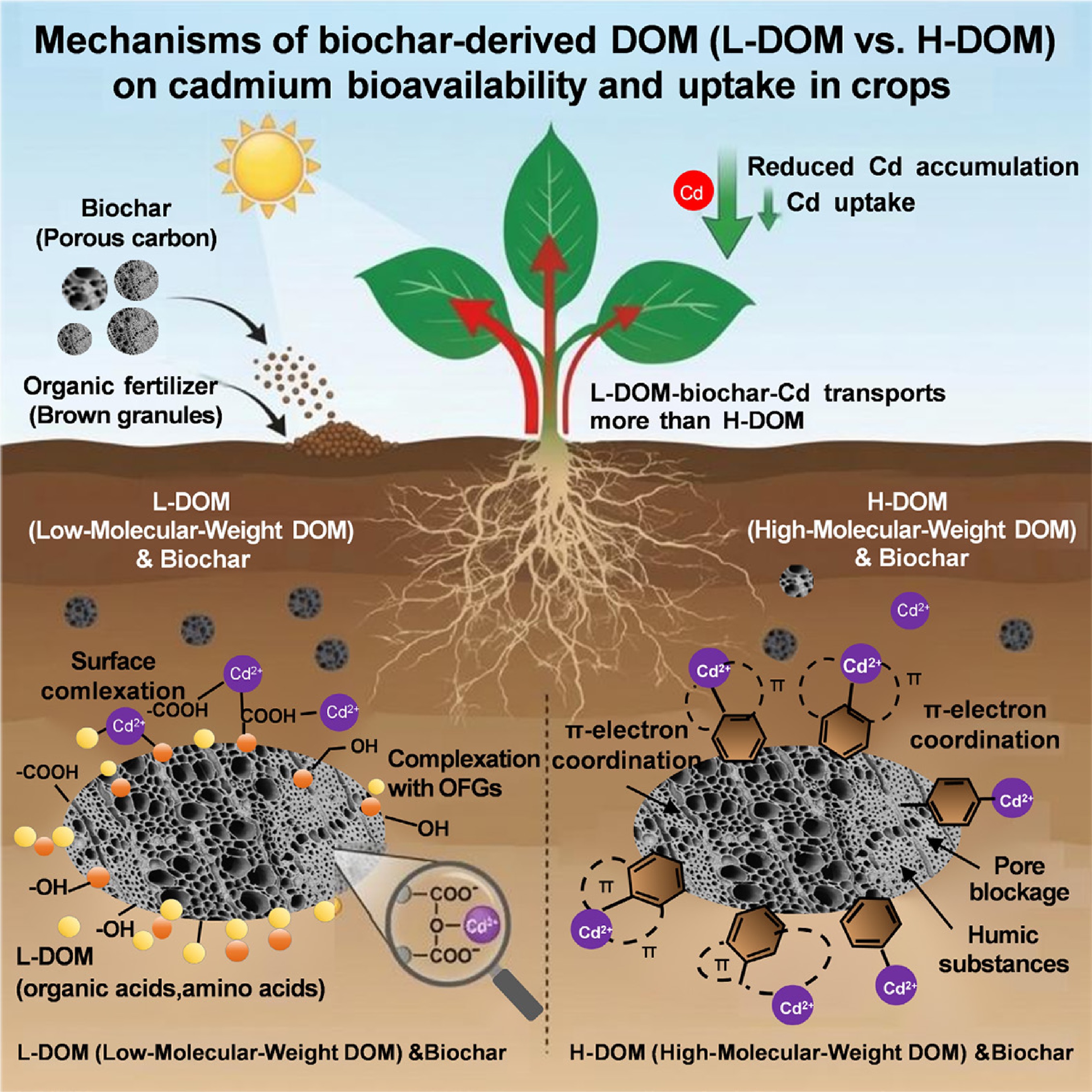
-
Cadmium (Cd) is a common non-essential element with high toxicity in contaminated soils. Cd concentrations can negatively affect the metabolism of microorganisms and growth of plants[1]. How to reduce the bioavailability of Cd in soil and prevent Cd from entering the food chain has been a problem of widespread concern. Moreover, the availability and migration of heavy metals (HMs) in soil are dependent on their chemical speciation and influenced by various environmental conditions such as soil agrotypes, soil pH, and dissolved organic matter (DOM)[2]. Organic fertilizer is widely used to improve soil quality and crop yield, and is also considered a cost-effective amendment for mitigating heavy metal pollution[3]. However, its role in remediating Cd-contaminated soils remains uncertain. Some research results showed that the content of Cd in soil is positively correlated with the content of organic matter. The higher the content of organic matter, the higher the risk of Cd pollution[4]. However, some other results indicate that organic fertilizer primarily enhances heavy metal mobility by increasing DOM content, rather than by altering soil adsorption behavior[5,6]. This implies that DOM components in organic fertilizer may play a major role.
As is well recognized, the composition and characteristics of DOM derived from both organic fertilizers and soil are highly complex and variable[7]. While current research has largely focused on the effects of DOM derived from soil or sediments on the adsorption of HMs[5−8], the binding efficiency between different DOM components and HMs remains debated[9]. Li et al.[10] demonstrated that soil DOM can form soluble complexes with Cd2+ and Pb2+, influencing their bioavailability, though its maximum sorption capacity for Cd varies considerably. Studies on molecular weight fractions have yielded conflicting results: some report that the < 0.5 kilodalton (kDa) fraction exhibits the highest metal-binding potential[11], while others suggest that the > 1 kDa fraction binds more strongly with metals such as Cu2+ and Hg2+ [12].
Biochar, as a usual soil amendment material, is characterized by its porous structure, high aromatization, and abundant surface functional groups[13]. These characteristics enable Cd2+ immobilization through multiple pathways[14]. Its hydrophilic and hydrophobic surface properties allow the adsorption of both polar and non-polar organic substances, and modifications (e.g., with chitosan) can further enhance performance by increasing cation exchange capacity, even if surface area is reduced[15−17]. Furthermore, the binding of HMs is strongly governed by the chemical composition, structure, and molecular weight of DOM[18]. DOM comprises a complex mixture of both polar low-molecular-weight compounds and nonpolar high-molecular-weight compounds.
The combined application of organic fertilizers and biochar as soil amendments for remediating heavy metal-contaminated soils has been extensively documented[19,20]. Field studies have demonstrated that this combination significantly reduces Cd bioavailability[21], and in seasonally frozen lands, enhances long-term immobilization by inhibiting soil aggregate cracking[22]. Although strong interactions are expected between biochar and organic fertilizer-derived DOM, the role of specific DOM fractions in Cd immobilization after binding to biochar remains poorly understood.
This study aims to elucidate the mechanisms underlying the combined effectiveness of organic fertilizer-derived DOM and biochar in Cd immobilization, and to provide conceptual guidance for developing next-generation composite amendments, such as incorporating DOM extracted from organic fertilizers onto biochar within existing production lines. To achieve this, DOM was extracted from organic fertilizer by ultrafiltration centrifugation and fractionated into three molecular weight components, which were then combined with different modified biochars to prepare composites. The resulting materials were evaluated for Cd2+ adsorption isotherms, their effects on key soil properties, and their impact on crop Cd uptake, thereby offering theoretical support for the development of biochar-based materials for Cd-contaminated soil remediation.
-
Biochar was produced from pomelo (Citrus maxima [Burm]. Merr.) branches. The raw materials were first washed with tap water to remove adhering dust, then rinsed with deionized water three times. After drying in an oven at 60 °C, the materials were ground and passed through a 2 mm sieve. A 50.00 g aliquot of each prepared biomass was weighed and then placed in a polytetrafluoroethylene (PTFE) crucible. The crucible was covered, sealed with tin foil, and transferred to a muffle furnace. The temperature was increased at a rate of 15 °C·min–1 to 300 °C and held for 4 h. After cooling to room temperature, the residue in the crucible was collected as the original biochar. A portion of this original biochar (designated Bc) was then washed three to five times separately with deionized water or a 0.1 mol·L–1 HCl solution. The washed samples were dried, yielding the final materials designated Bw (water-washed biochar) and Bh (acid-washed biochar), respectively.
The organic fertilizer used in this study was the commercially available 'Jiashifu' brand bio-organic fertilizer, a product developed from the technological achievements of the Institute of Agricultural Resources and Environment, Guangdong Academy of Agricultural Sciences. It was produced through thermophilic aerobic composting of livestock manure, mushroom residue, peanut bran, and rice bran. The fertilizer had a pH of 8.97, an organic matter content of 40.13 g·kg–1, and a dissolved organic carbon (DOC) content of 3.26 g·kg–1. To extract the DOM, 300 g of the organic fertilizer was weighed into a beaker and mixed with 900 mL of deionized water at a mass ratio of 1:3. The mixture was then treated with ultrasonication (40 kHz) at 50 °C for 1 h, followed by centrifugation at 4,000 rpm for 20 min. The supernatant was finally filtered through a 0.45 μm membrane to obtain the DOM solution.
By the ultrafiltration method, DOM was divided into three components based on molecular weight: < 3 kDa (Component 1, DOM<3k), 3k–10 kDa (Component 2, DOM3k–10k) and > 10 kDa (Component 3, DOM>10k). The procedure was as follows: A 3 kDa ultrafiltration centrifugal tube was first pre-rinsed with 15 mL of 0.1 mol·L–1 NaOH solution, followed by a rinse with 15 mL of deionized water and centrifugation at 4,000 rpm for 2 min. Subsequently, 12 mL of the pre-filtered DOM solution was added to the tube and centrifuged at 5,000 rpm for 30 min. The filtrate obtained was collected as the DOM<3k component. The remaining solution was then transferred to a 10 kDa ultrafiltration centrifugal tube. Following the same centrifugation protocol, the filtrate was collected as the DOM3k–10k component, and the residual solution was designated as the DOM>10k component.
After adjusting the concentration of the three DOM components to the same C content of 50 mg C·L–1, the three DOM component solutions obtained were fully mixed with Bw and Bh, respectively (at a mass ratio equivalent to 0.25% of the carbon content of biochar), and then freeze-dried at room temperature for 24 h. They were regarded as biochar DOM complexes, which were recorded as: water-washed biochar-DOM<3k complex (Bw1), water-washed biochar-DOM3k–10k complex (Bw2), water-washed biochar-DOM>10k complex (Bw3); acid-washed biochar-DOM<3k complex (Bh1), acid-washed biochar-DOM3k–10k complex (Bh2), and acid-washed biochar-DOM>10k complex (Bh3). The adsorption rate of DOM was 47.90%–68.27%, and the desorption rate was 4.10%–5.10%, indicating that biochar and DOM formed a relatively stable complex.
Isothermal adsorption experiment
-
Nine types of materials were weighed for the adsorption experiments: the three prepared original/washed biochars (Bc, Bw, Bh) and the six biochar-DOM composites (Bw1, Bw2, Bw3, Bh1, Bh2, and Bh3). For each sample, 0.1000 g was placed into a 50 mL centrifuge tube. Then, 30 mL of a series of Cd2+ solutions (with concentrations of 0, 10, 20, 40, 80, 120, and 160 mg·L–1, using 0.01 mol·L–1 CaCl2 as the electrolyte solution and adjusted to pH 5.5), was added to each tube. The suspensions were shaken thoroughly at 25 °C for 24 h. After shaking, they were allowed to settle for 20 min and then filtered through a 0.45 μm membrane. The Cd2+ concentration in the filtrate was determined. Each treatment was performed in triplicate. The adsorption data were fitted using the Langmuir, Freundlich, and Temkin isotherm models. At the same time, samples of the biochars and biochar-DOM complexes were collected, and the changes in surface functional groups before and after the adsorption experiments were determined by Fourier-transform infrared (FTIR) spectroscopy.
Soil incubation experiment
-
The soil used for the incubation and culture experiment was collected from farmland around a mining area in Yangshuo, Guangxi. Surface soil samples (0–20 cm) were gathered, and then plant residues and stones were manually removed. The soil was then spread evenly on plastic film, air-dried, thoroughly mixed, ground, and passed through a 2 mm sieve for standby. The main physical and chemical properties of the soil were as follows: pH 5.53, organic matter content 19.80 g·kg–1, total Cd content 5.74 mg·kg–1, and available Cd content 3.99 mg·kg–1.
Seven treatments were set up in the soil incubation experiment, which were: (1) control (CK), (2) Bw1, (3) Bw2, (4) Bw3, (5) Bh1, (6) Bh2, and (7) Bh3, with three replicates for each treatment. For each replicate, 150.00 g of soil was weighed into a plastic container. According to the experimental design, biochar-DOM complexes were added at a rate of 1% (w/w, relative to soil mass). The soil and amendments were mixed thoroughly. The containers were then covered with plastic wrap to minimize moisture loss and incubated at a constant temperature of 25 °C for 90 d. Throughout the incubation period, deionized water was added regularly to maintain the soil moisture at 70% of the soil water-holding capacity. Soil samples were collected every 10 d to determine the available Cd content. The fractions of Cd in the soil were tested after the incubation experiment.
Pot experiment
-
The soil used for the pot experiment had the same physicochemical properties as the soil in the incubation experiment. Seven treatments were set up, including a control (CK) with no amendment, and six treatments amended with 1% (mass ratio, w/w) of the respective biochar-DOM complex, each with three replicates. For each pot, 2 kg of soil was mixed with the designated biochar-DOM complex and 3 g of compound fertilizer. The mixture was placed into a polyethylene pot (20 cm in height, 15 cm in diameter). The soil moisture was adjusted to approximately 80% of the soil water-holding capacity. The pots were then kept at room temperature for 15 d and covered with plastic film to prevent excessive water loss. Chinese cabbage (Brassica rapa var. glabra Regel) seeds were purchased from the Wushan seed market in Tianhe District, Guangdong Province, in October 2024. Subsequently, three Chinese cabbage seeds were sown in each pot. After about 15 d, seedlings were thinned to retain one uniform plant per pot. Throughout the whole growth period, distilled water was applied to maintain soil moisture between 40% and 80% of the soil water-holding capacity.
Plants were harvested 40 d after sowing. The aboveground and underground parts were collected separately. They were first rinsed with tap water for 3–5 min, followed by three washes with approximately 100 mL of deionized water each. After air-drying at room temperature for 1 h, the samples were weighed to record the fresh weight, which was used for yield calculation. All samples were sterilized in an oven at 105 °C for 10 min, and dried at 80 °C to constant weight. The dried samples were ground for Cd content analysis. Soil samples were collected from each pot, air-dried, sieved according to the requirements of subsequent analyses, and used for the determination of soil available Cd and Cd fractions.
The enrichment factor (EF) is used to measure the concentration of Cd in plant tissues relative to that in soil, and the calculation formula is as follows[23]:
$ {EF=}\dfrac{{{C}}_{{Plant}}}{{{C}}_{{soil}}} $ (1) In Eq. (1), Cplant represents the concentration of Cd in plant tissues (mg·kg–1), and Csoil represents the concentration of Cd in soil (mg·kg–1).
The transfer coefficient (TF) is used to measure the ability of plants to transfer Cd from underground to aboveground parts. The calculation formula is as follows:
$ {TF=}\frac{{{C}}_{{shoot}}}{{{C}}_{{root}}} $ (2) In Eq. (2), Cshoot represents the concentration of Cd in the plant shoot (mg·kg–1), and Croot represents the concentration of Cd in the plant root (mg·kg–1).
Analytical methods
Soil analysis
-
Soil pH was measured by the glass electrode method with a water-to-soil ratio of 2.5:1. Soil available Cd content was determined following the GB/T 23739-2009 method. 5.00 g of air-dried soil (sieved to < 2 mm) was placed in a 100 mL conical flask with a stopper, and 25 mL of DTPA extraction solution (0.005 mol·L–1 DTPA, 0.1 mol·L–1 TEA, 0.01 mol·L–1 CaCl2) was added. The mixture was shaken at 25 °C for 2 h (180 times·min–1). After filtration, the Cd concentration in the filtrate was measured using an atomic absorption spectrophotometer (ICE 3500, Thermo Fisher Scientific Co., USA).
Cd fractions in the soil were analyzed using a modified Tessier sequential extraction procedure[24,25]. The fractions analyzed included water-soluble (WS), specifically adsorbed (SP), amorphous iron-manganese oxides (AIO), crystalline iron-manganese oxides (CIO), organic-bound (OB), and residual (RES) Cd[26]. The specific method was as follows: air-dried soil (sieved to < 0.25 mm) was dried in an oven at 60 °C for 2 h to achieve a constant weight. A 1.0000 g soil sample was weighed into a 50 mL brown light-proof centrifuge tube. Sequential extractions were then performed according to the steps in the article. Following each extraction step, 20 mL of deionized water was added to rinse the soil sample.
The Cd2+ concentration in all extracted solutions was determined using a graphite furnace atomic absorption spectrophotometer (Model AA600, PerkinElmer, USA). Each sample was measured in triplicate, and the results were reported as the mean value. To ensure the accuracy and reliability of the data, blank samples, certified reference materials, and spiked recovery samples were analyzed concurrently. The relative standard deviation (RSD) for the measurements was maintained below 10%. The method detection limit (MDL) for Cd was 0.05 μg·L–1.
Dissolved organic carbon (DOC) concentration was determined using a Shimadzu TOC-5050A total organic carbon analyzer.
Plant analysis
-
The content of Cd in the aboveground and underground parts of Chinese cabbage plants was digested with HNO3–HClO4 (volume ratio v : v = 5:1), and the content of Cd in the digestion solution was determined by inductively coupled plasma mass spectrometry (ICP-MS, 7800, Agilent Technology, USA). For quality assurance and control, reagent blanks and triplicate samples were set up during the digestion process of each batch of samples. Furthermore, the quality control of the whole digestion, analysis, and determination process was carried out by using the national reference material GBW10048 (GSB-26) (produced by the Institute of Geophysical and Geochemical Exploration, IGGE).
Other analysis
-
Surface functional groups were identified by Fourier transform infrared spectroscopy (FTIR, Model PerkinElmer FTIR-2000, USA).
Data processing and statistical analysis
-
All data were the mean values of three replicates. Statistical analysis was performed using IBM SPSS Statistics 26 (IBM Corp., USA). Significant differences among the various treatments were evaluated by one-way analysis of variance (ANOVA) at a significance level of p < 0.05, followed by Duncan's multiple range test for comparisons. All graphs were generated using OriginPro 2024b (OriginLab Corp., USA).
-
The Cd2+ adsorption isotherms for both the three types of biochar and the six biochar-DOM composites were well-fitted by the Langmuir, Freundlich, and Temkin models, with the Langmuir model providing a slightly better fit (Fig. 1). All corresponding R2 values reached a very significant level (p < 0.01) (Table 1) . The Langmuir constant (KL) ranged from 0.05 to 0.11. The KL values for the three original/washed biochars were the lowest, while those for Bw1, Bw2, and Bw3 were comparatively higher; 25.00%–120.00% higher than the others, indicating a weaker affinity for Cd2+ adsorption by the original biochars themselves. Washing with water or a dilute acid solution had no significant effect. The Cd2+ adsorption capacity of water-washed biochar with DOM complexes was significantly improved, but there was no significant difference among DOM complexes with different molecular weights. The KL values for acid-washed biochar (Bh) composites with high-molecular-weight DOM (> 3 kDa) increased significantly, confirming their enhanced Cd2+ adsorption affinity. The separation factor (RL) value ranged from 0.18 to 0.35, which is contrary to KL. It further supported the relatively stronger adsorption performance of Bw1, Bw2, and Bw3 for Cd2+. The calculated maximum adsorption capacity (qmax) varied between 54.53 and 84.25 mg·g–1. The qmax values for water-washed and acid-washed biochar increased by 19.00% and 22.00%, respectively, compared to the original biochar, indicating that the adsorption capacity of Cd2+ was improved. For the biochar-DOM complexes, the qmax values were enhanced by 2.00%–55.00%, with the extent of increase positively correlated with DOM molecular weight. This demonstrates that the adsorption capacity of Cd2+ in the complex was enhanced compared with biochar alone, with the enhancement being more pronounced for composites incorporating higher molecular weight DOM.
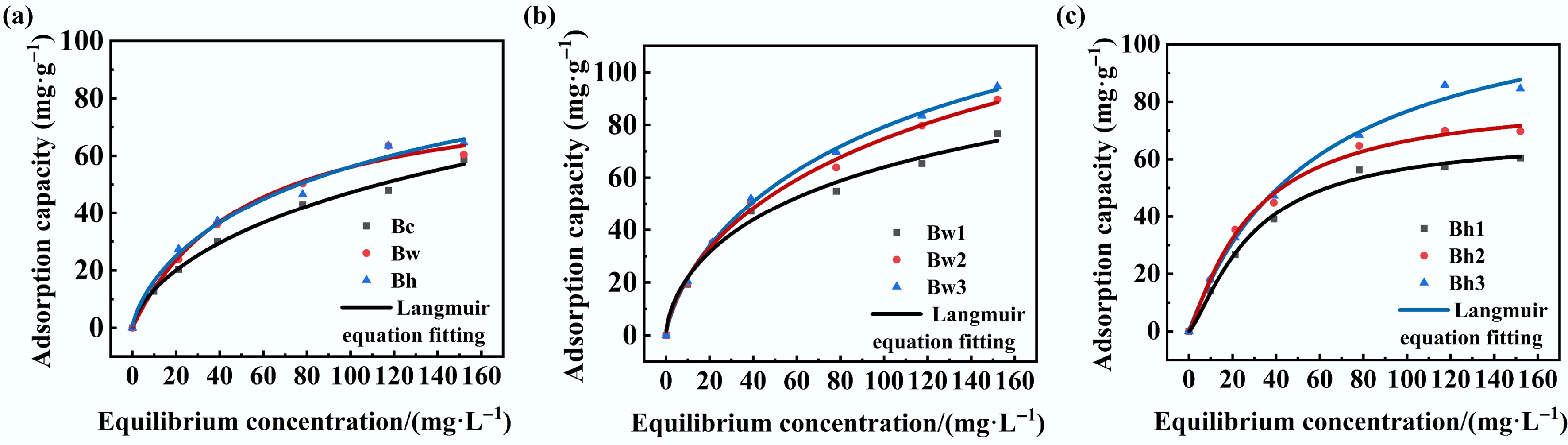
Figure 1.
Isothermal adsorption equations of Cd2+ by different modified biochar-DOM composites; (a) represents the Freundlich equation fitting of Cd adsorption by biochar alone; (b) represents the Freundlich equation fitting of Cd adsorption by water-washed biochar combined with DOM; and (c) represents the Freundlich equation fitting of Cd adsorption by acid washed-biochar combined with DOM.
Table 1. Correlation parameters for isothermal adsorption equations of Cd2+ by different biochar and modified biochar-DOM composites
Model Parameter Bc Bw Bh Bw1 Bw2 Bw3 Bh1 Bh2 Bh3 Langmuir qmax (mg·g–1) 54.53 64.94 66.71 67.98 79.30 81.04 71.84 76.80 84.25 KL (L·mg–1) 0.05 0.05 0.06 0.11 0.10 0.11 0.05 0.08 0.07 RL 0.33 0.33 0.31 0.18 0.20 0.19 0.35 0.24 0.26 R2 0.97 0.98 0.99 0.97 0.97 0.96 1.00 0.99 0.98 Freundlich 1/n 0.47 0.47 0.44 0.35 0.98 2.50 0.44 0.38 0.45 kf 5.78 6.99 8.19 13.88 1.60 0.00 8.07 1.00 10.82 R2 0.99 0.97 0.96 0.98 0.98 0.99 0.93 0.92 0.98 Temkin BT (J·mol–1) 14.07 15.88 15.36 14.61 18.65 19.85 14.92 15.39 20.55 KT (L·mg–1) 0.35 0.40 0.48 1.03 0.75 0.74 0.52 0.91 0.54 R2 0.96 0.97 0.96 0.96 0.97 0.97 0.98 0.97 0.97 The combination of Bw and DOM<3k is denoted as Bw1; the combination of Bw and DOM3k–10k is denoted as Bw2; the combination of Bw and DOM>10k is denoted as Bw3; the combination of Bh and DOM<3k is denoted as Bh1; the combination of Bh and DOM3k–10k is denoted as Bh2; and the combination of Bh and DOM>10k is denoted as Bh3. The same below. The results in Table 2 showed that the specific surface area of Bh increased by 64.85% compared with Bc, indicating that acid washing effectively removed mineral impurities and exposed more pore structures. For both water-washed and acid-washed biochars, loading with DOM further increased the specific surface area, and this increase was positively correlated with the molecular weight of DOM. Among all samples, Bh3 exhibited the highest specific surface area (4.65 m2·g–1). In addition, both acid washing and biochar-DOM composites effectively enhanced the total pore volume. The Bh3 composite showed the largest total pore volume, which was 87.15% higher than that of Bc. Bh significantly reduced the average pore size, from 12.62 nm in Bc to 10.96 nm in Bh, suggesting that acid washing may also etch the pore walls, leading to a decrease in pore diameter. Furthermore, DOM loading further reduced the pore size, with the composites incorporating high-molecular-weight DOM exhibiting the smallest average pore size.
Table 2. BET surface area, pore volume, and average pore size by different biochar and modified biochar-DOM composites
Treatment SABET
(m2·g–1)Pore volume
(cm3·g–1)Average pore
size (nm)Bc 2.69 0.013 12.62 Bw 3.89 0.013 11.12 Bh 4.44 0.018 10.96 Bw1 2.77 0.011 11.35 Bw2 3.26 0.015 11.46 Bw3 3.74 0.016 10.96 Bh1 3.77 0.018 11.91 Bh2 4.14 0.020 10.05 Bh3 4.65 0.025 9.92 FTIR
-
Following Cd2+ adsorption, the FTIR spectra showed significant differences for biochar composites bound with low-molecular-weight DOM (Bh1 and Bw1) compared to those recorded before the reaction (Fig. 2). A notable increase in peak intensity was observed in the 3,200–3,400 cm–1 region, corresponding to –OH/–NH stretching vibration peaks. Peak intensities decreased in the range of 1,200–1,280 cm–1 (C–C/C–O stretching vibrations) and below 800 cm–1 (representing the –CH/–OH bending vibrations). When the molecular weight of DOM increased, the group peaks of the composites became progressively more similar, with those containing the highest molecular weight DOM being nearly identical to Bh/Bw (Fig. 2a).
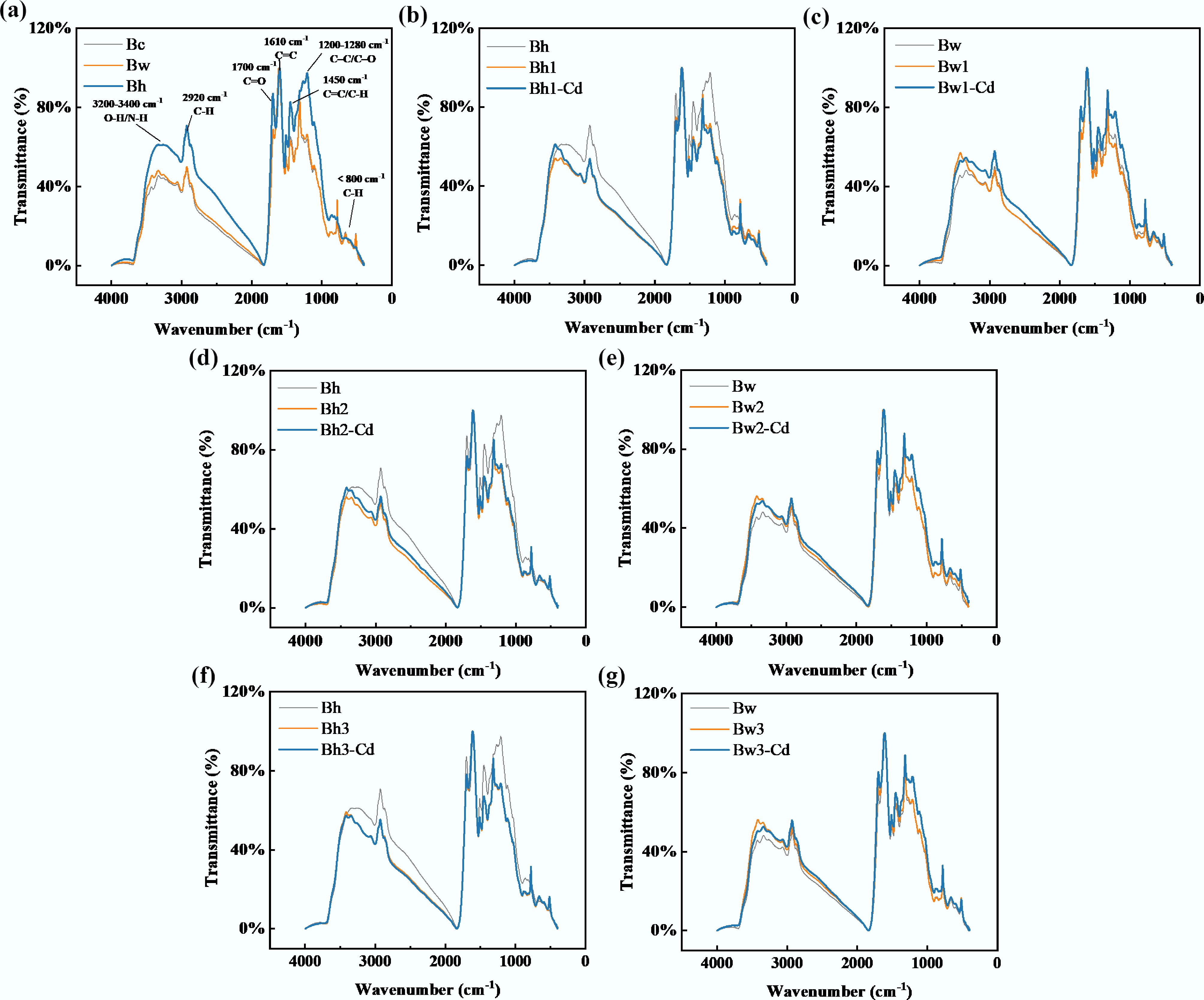
Figure 2.
FTIR spectra of the different modified biochar-DOM composites before and after Cd2+ adsorption; (a) represents the FTIR spectra of original biochar (Bc), acid-washed biochar (Bw), and water-washed biochar (Bh); (b) represents the spectra of Bh, Bh1, and Bh1-Cd; (c) represents the spectra of Bw, Bw1, and Bw1-Cd; (d) represents the spectra of Bh, Bh2, and Bh2-Cd; (e) represents the spectra of Bw, Bw2, and Bw2-Cd; (f) represents the spectra of Bh, Bh3, and Bh3-Cd; (g) represents the spectra of Bw, Bw3 and Bw3-Cd). The combination of Bw and DOM<3k is denoted as Bw1; Bw1 combines with Cd to form Bw1-Cd; the combination of Bw and DOM3k–10k is denoted as Bw2; Bw2 combines with Cd to form Bw2-Cd; the combination of Bw and DOM>10k is denoted as Bw3; Bw3 binds to Cd to form Bw3-Cd; the combination of Bh and DOM<3k is denoted as Bh1; Bh1 binds to Cd to form Bh1-Cd; the combination of Bh and DOM3k–10k is denoted as Bh2; Bh2 combines with Cd to form Bh2-Cd; the combination of Bh and DOM>10k is denoted as Bh3; Bh3 combines with Cd to form Bh3-Cd.
The spectral consistency between Bw1–3 (Bw-DOM composites) and Bw was higher than that observed for Bh1–3 (Bh-DOM composites) and Bh. Aside from a significant intensity increase in the 3,200–3,400 cm–1 region (representing the –OH/–NH stretching vibration peak), the peak patterns and intensities in the Bw1–3 composites were largely consistent with those of Bw. The group strength increased after Bh1–3 reacted with Cd2+, but only in the region of 3,200–3,400 cm–1 (representing the stretching vibration peak of –OH/–NH). The changes in group peak intensities before and after the reaction of Bw-DOM complexes with Cd2+ showed almost no dependence on the molecular weight of DOM.
Soil incubation experiment
-
The results of the soil incubation experiment demonstrated that the addition of biochar-DOM composites significantly increased soil pH (Fig. 3), which was significantly increased by 0.43–0.84 units compared with the control CK. The greatest increase was observed in the Bw3 treatment, which raised the soil pH by 0.84 units relative to the control. No significant differences in pH were found among the treatments with acid-washed biochar composites (Bh1, Bh2, Bh3). In contrast, the higher the molecular weight of washed biochar-DOM complexes, the greater the increase in soil pH value. These results indicate that applying composite materials synthesized from biochar and high-molecular-weight DOM is effective in enhancing soil pH.
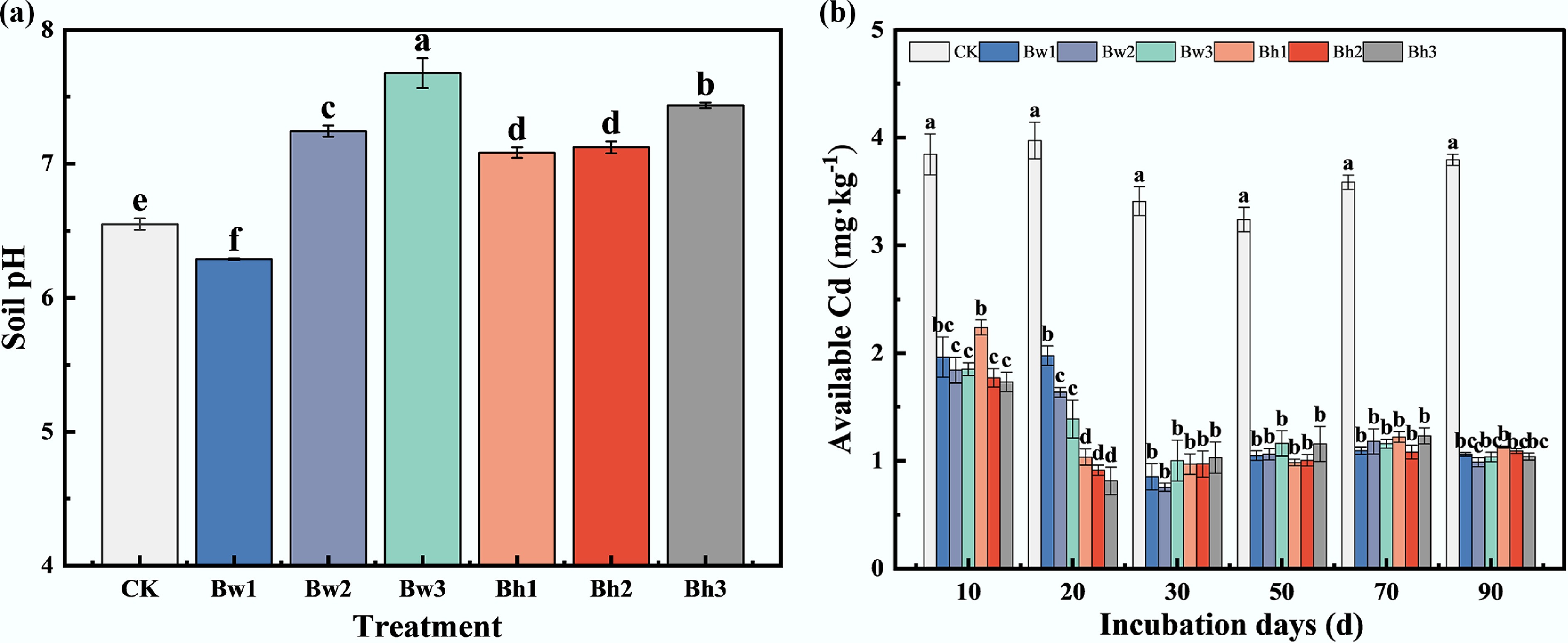
Figure 3.
Effects of different biochar-DOM complexes on (a) soil pH, and (b) available Cd content. CK: control treatment; Bc: biochar; Bw: biochar washed with deionized water; Bw1: Bw + DOM<3k; Bw2: Bw + DOM3k–10k; Bw3: Bw + DOM>10k; Bh: biochar washed with dilute hydrochloric acid; Bh1: Bh + DOM<3k; Bh2: Bh + DOM3k–10k; Bh3: Bh + DOM>10k. Values in the figure are presented as means (n = 3), and different letters indicate significant differences among treatments (p < 0.05). The same applies below.
During the incubation period, the available Cd content in the control (CK) soil remained at a high level (> 3.0 mg·kg–1). After adding biochar-DOM composites, all amended treatments exhibited a rapid decline in available Cd. By day 10, the available Cd content of all treatments was significantly lower than that in the control, with reductions ranging from 41.82% to 54.95%. The reduction was more pronounced with increasing DOM molecular weight; specifically, the Bw3 and Bh3 treatments reduced available Cd by 51.86% and 54.95%, respectively, compared to the control. By day 20, the acid-washed biochar-DOM composites (Bh1, Bh2, Bh3) demonstrated a significantly greater reduction in soil available Cd than the water-washed composites, achieving a decrease of 73.94% to 79.53% relative to the Bw. After 30 d, the effect of each treatment on soil Cd content stabilized. The available Cd concentrations across all treatments remained within a narrow range of 0.8–1.0 mg·kg–1, and there was no significant difference between the treatments.
In general, the effect of Bw and Bh composites on the content of available Cd in the soil was significantly different in the early stage of the experiment, but the difference was not obvious in the late stage of the experiment.
The changes in soil Cd fractions of each treatment are shown in Fig. 4. Compared to CK, all biochar-DOM composite treatments significantly reduced the content of soil water-soluble Cd, with reductions ranging from 28.53% to 52.84%. Among them, the Bh2 treatment showed the most pronounced effect. The specifically adsorbed Cd was significantly higher in Bh1–3 composite treatments than in the control, with an increase of 10.43% to 14.34%. However, there were no significant differences among treatments with different DOM molecular weights. No significant changes were detected in the contents of Cd bound to amorphous Fe oxides, crystalline Fe oxides, or organic matter across all treatments. The residual Cd fraction showed a significant difference after treatment, which was 28.82%–123.77% higher than that of the control, and increased with the increase of DOM molecular weight. Furthermore, for a given DOM molecular weight, the Bh composites consistently resulted in a higher residual Cd content than the Bw composites.
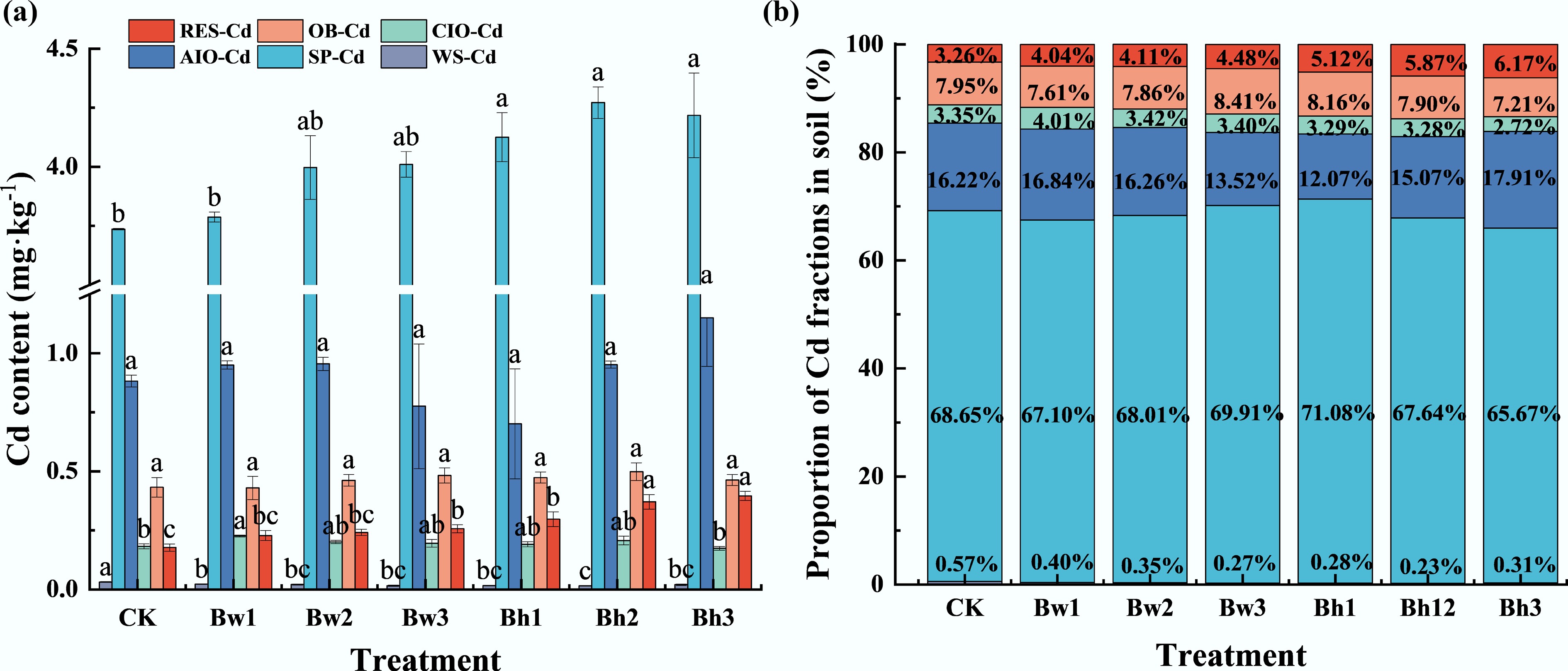
Figure 4.
Effects of different biochar-DOM composites with varying molecular weight ranges on soil Cd fractions. (a) represents the concentration of different forms of Cd in soil under different molecular weight biochar-DOM composites; (b) represents proportion of Cd fractions in soil under different molecular weight biochar-DOM composites.
Similarly, the proportion of different forms of Cd fractions also showed that the content of water-soluble Cd decreased after treatment. The proportion of water-soluble Cd decreased by 0.18%–0.34% relative to the control, while the proportion of residual Cd increased by 0.79%–2.92%.
Biomass and Cd uptake of Chinese cabbage in the pot experiment
-
Overall, the application of biochar-DOM composites had a limited impact on the yield of Chinese cabbage. A significant reduction in yield was observed only in the Bw3 and Bh3 treatments, which decreased by 28.00% and 60.00%, respectively (p < 0.05). This suggests that biochar composites incorporating high-molecular-weight DOM may inhibit the growth of Chinese cabbage.
The effects of different biochar-DOM composites on Cd uptake by Chinese cabbage plants are shown in Fig. 5. Compared to the control group, all biochar-DOM composite treatments significantly inhibited Cd accumulation in the shoots of Chinese cabbage. The Cd content in the shoots was reduced by 18.48%–74.46%. Among the treatments, Bw3 and Bh3 were the most effective, significantly reducing the Cd content in shoots of Chinese cabbage by 67.27% and 74.46%, respectively.
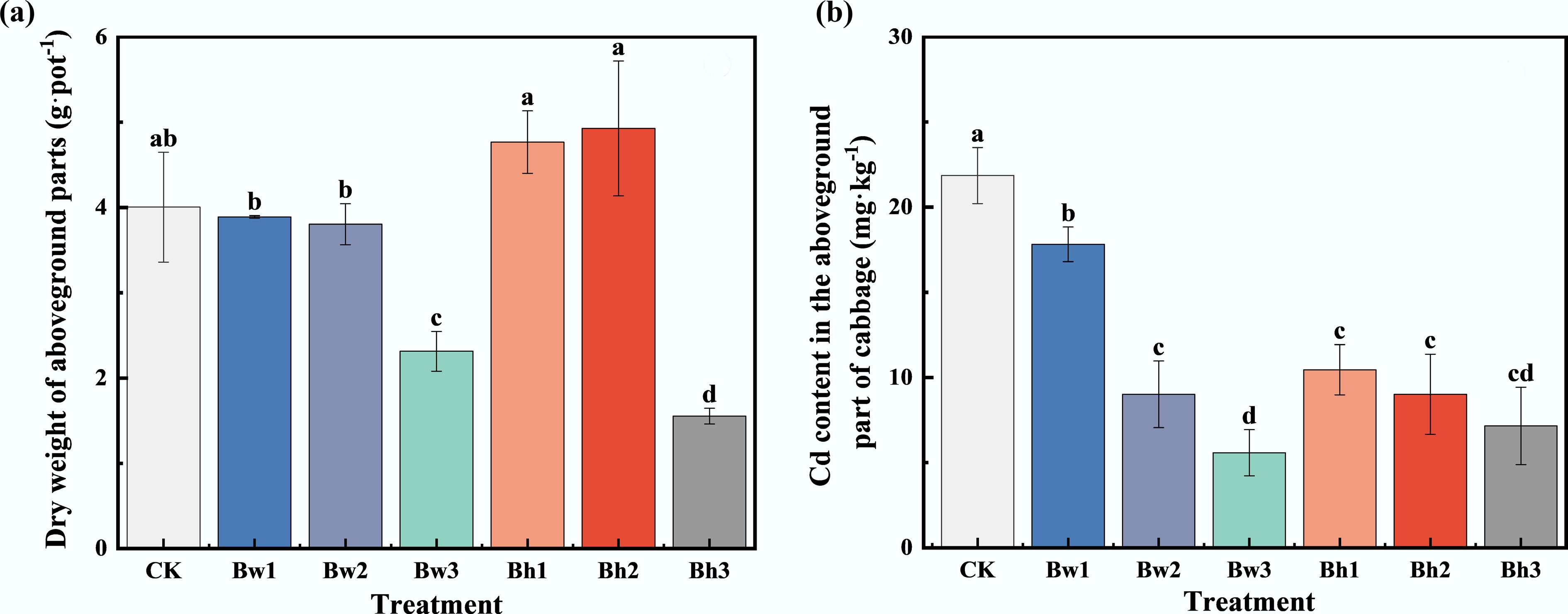
Figure 5.
Differences in (a) cabbage yield, and (b) Cd content with the addition of different biochar-DOM composites.
In addition, compared with Bw1–3 composites, the Bh1 (Bh incorporating low-molecular-weight DOM) composites had a more significant inhibitory effect on Cd uptake in the shoots. With the increase in the molecular weight of DOM, the inhibitory effect of biochar-DOM composites on Cd was more prominent.
The enrichment factor (EF) serves as an indicator to quantify the capacity of plants to accumulate heavy metals in soil (Fig. 6). Compared with the control (CK), only the Bw1 treatment exhibited no statistically significant difference (p < 0.05), and all other treatments decreased the EF by 50.72%–72.65%. For both water-washed and acid-washed biochar-DOM composites, the EF values decreased with increasing DOM molecular weight. These results indicate that biochar-DOM composites synthesized with higher molecular weight DOM can significantly reduce the accumulation of Cd in the aboveground tissues of plants.
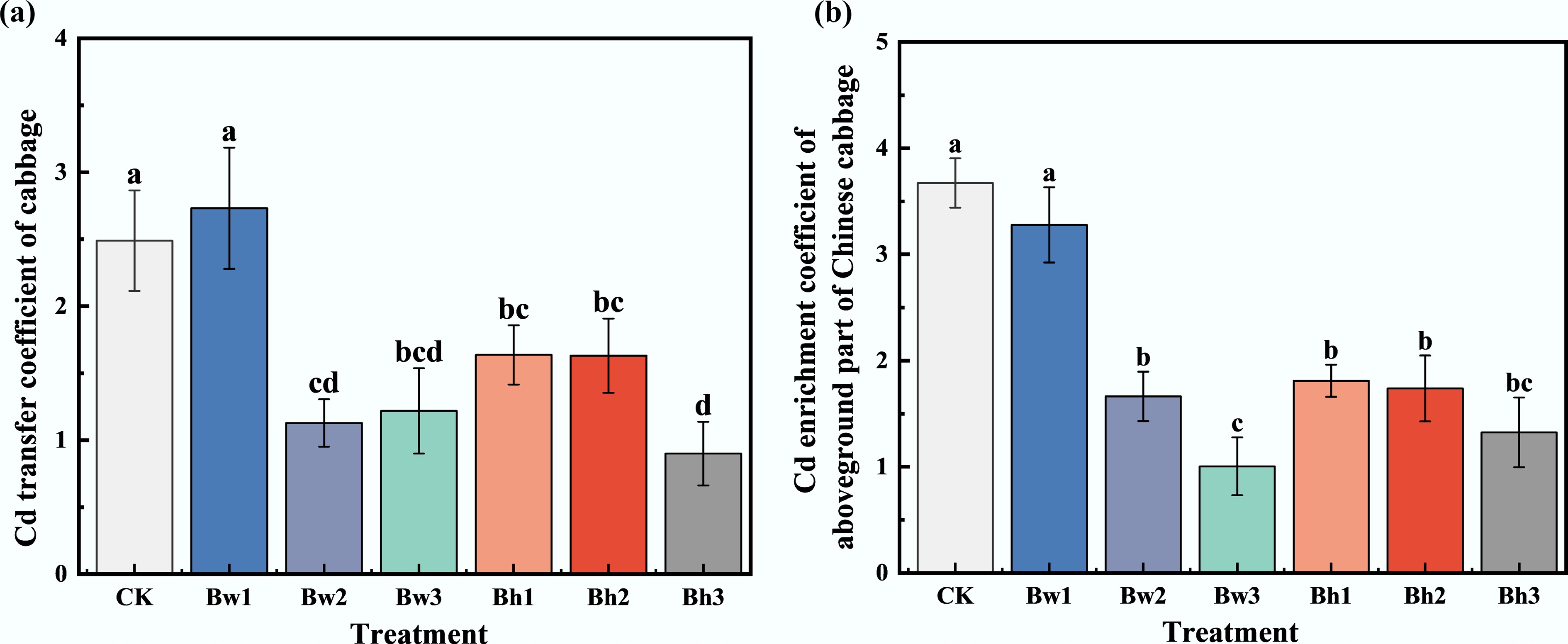
Figure 6.
Differences in (a) Cd transfer coefficient, and (b) enrichment coefficient of Chinese cabbage with different biochar-DOM composites.
The soil pH measured after harvesting cabbage in the pot experiment is presented in Fig. 7. Compared with CK, all treatments except Bw1 increased the soil pH by 0.53–1.13 units. Among them, the Bw3, Bw2, and Bh3 treatments resulted in a significantly higher soil pH (p < 0.05), reaching 7.68, 7.24, and 7.44, respectively, whereas the pH values in other treatments remained below 7. The results showed that biochar composites combined with higher molecular weight DOM were more effective in elevating soil pH.
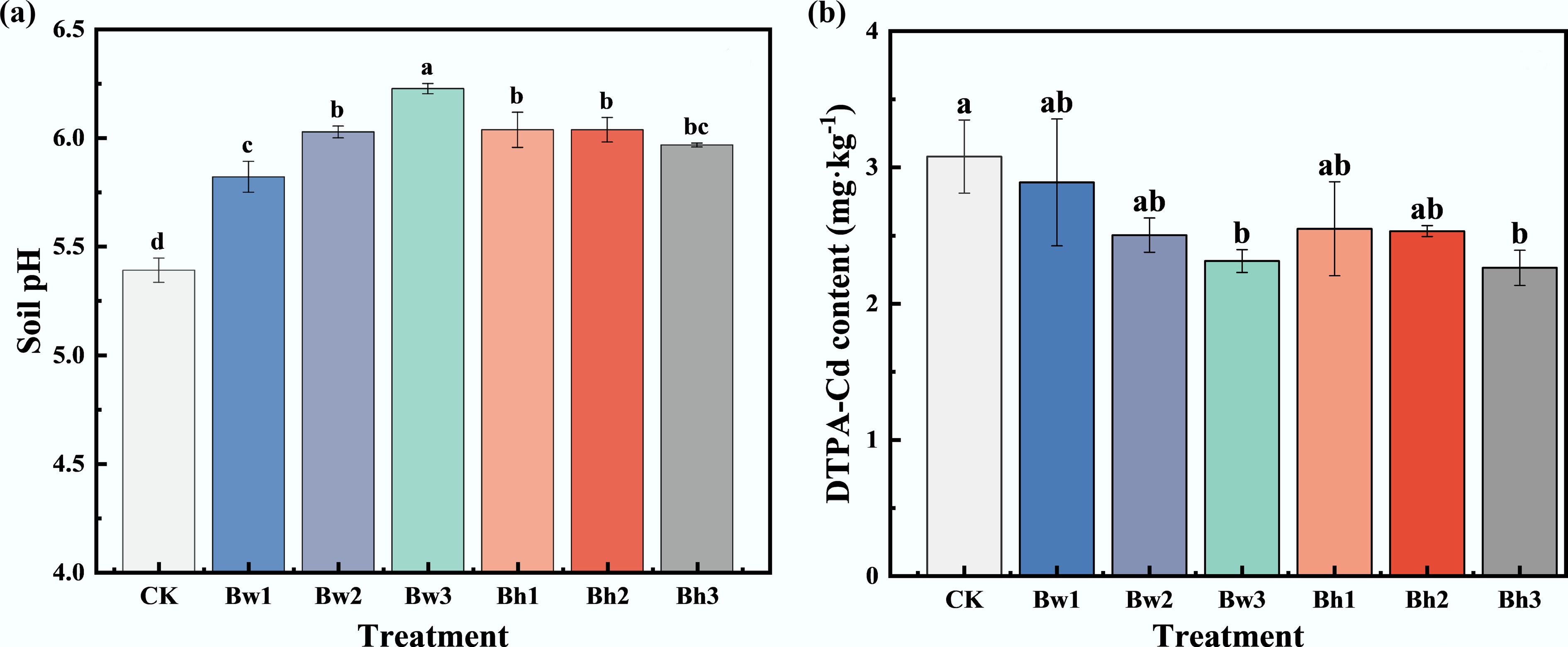
Figure 7.
Effects of different biochar-DOM composites on (a) soil pH, and (b) available Cd content in the pot experiment.
Fig. 7 also illustrates the changes in soil available Cd following the harvest of cabbage under different biochar-DOM composite treatments. Compared with CK, reductions in soil available Cd were statistically significant (p < 0.05) only for the Bw3 and Bh3 treatments, which decreased by 24.89% and 26.52%, respectively. For composite materials formed from either Bw or Bh with DOM of varying molecular weights, the reduction in soil available Cd became more pronounced as the molecular weight of the DOM increased.
Fig. 8 shows the changes in soil Cd fractions following cabbage harvest with the application of different biochar-DOM composites. Compared with CK, treatments with various biochar-DOM composites significantly (p < 0.05) reduced the contents of WS-Cd and SP-Cd. The most significant reduction in WS-Cd was observed in the Bw3 and Bh3 treatments, which decreased by 59.65% and 50.20%, respectively, relative to CK, while the other treatments showed no significant difference. For SP-Cd, the Bh1–3 composites were significantly lower than the control by 10.43%–14.34%, though no significant differences were detected among different molecular weight treatments. Across all treatments, no significant changes were observed in the contents of AIO-Cd, CIO-Cd, OB-Cd, or RES-Cd.
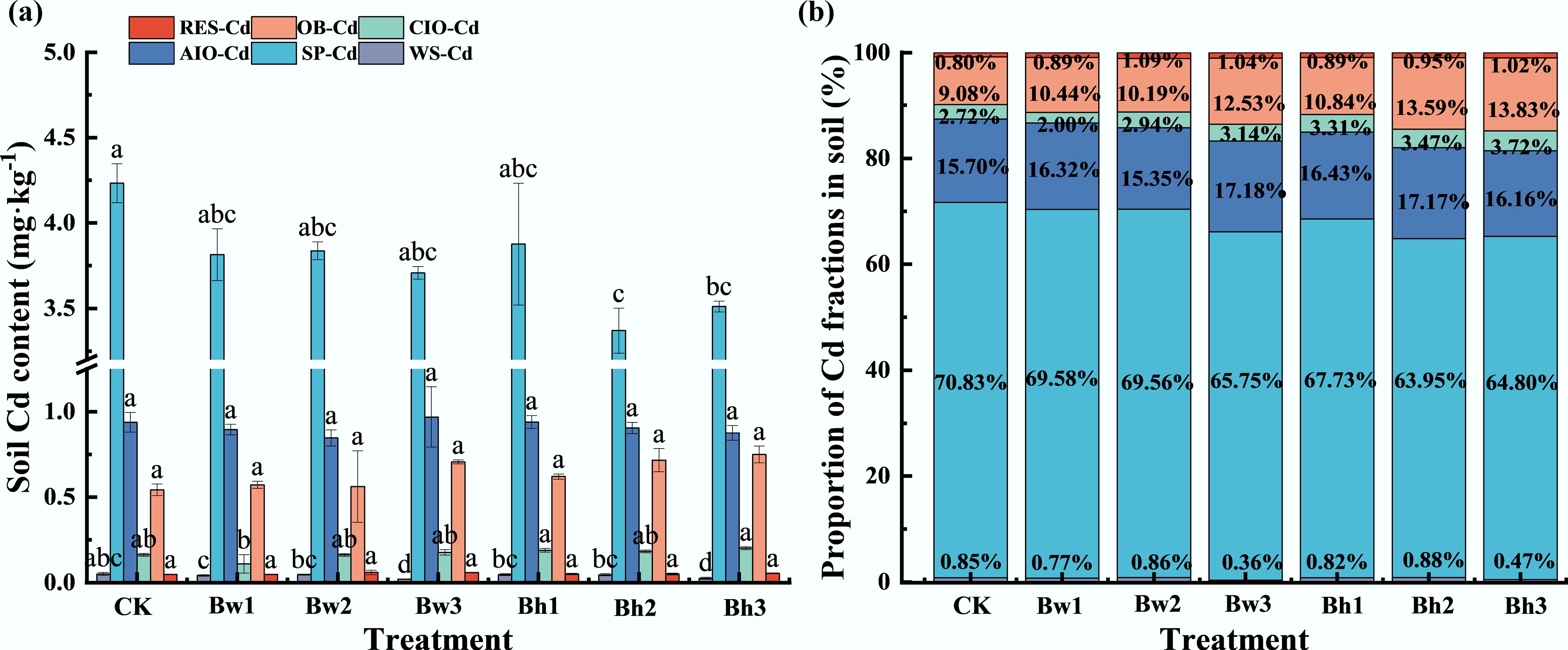
Figure 8.
Effects of different biochar-DOM composites on soil Cd fractions in the pot experiment. (a) represents the concentration of different forms of Cd in pot soil under different molecular weight biochar-DOM composites; (b) represents proportion of Cd fractions in pot soil under different molecular weight biochar-DOM composites.
The application of different biochar-DOM composites altered the proportional distribution of Cd fractions in soil, and the change trend was consistent with the concentration. Following treatment with either Bw or Bh-DOM composites, the proportion of WS-Cd decreased to a comparable extent, with mean reductions of 27.89% and 22.57%, respectively, relative to the control. This indicates a greater conversion of WS-Cd into other forms. The proportion of SP-Cd in the two composite treatments was also lower than that in the control, with a more significant decrease induced by Bh1–3 (a mean decrease of 15.29%). The proportions of CIO-Cd, OB-Cd, and RES-Cd all increased compared with the control. The acid-washed biochar-DOM composites led to more significant increases in CIO-Cd and OB-Cd (mean increases of 17.63% and 28.09%, respectively, relative to the control), whereas the Bw1–3 resulted in a greater increase in RES-Cd (mean increase of 16.46%).
Regarding the influence of DOM molecular weight on Cd fractions, higher molecular weight DOM exhibited a significant effect on the transformation of WS-Cd into other fractions. In Bh-DOM composites, treatments with larger molecular weight DOM further favored the transformation into CIO-Cd and OB-Cd. However, for Bw-DOM composites, larger molecular weight DOM primarily increased the proportions of AIO-Cd and OB-Cd.
Overall, composites formed with higher molecular weight DOM were more effective in reducing both the content and proportion of WS-Cd and SP-Cd, while increasing the proportions of OB-Cd and RES-Cd. These results suggest that the application of such organic amendments can promote the transformation of soil Cd from bioavailable to non-bioavailable forms, and then reduce the absorption of Cd by plants.
-
Previous studies[27−30] have indicated that acid-washed biochar alters biochar's adsorption mechanisms by enhancing complexation, π-electron interactions, and precipitation, while increasing oxygen-containing functional groups. In this study, all biochar-DOM composites exhibited significantly improved Cd2+ adsorption capacity, with high-molecular-weight DOM-biochar (Bh) composites showing the greatest enhancement. This may be attributed to the increased acidic functional groups (e.g., carboxyl and phenolic hydroxyl groups) on the Bh surface and the enrichment of oxygen-containing groups from acid washing, which is consistent with the FTIR results.
The dominant immobilization mechanism is molecular-weight-dependent: for high-molecular-weight DOM composites, π-electron coordination with aromatic structures predominates; for low-molecular-weight DOM composites, complexation with oxygen-containing functional groups plays a more significant role[31,32]. FTIR data support this distinction, showing minimal changes in Bh composites after Cd binding, suggesting π–π interactions rather than chemical bond formation as the primary driver.
Adsorption behavior further corroborates this mechanism. Low- and medium-molecular-weight DOM composites (Bw1, Bw2) followed the Freundlich model, indicating heterogeneous multi-layer adsorption through ion exchange and electrostatic attraction. This is consistent with findings that such DOM provides additional adsorption sites (e.g., citric acid) and facilitates Cd uptake through ion exchange and electrostatic attraction, despite its lower aromaticity[31,33,34]. In contrast, high-molecular-weight DOM composites conformed to the Langmuir model, suggesting monolayer adsorption on a homogenized surface, likely due to physical coverage of heterogeneous sites or micropore blocking by larger DOM molecules[35,36]. The stronger Cd affinity of humic-like, aromatic components in high-molecular-weight DOM further supports this finding.
The combination of DOM and biochar preserved surface functional groups, with interactions dominated by physical forces rather than chemical bonding. Overall, the adsorption mechanisms involve ion exchange, complexation with oxygen-containing groups, π-electron coordination, and mineral precipitation[37]. Low-molecular-weight DOM composites rely primarily on functional group complexation, while high-molecular-weight DOM composites depend mainly on π-electron coordination within aromatic systems[38,39]. This molecular-weight-dependent continuum governs the enhanced Cd immobilization by biochar-DOM composites.
Regulatory behavior of Bw/Bh-DOM composites on soil Cd2+
-
The reduction in soil available Cd by biochar-DOM composites operates through both direct and indirect mechanisms[40]. Initially, all biochar-DOM composites significantly decreased available Cd in soil primarily by increasing soil pH, which suppressed Cd2+ solubility through enhanced hydroxyl coordination and reduced ion activity[41]. This effect was particularly pronounced for composites formed with higher molecular weight DOM. Their greater aromaticity and hydrophobicity promote the formation of insoluble Cd complexes that further transform into stable fractions by hydrophobic interactions with soil particles[42,43]. Therefore, biochar-DOM composites synthesized with higher molecular weight DOM (e.g., the Bw3 treatment) exhibited the strongest immobilization capacity. However, with the passage of time, the immobilization rate slowed as adsorption sites became saturated, shifting toward chemical stabilization mechanisms[44].
The fraction of Cd in soil determines its bioavailability and environmental risk. Water-soluble Cd (WS-Cd) poses the highest biological toxicity, while residual Cd (RES-Cd) is largely inaccessible to plants[45,46]. Although low-molecular-weight DOM can form soluble complexes with Cd and potentially increase its mobility[6,47,48], all composites in this study significantly reduced WS-Cd and increased RES-Cd. Composites synthesized with higher molecular weight DOM (e.g., Bw3 and Bh3) demonstrated the strongest transformation capacity, as their highly aromatized, hydrophobic nature favors the formation of stable, insoluble Cd complexes[49,50]. This led to the most significant decrease in WS-Cd and a significant increase in RES-Cd.
Influence of Bw/Bh-DOM composites on Cd2+ toxicity in the soil–plant system
-
The effects of biochar-DOM composites on plant growth also exhibited molecular-weight-dependent patterns: high-molecular-weight DOM composites had a significant negative effect on above-ground biomass but most effectively reduced Cd accumulation in shoots and root-to-shoot translocation, whereas low/medium-molecular-weight composites showed no significant effect on plant growth. High-molecular-weight DOM might have a negative impact on root growth by inhibiting microbial activity[51,52], while applying an appropriate amount of low-molecular-weight organic acids to soil can promote plant uptake of heavy metal ions[53].
The composites reduced Cd bioavailability through direct adsorption[54] and by increasing soil pH[55−57], which suppressed Cd solubility. High-molecular-weight DOM composites most effectively decreased water-soluble Cd and increased residual Cd fractions by forming stable, insoluble complexes, while low-molecular-weight DOM tended to form soluble complexes potentially available for plant uptake. Notably, SP-Cd decreased in pot experiments, but increased in incubation experiments, likely due to root exudates modifying composite surfaces during plant growth[58].
Both incubation and pot experiments in this study demonstrate that the molecular weight of organic fertilizer-derived DOM critically governs the Cd immobilization capacity of biochar composites. High-molecular-weight DOM enhances adsorption via π-electron coordination and functional group complexation, thereby reducing Cd bioavailability and plant uptake. These findings highlight the critical role of DOM molecular weight in soil remediation and provide a theoretical basis for designing optimized composite amendments. However, the molecular-scale interactions require further spectroscopic validation, and findings are limited by single-source DOM and controlled pot conditions. Future research should validate sustained effectiveness through long-term field trials with cost-benefit analyses, and explore DOM from diverse sources to develop tailored remediation strategies.
-
The maximum adsorption capacity of modified biochar-DOM composites for Cd was significantly higher than that of biochar alone and increased with the molecular weight of the DOM. The primary mechanism governing Cd adsorption by these composites was coordination with π-electrons, followed by complexation with oxygen-containing functional groups.
Different biochar-DOM complex treatments could increase soil pH, reduce the content of available Cd, significantly decrease the content of WS-Cd, and increase the proportion of RES-Cd in soil.
The addition of biochar-DOM composites had no effect on cabbage yield, but significantly inhibited Cd uptake in the above-ground parts. With the increase of the molecular weight of the complex, the inhibitory effect of biochar-DOM complexes on Cd was more prominent. Biochar composites synthesized with higher molecular weight DOM were particularly effective in reducing both the translocation factor and the enrichment factor of Cd in plants.
The mechanism transition is governed by DOM molecular weight, aromaticity, functional groups, and environmental factors. This framework provides a theoretical basis for designing targeted composite formulations and preparation processes for site-specific soil remediation.
-
The authors confirm contributions to the paper as follows: all authors contributed to the study conception and design; Lan Wei: data collection and analysis; writing the first draft; Danni Liu: data collection and analysis; Weisheng Chen: data collection and analysis; Lianxi Huang: data collection and analysis; Shaojun Jiang: methodological approach; Xiaodong Zheng: methodological approach; Zhongzhen Liu: language editing and manuscript review; Yanhong Wang: data collection and manuscript review; all authors commented on previous versions of the manuscript. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets used or analyzed during the current study are available from the corresponding author upon reasonable request.
-
This study was funded by the Young Scientists Fund of the National Natural Science Foundation of China (Grant Nos 42307343, 42207316), Guangxi Key Natural Science Foundation (Guangdong-Guangxi Joint Fund Project) (Grant No. 2022GXNSFDA080008), and the Provincial Science and Technology Project of Guangdong (Key Assignment of Rural Science and Technology Commissioner) (Grant No. KTP20240938).
-
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
-
Full list of author information is available at the end of the article.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Wei L, Liu D, Chen W, Huang L, Jiang S, et al. 2026. Enhancement of organic fertilizer-derived dissolved organic matter fractions on cadmium immobilization by biochar composites in contaminated soil. Agricultural Ecology and Environment 2: e013 doi: 10.48130/aee-0026-0008
Enhancement of organic fertilizer-derived dissolved organic matter fractions on cadmium immobilization by biochar composites in contaminated soil
- Received: 30 January 2026
- Revised: 10 March 2026
- Accepted: 07 April 2026
- Published online: 06 May 2026
Abstract: The influence of DOM molecular diversity from organic fertilizers on Cd immobilization by biochar composites remains unclear. This study systematically investigated the preparation, adsorption characteristics, and mechanisms of biochar composites loaded with DOM of different molecular weights, as well as their effectiveness in remediating Cd-contaminated soil. It was found that Cd2+ adsorption by DOM-loaded biochar composites was significantly enhanced with increasing DOM molecular weight. The qmax values of biochar DOM>10k composites were 2.19% to 19.21% higher than those of DOM<3k and DOM3k–10k composites, respectively. The primary immobilization mechanisms were identified as π-electron coordination, and complexation with oxygen-containing functional groups. Soil incubation and pot experiments confirmed that the composites consistently increased soil pH (by 0.43–1.13 units) and reduced Cd bioavailability (by 70.70%–73.99% after 90 d in incubation; and by 6.17%–26.52% in pot experiments). Notably, they transformed Cd from exchangeable to more stable residual forms, with residual Cd increasing by 28.82%–123.77% in incubation and 1.46%–25.21% in pot experiments. High-molecular-weight DOM composites significantly reduced Cd uptake in Chinese cabbage shoots without affecting yield, primarily by inhibiting Cd translocation and accumulation in plants. This study elucidates the critical role of DOM molecular weight in governing biochar-based amendment performance, and provides a theoretical basis for designing targeted remediation strategies in heavy metal-contaminated agricultural soils.