-
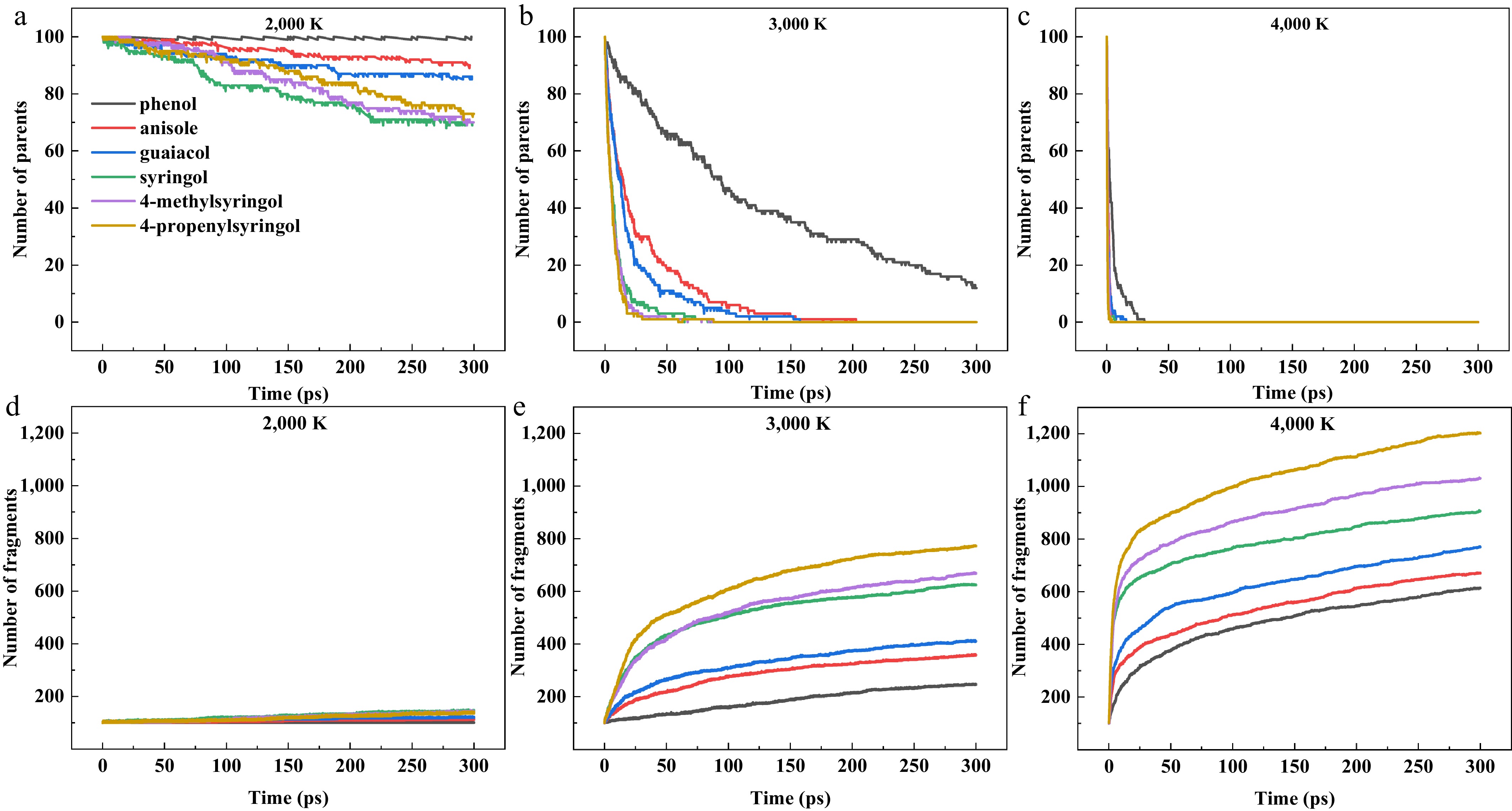
Figure 1.
(a)–(c) Time evolution of parent molecule and fragment. (d)–(f) Quantities for six model compounds at pyrolysis temperatures of 2,000, 3,000, and 4,000 K.
-
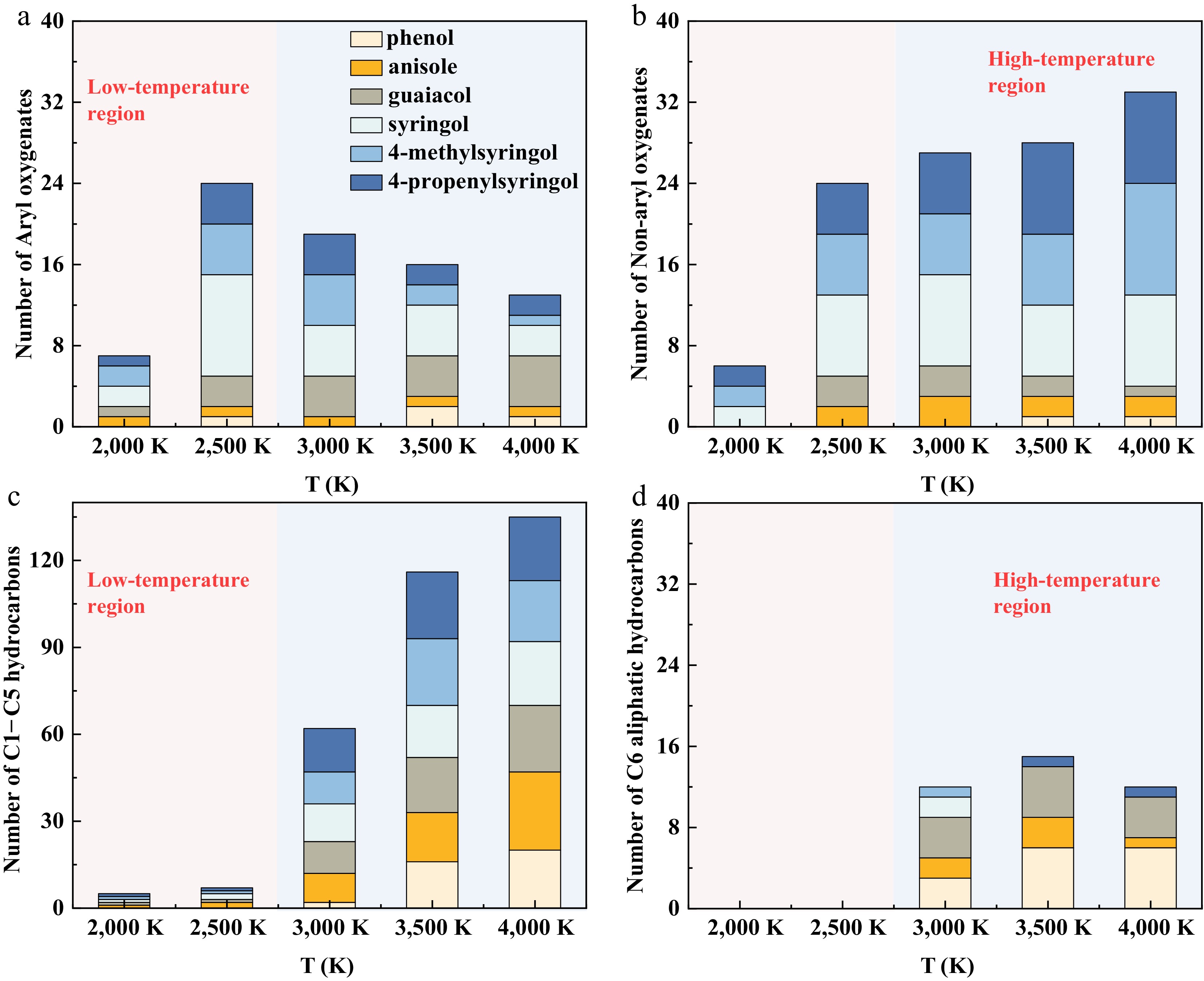
Figure 2.
Distribution of pyrolysis products for six model compounds at five temperatures. (a) Aryl oxygenates. (b) Non-aryl oxygenates. (c) C1−C5 hydrocarbons. (d) C6 aliphatic hydrocarbons.
-
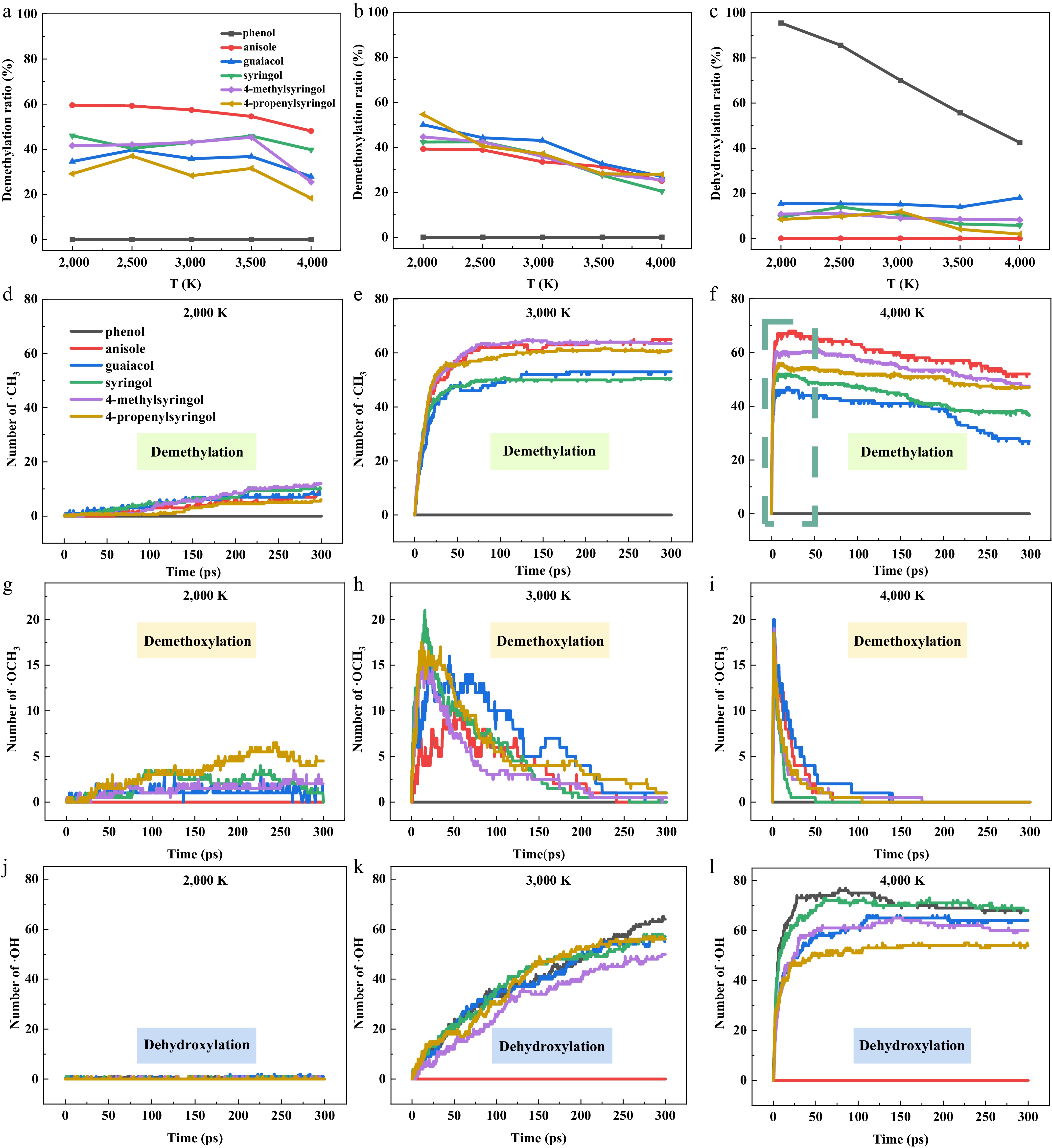
Figure 3.
Temperature dependence of initial pyrolysis reaction branching ratios and key radical numbers for six model compounds. (a)–(c) Branching ratios of the initial pyrolysis reaction pathways. (d)–(l) The number of ·CH3, ·OCH3, and ·OH radicals obtained at 2,000, 3,000, and 4,000 K. The number of ·CH3 and ·OCH3 radicals for the syringol derivatives (syringol, 4-methylsyringol, and 4-propenylsyringol) was divided by 2.
-
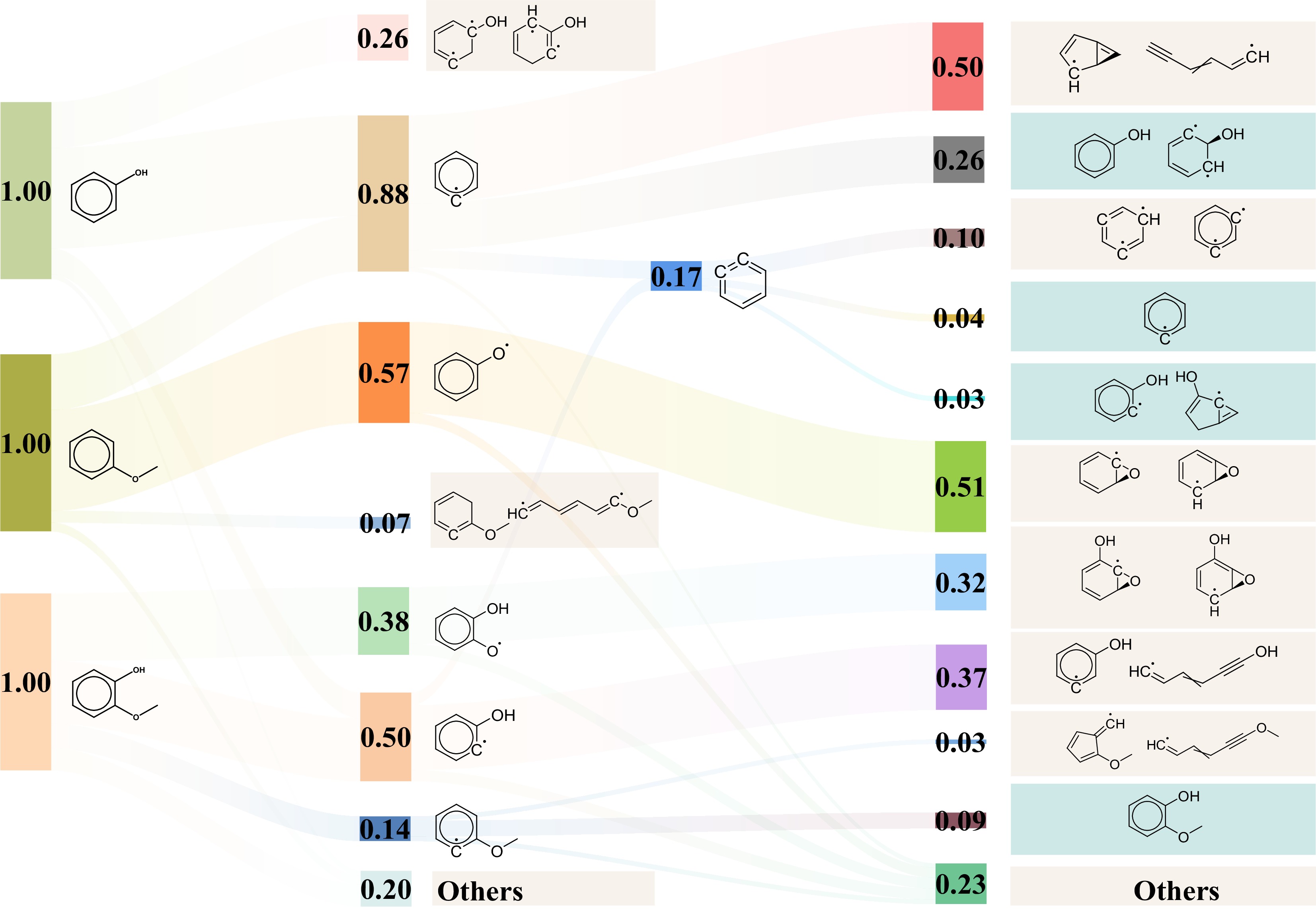
Figure 4.
Sankey diagram of the initial pyrolysis mechanisms of phenol, anisole, and guaiacol at 3,500 K. The isomerization products are highlighted in blue, and the recombination reaction products in cyan, both located in the far right.
-
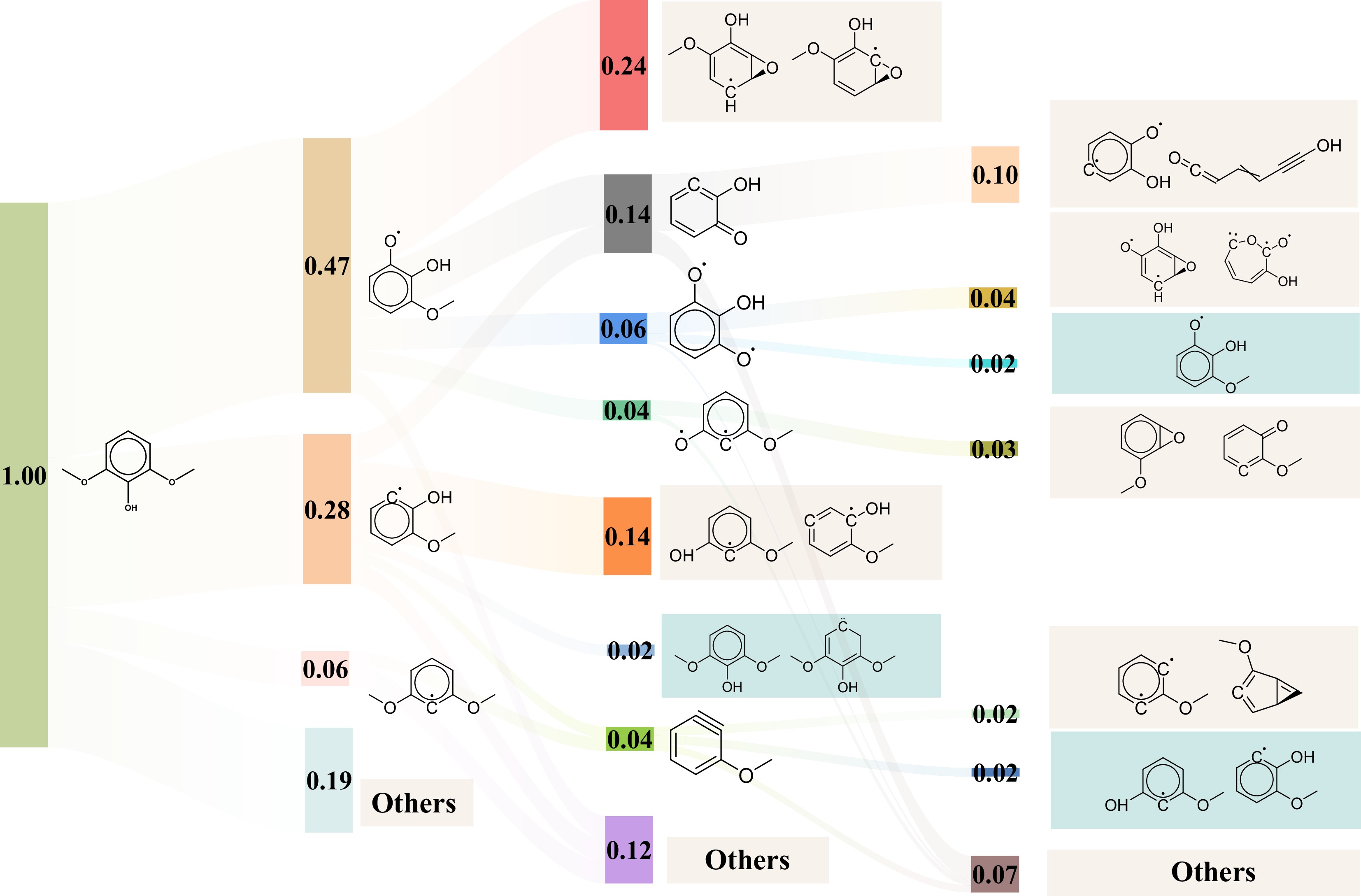
Figure 5.
Sankey diagram of the initial pyrolysis mechanisms of syringol at 3,500 K.
-
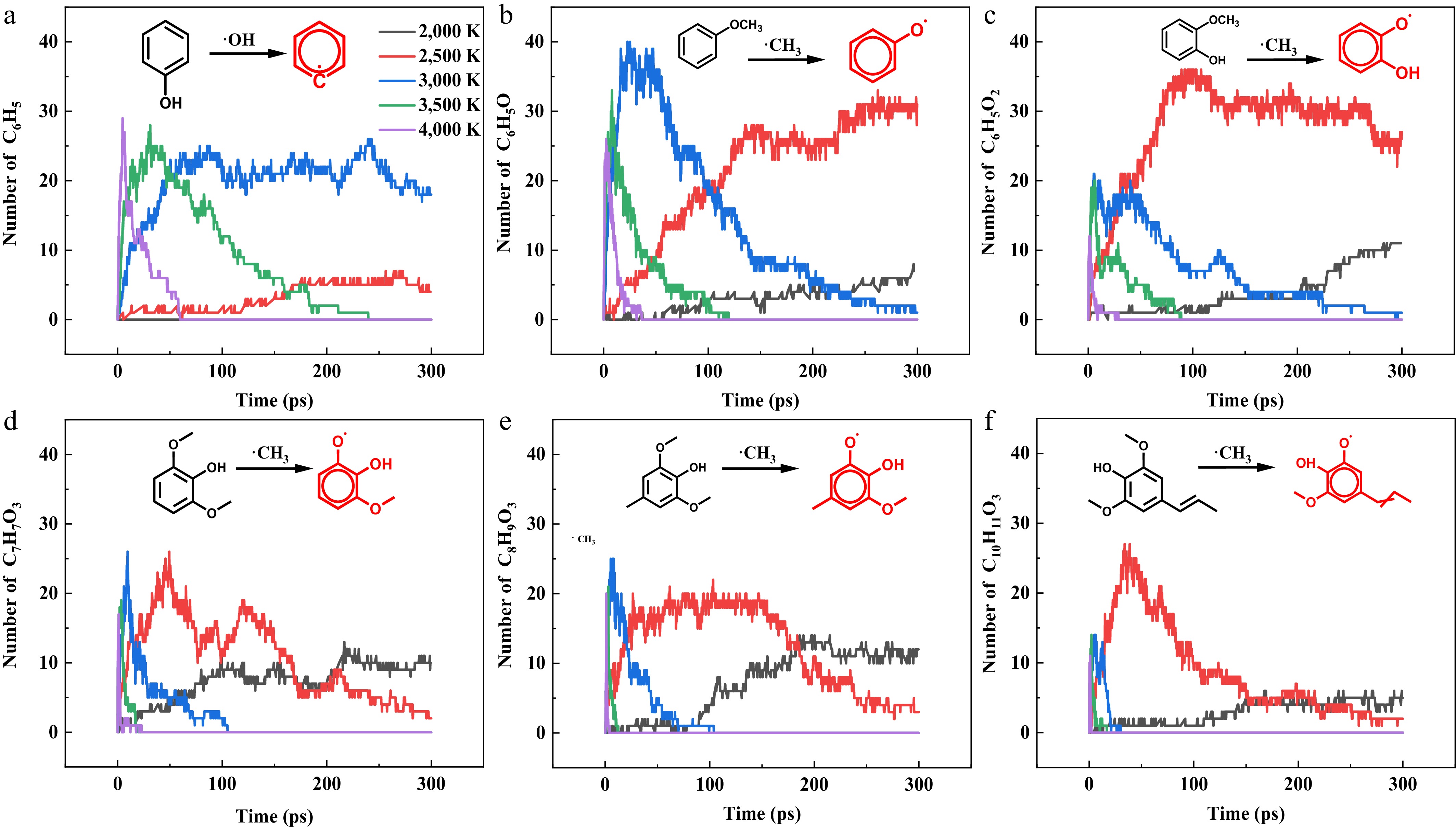
Figure 6.
Temporal evolution of six characteristic initial radicals across five pyrolysis temperatures (2,000, 2,500, 3,000, 3,500, and 4,000 K).
-
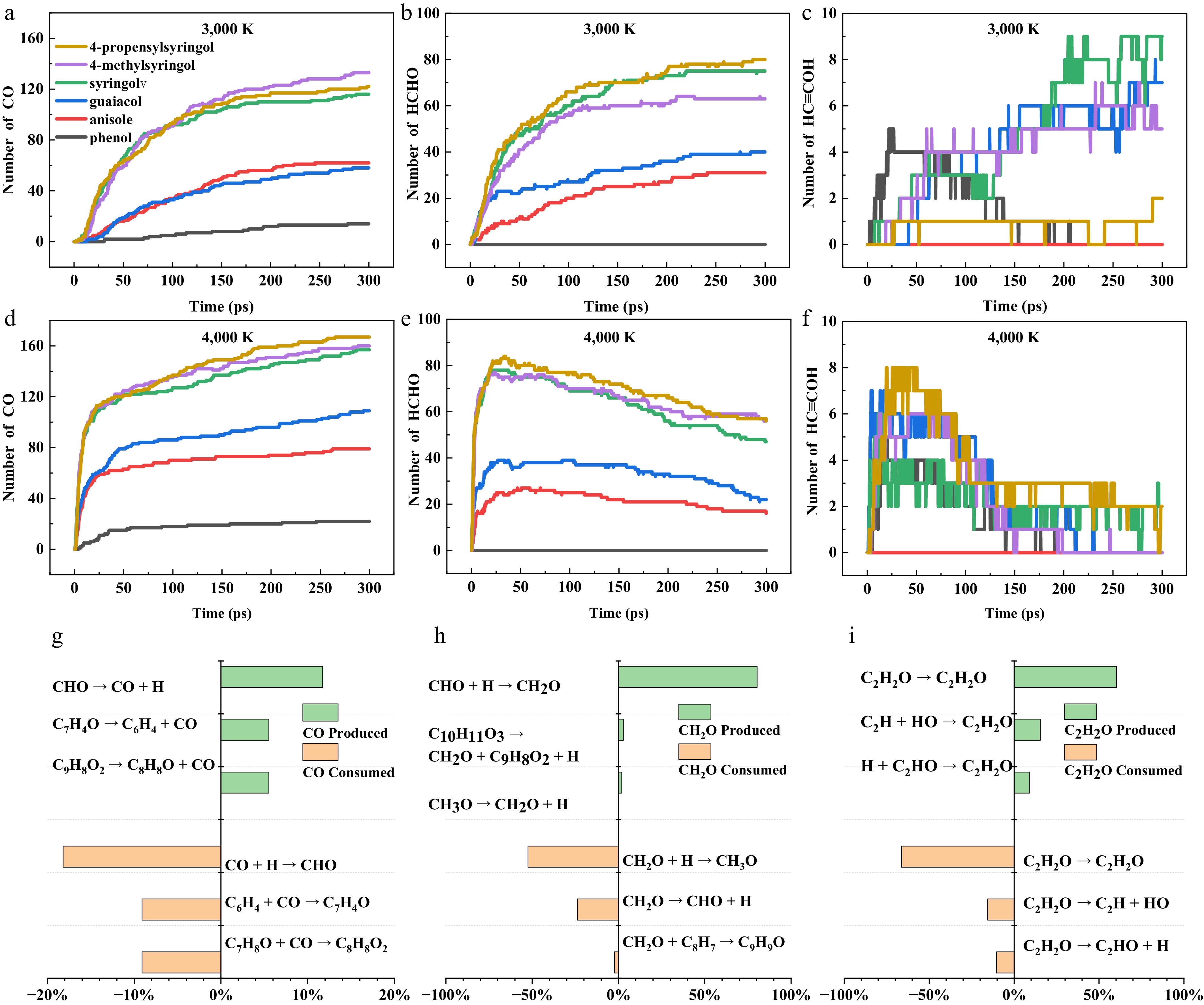
Figure 7.
Quantitative evolution and pathway analysis of key oxygenated small molecules during the pyrolysis of six model compounds. (a)–(c) Quantity-time profiles of CO, HCHO, and HC≡COH generated from the six model compounds at 3,000 K. (d)–(f) Quantity-time profiles of the same three products at 4,000 K. (g)–(i) Generation and consumption pathways of CO, HCHO, and HC≡COH during the pyrolysis of 4-propenylsyringol at 3,500 K.
-
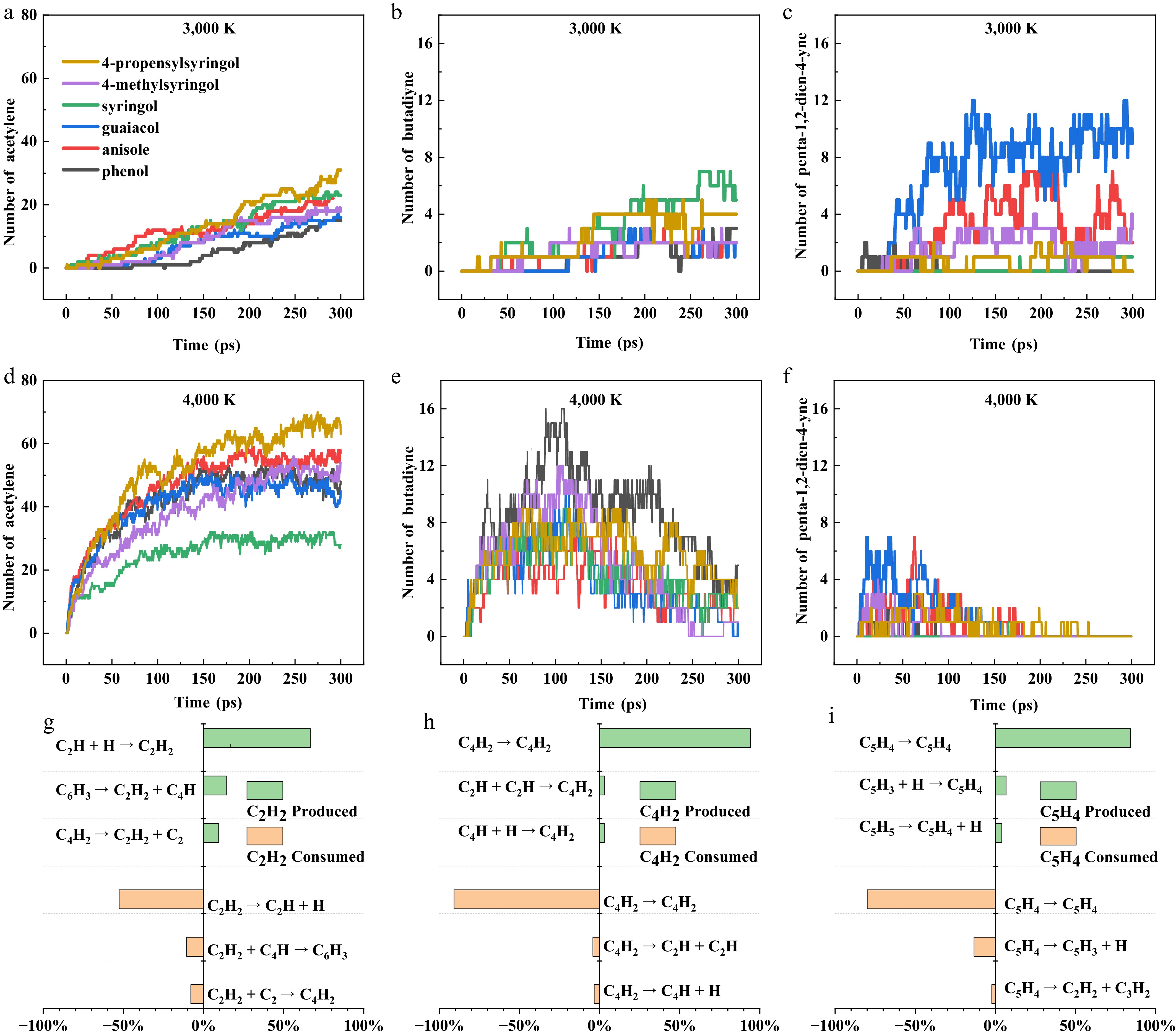
Figure 8.
Quantitative evolution and pathway analysis of key hydrocarbons during the pyrolysis of six model compounds. (a)–(c) Number-time evolution profiles of acetylene (C2H2), butadiyne (C4H2), and penta-1,2-dien-4-yne (C5H4) generated from the six model compounds at 3000 K. (d)–(f) Number-time evolution profiles of the same three products at 4000 K. (g)–(i) Generation and consumption pathways of C2H2, C4H2, and C5H4 during the pyrolysis of 4-propenylsyringol at 3,500 K.
Figures
(8)
Tables
(0)