-
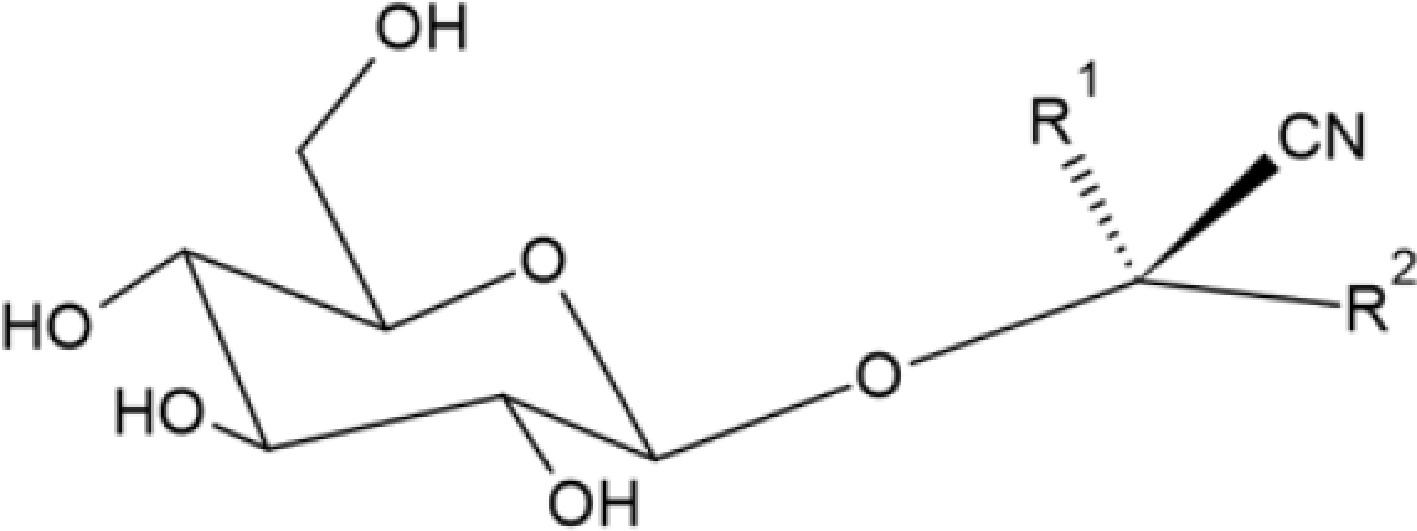
Figure 1.
General structure of cyanogenic glycosides (structure was made with ChemSketch software, version 2024.2.3).
-
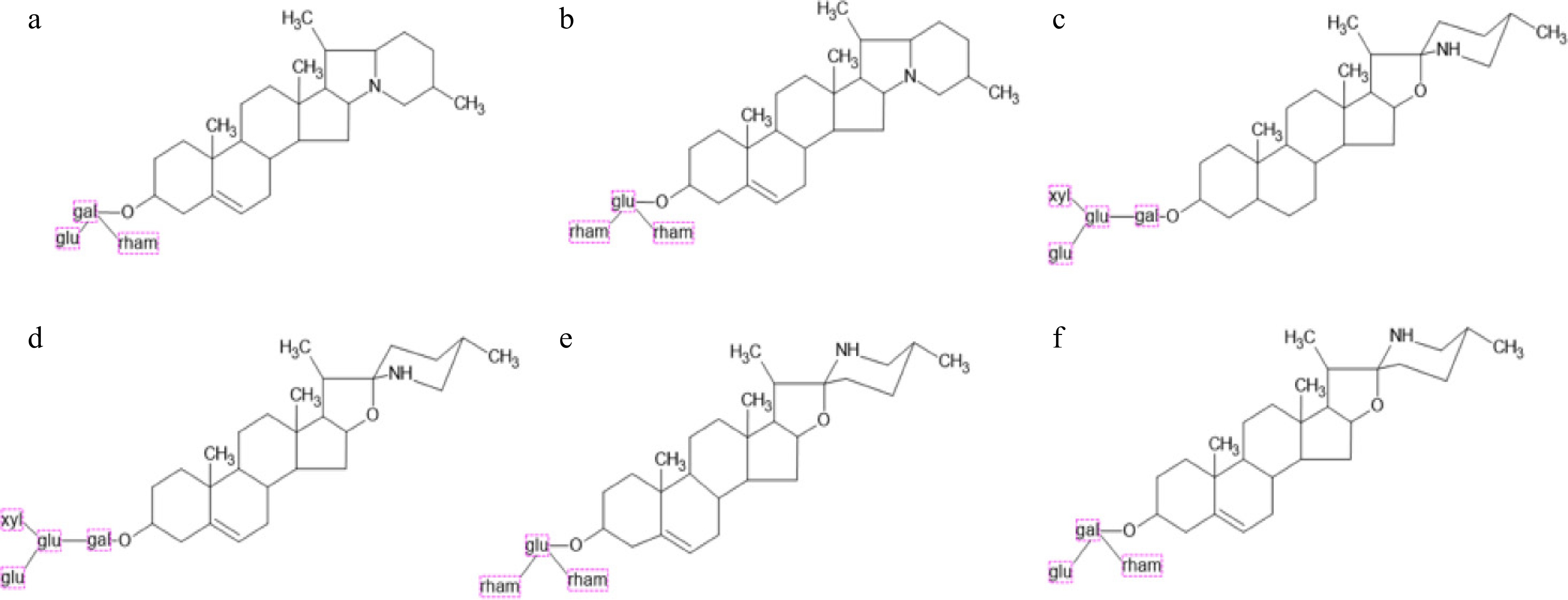
Figure 2.
Structures of the most important glycoalkaloids. (a) α-solanine, (b) α-chaconine, (c) α-tomatine, (d) dehydrotomatine, (e) solamargine, and (f) solasonine (structures were made with ChemSketch software, version 2024.2.3).
-
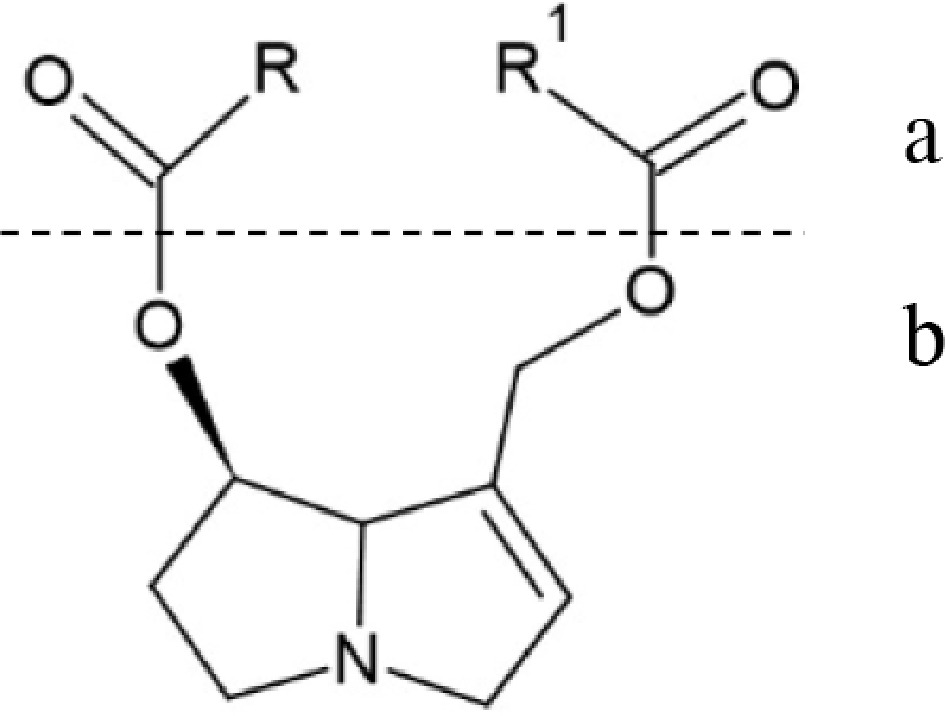
Figure 3.
General structure of Pyrrolizidine alkaloids. (a) Necic acid. (b) Necine base (structure was made with ChemSketch software, version 2024.2.3).
-
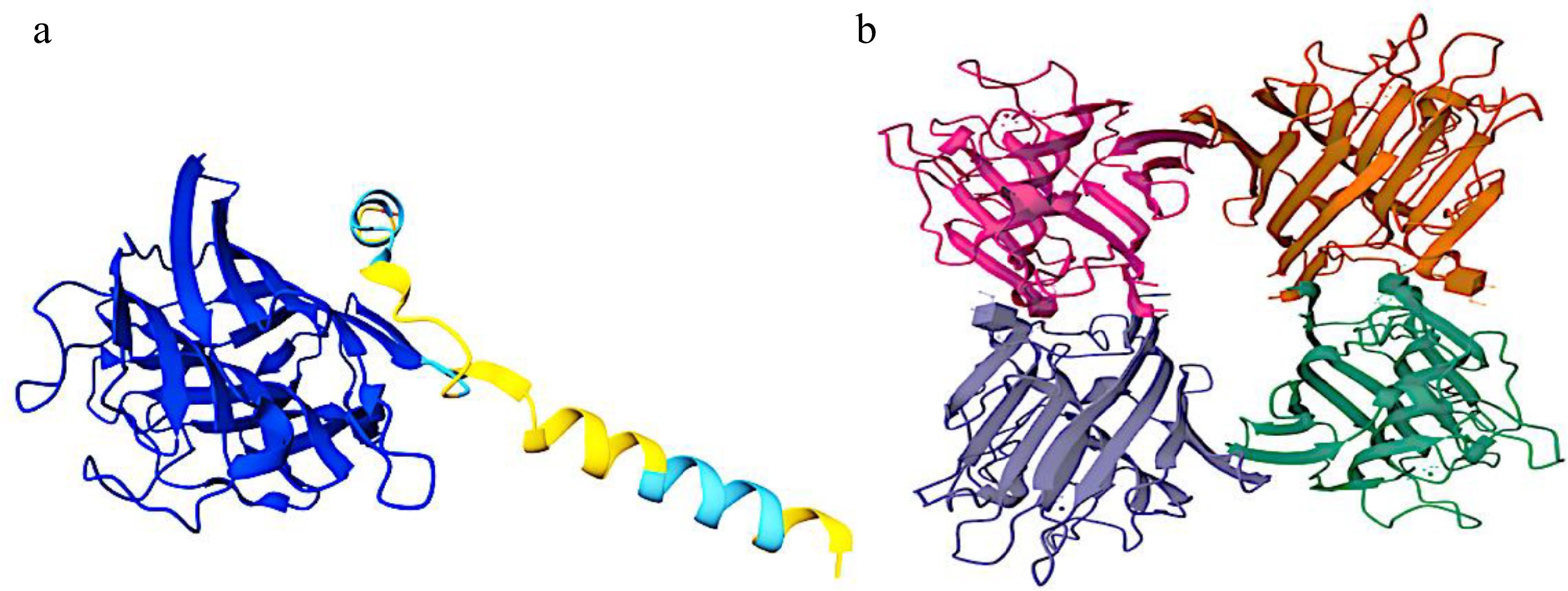
Figure 4.
Three-dimensional structures of the (a) PHA and (b) PHA-L of Phaseolus vulgaris.
-
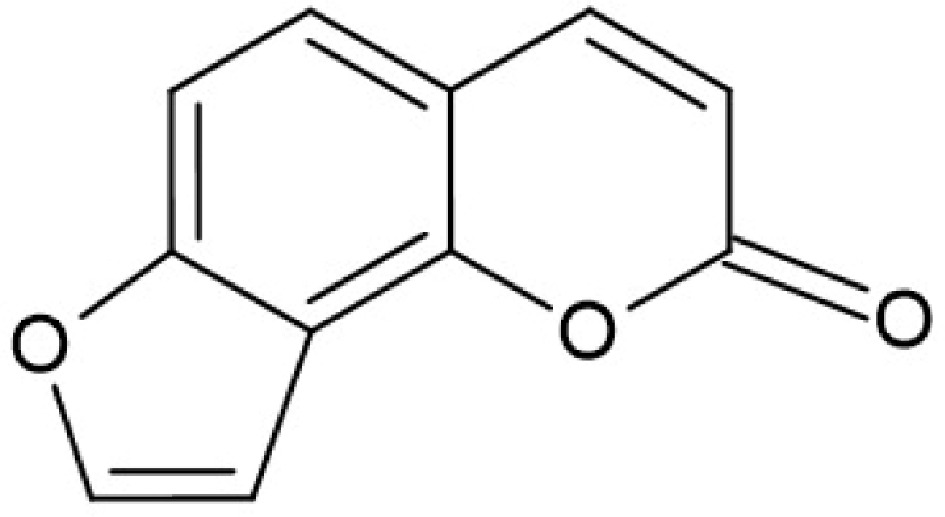
Figure 5.
Chemical structure of furanocoumarin (structure was made with ChemSketch software, version 2024.2.3).
-
Food group Toxic molecule Food Mechanism of toxicity Ref. Fruits Amygdalin Apple seeds, bitter almonds, apricots Release of HCN by enzymatic hydrolysis, inhibiting cytochrome C oxidase and causing cellular hypoxia. [43−45] Furanocoumarins Grapefruit, lime, bergamot UV photoactivation, generating ROS that damage cell membranes and DNA. [46−48] Vegetables Nitrates Spinach, lettuce, beets, celery Conversion to carcinogenic nitrosamines in the gastrointestinal tract. [41,49] α-Solanine Green or sprouted potatoes, eggplants Inhibition of acetylcholinesterase and disruption of cell membranes. [50,51] Cereals Phytic acid Wheat bran, brown rice, legumes Chelation of minerals (Fe, Zn, Ca), reducing their bioavailability. [52,53] Aflatoxin B1 Corn, peanuts, nuts, rice stored in humid conditions Chelation of minerals (Fe, Zn, Ca), reducing their bioavailability. [42,54] Dairy Aflatoxin M1 Milk, cheese, yogurt (from cows that consumed feed contaminated with aflatoxin B1) Hepatotoxic metabolite that forms DNA adducts and causes oxidative stress. [54,55] Egg Avidin Raw egg white Binding to biotin (vitamin B7), preventing its absorption and causing deficiency. [56] Fish and mollusks Tetrodotoxin Globin fish, gecko, some bivalve molluscs Blockage of voltage-gated sodium channels, causing paralysis. [57,58] Mercury (methylmercury) Tuna, swordfish, shark, pike Neurotoxicity by binding to thiol groups in neuronal proteins. Mushrooms α-Amanitin Amanita phalloides, Amanita virosa (poisonous mushrooms) Inhibition of RNA polymerase II, stopping protein synthesis. [59−61] Poultry meat Cytolethal distending toxin (CDT) Chicken contaminated with Campylobacter jejuni Actin depolymerization, causing cell distension and death. [62] Pork and beef Heterocyclic amines Grilled meat, well-cooked fried meats Formation of mutagenic DNA adducts during high-temperature cooking. [25,63,64] Natural infusions Pyrrolizidine alkaloids (PAs) Herbal teas (senecio, borage, comfrey) Metabolization to pyrroles that damage the liver (hepatic veno-occlusion). [65,66] Table 1.
Toxic molecules in some popular foods and their mechanism of toxicity.
-
Subclass Common name Systematic name Chemical structure Aminoimidazoazarenes PhIP 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine 
IQ 2-amino-3-methylimidazo[4,5-f]quinoline 
MeIQ 2-amino-3,4-dimethylimidazo[4,5-f]quinoline 
MeIQx 2-amino-3,8-dimethylmidazo[4,5-f]quinoxaline 
β-carbolines β-carboline-1 carboxylic acid 9H-pyrido[3,4-b]indole-1-carboxylic acid 
Harmane 1-methyl-9H-pyrido[3,4-b]indole 
Norharmane or β-carboline 9H-pyrido[3,4-b]indole 
(Structures were made with ChemSketch software, version 2024.2.3). Table 2.
Structures and systematic names of some heterocyclic aromatic amines.
Figures
(5)
Tables
(2)