasidium Jülich 1972) Xeroceps Audet 2010 -
Order Families Number of genera Russulales Albatrellaceae 8 Aleurocystidiellaceae 1 Auriscalpiaceae 6 Bondarzewiaceae 8 Echinodontiaceae 5 Gloeocystidiellaceae 1 Gloeodontiaceae 1 Hericiaceae 6 Peniophoraceae 17 Russulaceae 7 Stereaceae 23 Terrestriporiaceae 1 Wrightoporiaceae 1 Incertae sedis 13 Table 1.
Number of genera within families of Russulales.
-
Gene
regionPrimer
pairsSequence (5′−3′) Annealing temperature (°C) Ref. ITS ITS5 GGAAGTAAAAGTCGTAACAAGG 55 [ 56] ITS4 TCCTCCGCTTATTGATATGC nLSU LR0R ACCCGCTGAACTTAAGC 48 [ 61] LR7 TACTACCACCAAGATCT mtSSU MS1 CAGCAGTCAAGAATATTAGTCAATG 52 [ 56] MS2 GCGGATTATCGAATTAAATAAC tef1- α ef1-983F GCYCCYGGHCAYCGTGAYTTYAT 60 [ 62] ef1-2218R ATGACACCRACRGCRACRGTYTG rpb1 rpb1-Af GARTGYCCDGGDCAYTTYGG 52 [ 59, 60] rpb1-Cf CCNGCDATNTCRTTRTCCATRTA rpb2 brpb2-6F TGGGGYATGGTNTGYCCYGC 52 [ 58] brpb2-7.1R CCCATRGCYTGYTTMCCCATDGC Table 2.
Gene regions and respective primers used in the study.
-
Order/family Species Sample no. GenBank accession no. Ref. ITS nLSU rpb2 tef1- α Agaricales Asterophora lycoperdoides CBS 170.86 AF357037 AF223190 DQ367431 DQ367424 [ 77] Gymnopilus picreus ZRL2015011 LT716066 KY418882 KY419027 KY419077 [ 77] Amylocorticiales Amylocorticium cebennense HHB-2808 GU187505 GU187561 GU187770 GU187675 [ 78] Anomoloma myceliosum MJL-4413 GU187500 GU187559 GU187766 GU187677 [ 78] Atheliales Athelia arachnoidea CBS 418.72 GU187504 GU187557 GU187769 GU187672 [ 78] Leptosporomyces raunkiaerii HHB-7628 GU187528 GU187588 GU187791 [ 78] Auriculariales Auricularia heimuer Xiaoheimao LT716074 KY418890 KY419035 KY419083 [ 78] Exidia sp . PBM2527 DQ241774 AY700191 DQ408144 [ 78] Boletales Coniophora arida FP104367 GU187510 GU187573 GU187775 GU187684 [ 78] Gomphidius roseus MB 95-038 DQ534570 DQ534669 GU187818 GU187702 [ 78] Dacrymycetales Calocera cornea AFTOL 438 AY789083 AY701526 AY536286 AY881019 [ 78] Dacryopinax spathularia AFTOL 454 AY854070 AY701525 AY857981 AY881020 [ 78] Geastrales Geastrum taylorii OSC59760 DQ218520 DQ219060 DQ219235 [ 79] Schenella pityophila OSC59743 DQ218519 DQ219057 DQ219232 [ 79] Gomphales Clavariadelphus truncatus OSC67280 AY574649 DQ219064 DQ219240 [ 78] Kavinia alboviridis 0102140 AY574692 DQ219073 DQ219250 [ 78] Hymenochaetales Fomitiporia mediterranea AFTOL688 AY854080 AY684157 AY803748 AY885149 [ 78] Phellinus hartigii Dai 11766 KT203287 KT203308 KJ651721 [ 78] Hysterangiales Aroramyces gelatinosporus H4010 DQ218524 DQ218941 DQ219118 [ 78] Chondrogaster pachysporus OSC49298 DQ218538 DQ218958 DQ219136 [ 78] Jaapiales Jaapia argillacea CBS252.74 GU187524 GU187581 GU187788 GU187711 [ 78] Polyporales Fomitopsis pinicola AFTOL 770 AY854083 AY684164 AY786056 AY885152 [ 78] Polyporus squamosus Cui 10595 KU189778 KU189809 KU189988 KU189925 [ 78] Russulales/Albatrellaceae Albatrellus ovinus PS11795 MW269673 MW269685 MW290304 MW290320 [ 26] Byssoporia terrestris Hjm18172 EU118608 EU118608 [ 45] Polyporoletus sublividus JA 030918 DQ389663 DQ389663 [ 80] –/Aleurocystidiellaceae Aleurocystidiellum bernicchiae MR12636 MT831037 MT831017 [ 46] Aleurocystidiellum bernicchiae SPG3217 MT831016 [ 46] Aleurocystidiellum subcruentatum He2886 KU559341 KU574847 KU992720 [ 81] Aleurocystidiellum subcruentatum HHB-17353-sp KU559360 KU574818 [ 81] Aleurocystidiellum tsugae He4025 KY706211 KY706223 [ 81] Aleurocystidiellum tsugae He4024 KY706210 KY706222 [ 81] –/Auriscalpiaceae Auriscalpium vulgare HKAS93484 MK211170 KY485984 KY495319 KY474614 Unpublished Dentipratulum bialoviesense GG1645 AF506389 AF506389 [ 26] Lentinellus cochleatus KGN960928 AF506417 AF506417 [ 26] –/Bondarzewiaceae Bondarzewia occidentalis HHB-14803 KM243329 KM243332 KX066163 KX066142 [ 46] Heterobasidion annosum Korhonen 06129/6 KJ583211 KJ583225 KF006499 KX252741 [ 46] Laurilia sulcata He20120916-7 KY172894 KY172909 [ 82] Lauriliella taxodii FP-105464-Sp KY172896 KY172912 [ 82] –/Echinodontiaceae Amylostereum chailletii NH8031 AF506406 AF506406 [ 26] Echinodontiellum japonicum Dai 7378 KY172887 KY172902 [ 82] Echinodontium tinctorium HHB-12866-Sp KY172888 KY172903 MH550371 [ 26] Larssoniporia tropicalis Ryvarden 45363 KJ513294 KJ807089 [ 44] Subulicystidiella murina CLZhao 30728 PV771055 PX418375 Present study Subulicystidiella murina CLZhao 35801* PV441140 PV441154 Present study –/Gloeocystidiellaceae Gloeocystidiellum clavuligerum He3376 KY860377 KY860434 Unpublished Gloeocystidiellum granulatum He4301 KY860391 KY860449 Unpublished Gloeocystidiellum membranaceum CLZhao 37038 PV940928 PX070092 PX432797 PX439082 Present study Gloeocystidiellum porosellum Hjm 8851 AY048878 AY048878 Unpublished Gloeocystidiellum porosum HHB-15589-Sp MK625627 MK625555 MN031003 Unpublished Gloeocystidiellum punctatum CLZhao 20755* PP356586 PP785346 Present study Gloeocystidiellum purpureum Wu9310-45 AF441338 AF441338 [ 26] –/Gloeodontiaceae Gloeodontia columbiensis NH 11118 AF506444 AF506444 [ 26] Gloeodontia pyramidata Ryvarden 15502 AF506446 AF506446 [ 26] Gloeodontia sinensis CLZhao 34748 PV147171 PV185857 PV400175 Present study Gloeodontia subasperispora GB/KHL8695 AF506404 AF506404 [ 26] Gloeodontia discolor KHL 10099 AF506445 AF506445 [ 26] Gloeodontia eriobotryae Dai 12080 JQ349116 JQ349103 [ 30] Gloeodontia yunnanensis CLZhao 10504 MN908252 MN908254 [ 83] –/Hericiaceae Dentipellis fragilis Dai14767 MH085943 MH085958 Unpublished Hericium americanum DAOM21467 AF506458 AF506458 [ 26] Laxitextum bicolor Dai14056 KY860393 KY860451 [ 82] –/Peniophoraceae Asterostroma muscicola He4397 MK625630 MK625563 MN030965 [ 82] Baltazaria galactina He4999 MK625618 MK625547 MN030977 [ 84] Dichostereum boidinii He4410 MH538315 MH538331 MH550361 [ 43] Gloiothele lamellosa CBS 404.83 AF506487 AF506487 [ 26] Lachnocladium schweinfurthianum KM49740 MH260033 MH260051 [ 85] Peniophora quercina CBS 408.50 MH856688 MH868205 [ 84] Scytinostroma portentosum EL 11-99 AF506470 AF506470 [ 26] Vararia trinidadensis CBS:650.84 MH873495 MH873495 [ 84] –/Russulaceae Lactarius torminosus CBS 197.72 MH860447 MH872175 [ 84] Multifurca ochricompacta JJ2010.08 (PC0723658) MH063879 MH063844 MH061176 [ 86] Russula delica FH12-272 KF432955 KR364224 KR364340 [ 61] –/Stereaceae Acanthobasidium delicatum CBS 233.86 MH861948 MH873638 [ 84] Aleurobotrys botryosus DAOM211598 AF506398 AF506398 [ 26] Aleurodiscus amorphus Ghobad-Nejhad2464 KU559342 KU574832 KU992717 [ 81] Confertotrama rugulosa LodgeSJ 110.1 AF506441 AF506441 [ 87] Gelatinostereum phlebioides He4492 MW533096 MW528942 Unpublished Gloeosoma vitellinum 646cc MT831039 MT831019 [ 46] Megalocystidium leucoxanthum HK9808 AF506420 AF506420 [ 26] Stereodiscus antarcticus MR11265 MT831048 MT831028 [ 46] Stereum hirsutum He3504 MK625629 MK625557 MN031010 Unpublished Xylobolus frustulatus He2231 MH121216 KU574825 KU992704 [ 81] –/Terrestriporiaceae Terrestriporia alba Dai 18548 MT068564 MT068560 MW290307 MW290324 [ 21] Terrestriporia alba Dai 18556 MT068565 MT068561 MW290308 MW290325 [ 21] –/Wrightoporiaceae Wrightoporia avellanea LR 41710 AF506488 AF506488 [ 26] Wrightoporiaceae Wrightoporia subavellanea Dai 11484 KJ513295 KJ807085 [ 26] Sebacinales Sebacina sp. AFTOL 1517 DQ911617 DQ521412 [ 78] Tremellodendron pallidum AFTOL 699 DQ411526 AY745701 DQ408132 DQ029196 [ 78] Sordariales Neurospora crassa OR74A HQ271348 AF286411 AF107789 XM959775 [ 78] Thelephorales Boletopsis leucomelaena PBM2678 DQ484064 DQ154112 GU187820 GU187763 [ 78] Thelephora ganbajun ZRL20151295 LT716082 KY418908 KY419043 KY419093 [ 78] Table 3.
Taxa used in molecular clock analysis.
-
Node Means of stem age (Mya) 95% HPD (Mya) posterior probabilities A: Peniophoraceae/Echinodontiaceae 150.57 113.56–190.2 B: Gloeocystidiellaceae 163.59 125.46–202.89 C: Gloeodontiaceae 178.7 139.36–218.13 D: Auriscalpiaceae 174.07 128.22–221.01 E: Gloeocystidiellaceae/Russulaceae 133.39 79.96–186.84 F: Bondarzewiaceae/Stereaceae 179.84 126.6–228.84 G: Wrightoporiaceae 200.67 150.99–248.34 H: Hericiaceae/Aleurocystidiellaceae 140.94 87.46–196.71 I: Albatrellaceae/Terrestriporiaceae 127.75 71.29–188.91 C1: Hymenochaetales 142.43 135.31–150.67 C2: Agaricales 109.02 101.26–117.85 C3: Ascomycota/Basidiomycota 486.42 413.94–727.32 Table 4.
Estimated divergence time of each node.
-
Key to 25 species of Vararia s.l. in China 1 Generative hyphae with clamp connections 2 – Generative hyphae with simple septa 8 2 Basidia with 2 sterigmata 3 – Basidia with 4 sterigmata 4 3 Subcylindrical to fusiform basidiospores (12–17 × 4.5–5.5 µm), slightly thick-walled gloeocystidia two types: subglobose (15–30 × 6–8 µm), and subcylindrical or gradually narrower (25–40 × 4.5–6 µm) Vararia amphithallica – Fusiform to cylindrical basidiospores (18–22 × 6–7.2 µm), thick-walled, ventricose, gloeocystidia with an apical papilla (20–40 × 9–12 µm) V. bispora 4 Thin to thick-walled generative hyphae, subcylindrical basidia (26–46 × 5–8 µm), allantoid basidiospores (9–13 × 3.5–5 µm), ellipsoid to ovoid to subcylindrical gloeocystidia (9–23× 7–10.5 µm) V. daweishanensis – Thin-walled generative hyphae 5 5 Gloeocystidia two types: thin-walled, subglobose (11–23 × 6–12 µm), and subulate (25.5–43 × 7–11 µm), ellipsoid to broadly ellipsoid basidiospores (5–10 × 3–7 µm) V. fissurata – Gloeocystidia one type 6 6 Basidiospores < 5 µm wide V. investiens – Basidiospores > 5 µm wide 7 7 Broadly ellipsoid basidiospores (12–16 × 9.5–13 µm), clavate basidia (70–110 × 10–16 µm), clavate gloeocystidia (50–100 × 4–9 µm) V. montana – Spherical basidiospores (8–10 × 7.5–8.5 µm), cylindrical to clavate basidia (40–45 × 6–7.5 µm), subcylindrical to fusiform gloeocystidia (48–80(–105) × 7–11(–14) µm) V. sphaericospora 8 Dichohyphae distinctly thick-walled 9 – Dichohyphae thin-walled or slightly thick-walled 20 9 Generative hyphae thin- to slightly thick-walled 10 – Generative hyphae thin-walled 14 10 Gloeocystidia one type 11 – Gloeocystidia two or three types 12 11 Gloeocystidia clavate to cylindrical, thick-walled (12–21 × 5–9 µm), subcylindrical basidia with 4 sterigmata (11–25 × 4–7 µm), ellipsoid basidiospores (6–10 × 4–6 µm), white to cream hymenial surface V. punctata – Gloeocystidia spindle to subcylindrical (38–47 × 8–13 μm), basidiospores sub-fusiform to navicular (9–13 × 5–8 µm), subcylindrical basidia with four sterigmata (33–39 × 7–9 μm), spindle to subcylindrical gloeocystidia (38–47 × 8–13 μm), cream to isabelline to slightly brown hymenial surface V. isabellina 12 Gloeocystidia three types: subcylindrical (16.5–58.5 × 4–10 µm), fusiform (18.5–43.5 × 7–9 µm), and tapering (27.5–42 × 5.5–9 µm), slightly thick-walled, ellipsoid basidiospores (5.9–11.5 × 4.7–8.6 µm), cylindrical basidia (17.5–32 × 5–9.5 µm), thin- to slightly thick-walled V. yunnanensis – Thin-walled ellipsoid basidiospores 13 13 Gloeocystidia two types: elliptical to ovoid (5.8–16 × 3.5–7 µm), subulate (16.5–27 × 4–7 µm), subcylindrical basidia with 4 sterigmata (13–23.5 × 3–4.5 µm), ellipsoid basidiospores (3.5–5.5 × 2.5–3.5 µm), buff to ochraceous hymenial surface V. fragilis – Gloeocystidia two types: thick-walled, fusiform (28.5–50 × 6–12.5 µm), subglobose (11–27 × 7–11 µm), ellipsoid basidiospores (7.6–10.8 × 5.7–7.8 µm), subclavate to subcylindrical basidia (23–46 × 5–8 µm) V. yaoshanensis 14 Thick-walled fibriform skeletal hyphae absent 15 – Thick-walled fibriform skeletal hyphae present 16 15 Hymenial surface white to cream, gloeocystidia ventricose (16–19.5 × 5.5–7.5 µm) and obclavate (15–18 × 5.5–7 µm), subcylindrical to subclavate basidia (33.5–40 × 4.5–6 µm), subcylindrical to narrowly ellipsoid basidiospores (7.5–9.5 × 4–5 µm) V. wumengshanensis – Hymenial surface white to slightly cream to slightly greyish, gloeocystidia urniform (10–18 × 7–10 µm) and fusiform (26.1–36.3 × 4.2–7.9 µm), ellipsoid basidiospores (8.1–9.2 × 4.8–6.9 µm) V. membranacea 16 Gloeocystidia two types: subcylindrical to fusiform (24–40 × 4–8 µm), cylindrical to subclavate (12–18 × 6–7 µm), fusiform to subcylindrical basidia with 4 sterigmata (14–39 × 5–7 µm), ellipsoid basidiospores (5.8–8.9 × 3.5–5.5 µm), white to cream hymenial surface V. muscicola – Gloeocystidia one type 17 17 Basidia clavate 18 – Basidia fusiform to subcylindrical 19 18 Basidia larger (25–35 × 6–7 μm), hymenial surface white to slightly pink, gloeocystidia subulate (17–35 × 6–7 μm), sub-fusiform to navicular basidiospores (6–11 × 4–6 µm) V. sinensis – Basidia smaller (11–17.5 × 2–4 μm), hymenial surface white to cream, gloeocystidia subglobose, and clavate to fusiform (6.5–16 × 3–5 µm), ellipsoid basidiospores (3.5–5.5 × 2.5–4 µm) V. lincangensis 19 Basidia subcylindrical (20–29 × 6–7.5 µm), hymenial surface cream to yellowish, gloeocystidia fusiform to subcylindrical (15–21 × 5–7 µm), broadly ellipsoid basidiospores (2.5–4 × 1.5–2.5 µm) V. asiana – Basidia fusiform to subcylindrical (15–28 × 3–5.3 µm), hymenial surface ferruginous to yellowish-brown, gloeocystidia obclavate (14–25 × 3–6 µm), basidiospores ellipsoid (5.9–8.8 × 3.1–5.1 µm) V. ferruginosa 20 Thick-walled fibriform skeletal hyphae present 21 – Thick-walled fibriform skeletal hyphae absent 22 21 Gloeocystidia three types: urniform (16.3–28 × 10–14 µm), mushroom-shaped (21.3–37 × 12.7–13 µm), fusiform (35.4–47.3 × 7.8–14.1 µm), cylindrical to clavate basidia (29–57 × 8.5–12.3 µm), cylindrical to fusiform basidiospores (22.1–25.1 × 8.7–10.1 µm) V. bambusicola – Gloeocystides absent, oblong to subellipsoid basidiospores (9–13 × 5–7.2 µm), subcylindrical basidia (45–65 × 8–10 µm) V. cinnamomea 22 Rose to orange subfusiform basidiospores ((14–)16–19(–21.5) × 4.2–6 µm), cylindrical basidia (30–53 × 6.5–7.5 µm), thick-walled, subcylindrical gloeocystides (50–65 × 6–7(–8.5) µm) V. breviphysa – Basidiospores colorless 23 23 Gloeocystidia subcylindrical to fusiform (26–40 × 4.5–9 µm), cylindrical basidiospores (6–8 × 2–3 µm), cylindrical basidia (30–40 × 4–5 µm) V. racemosa – Gloeocystidia two types 24 24 Hymenial surface cream to buff, gloeocystidia two types: urniform (12.5–16.5 × 3.5–5.5 µm), capitate (10.5–13 × 5–6 µm); subcylindrical to subclavate basidia (34.5–37.5 × 4–5.5 µm), broadly ellipsoid basidiospores (6–7.5 × 3.5–5 µm) V. lacerata – Hymenial surface white to pinkish buff to cinnamon buff, gloeocystidia two types: urniform (9–20 × 8–14 µm), subcylindrical to fusiform (23–42 × 6–10 µm), subclavate basidia (20.5–26 × 5.5–7 µm), fusiform to ellipsoid basidiospores (11.8–16.1 × 5.6–7.7 µm) V. bannaensis
Figures
(56)
Tables
(6)