-
Winter conditions are predicted to feature reduced protective snow cover, heightened severe storms, and more fluctuating temperatures in areas such as Michigan for overwintering crops such as winter wheat (Triticum aestivum)[1,2]. For agricultural fields, ice encasement can occur in large areas due to freezing rain or fluctuating temperatures that could melt and refreeze snow layers. Ice encasement can also occur in localized or problematic areas, such as those that are low-lying or poorly drained. Targeted management strategies to promote plant survival of ice encasement could reduce crop loss associated with widespread or localized ice encasement in challenging field or environmental conditions. If persistent, ice encasement is a severe stress that restricts essential plant needs, including light interception and gas exchange, and can result in hypoxic or anoxic conditions at the plant and soil interface[3]. For winter wheat, damage can occur to plants in as little as 7 d of ice encasement[4]. Research is needed to better understand management strategies to prevent winter wheat yield losses associated with this stress.
Various fall management and cultural strategies can be utilized to influence plant productivity and yield potential of cereal grasses, including planting rate, seeding rate, and seeding depth[5−7]. Deep planting was originally recommended in northern areas to increase winter survival of winter wheat[8]. This was prior to the increase in no-till practices, which became useful since stubble in fields served as windbreaks and snow-traps to better insulate young winter wheat plants over the winter. Within no-till production and due to the added protection of the stubble and snow cover, shallower planting depths were feasible and resulted in higher yields and better winter survival[9]. The recommended seeding depth in Michigan, for example, is 2.54–3.81 cm, but is often highly variable due to field conditions and equipment utilized[10]. Northern USA region farmers utilize a mix of both no-till and till-based winter wheat farming practices and therefore would benefit from additional insight into how seeding depth influences ice encasement survival.
Seeding depth ultimately impacts radicle, seminal root, coleoptile, and other embryonic structures' interactions with the soil and atmosphere environments. Therefore, the ability of embryo structures to access gases, water, and light is influenced by seeding depth and could play a role in photosynthetic productivity and efficiency. In winter wheat, emergence from deep planting was only partly explained by coleoptile length[11]. In wheat and barley (Hordeum vulgare), the length of elongating and primordial leaves was shorter when sown deeply[7]. If the seed is sown deeper than the coleoptile's length and the true leaf emerges below the soil, lack of access to photosynthetic inputs could occur due to the absence of available light and therefore inhibit photosystem II (PSII) function. In addition to emergence constraints, winter conditions like freezing soil or ice encasement could further reduce photosynthetic productivity.
Plant stresses are known to reduce photosynthetic efficiency[12]. A key contributor to photosynthetic efficiency is PSII functioning and chlorophyll fluorescence measurements, which are widely used to investigate PSII function in response to environmental conditions. Plastic responses of PSII are thought to play a major role in temperature acclimation processes in winter wheat[13]. One primary method for assessing PSII efficiency is the OJIP test, which is a chlorophyll fluorescence transient curve that measures: (O) initial fluorescence of a leaf in a dark-adapted state, (J and I) the intermediate phases of fluorescence after light has been received by chlorophyll and energy has started to transfer to PSII, and (P or often denoted as Fm) when PSII is fully saturated with light[14]. Whether planting depth impacts these four steps of the chlorophyll a fast induction curve (OJIP) parameters has not been investigated, and could be used as a proxy to indicate plant stress associated with planting depth. In wheat, OJIP parameters correlated with winter hardiness and were a reliable indicator of freezing tolerance[15]. OJIP test curves give information about photosystem II (PSII) functional antenna size and photochemistry, as well as the functioning of the entire photosynthetic electron transport[16].
The two genotypes used in this study were 'DynaGro 9242W' (DynaGro) and 'WhiteTail', which exhibit coleoptile lengths that are determined by specific gene alleles. DynaGro carries the Rht-1 allele, which is associated with a longer coleoptile. In contrast, WhiteTail possesses the Rht-2 allele, which leads to a shorter coleoptile. Coleoptile length plays a crucial role in seedling emergence, particularly under varying planting depths. Plant growth is influenced by short or deep planting depths. Deep crowned cultivars survived better than shallower-crowned cultivars in Nebraska[5]. However, not all winter or winter stresses are equal. It is possible that deeply planted or crowned genotypes may suffer to a greater extent with oxygen-limiting stresses such as waterlogging or ice encasement conditions. In field conditions prone to hypoxia, it is possible that shallower planted seeds or shallower crowning plant types may have greater survival. To date, no literature seems to be available on how seeding depth interacts with ice encasement survival or how seeding depth influences the photosynthetic efficiency of plants. As targeted and field condition-based management practices become more prevalent in agronomy, a better understanding of specific winter stresses and specific cultural practices is needed.
Therefore, this study investigated OJIP parameters during the seedling stage and during cold acclimation of two winter wheat genotypes (DynaGro and WhiteTail) sown at shallow and deep seeding depths (1.27 and 6.35 cm below the surface). Wheat genotypes contrasting in coleoptile length were utilized to determine whether coleoptile length and planting depth influence overwintering emergence and ice encasement survival. It was hypothesized that OJIP transients in shallow sown plants will exhibit higher photosynthetic efficiency during the seedling stage and acclimation due to more effective light capture. Additionally, it was also hypothesized that shallow planting depths or genotypes that elongate the first internode in response to deep planting may improve plant survival of ice encasement, which could be related to oxygen availability. This hypothesis was tested in controlled conditions to reduce variability associated with planting depth in field conditions.
-
Two genotypes of soft white winter wheat seeds were planted in pots, with one seed sown per pot (Deepot tree containers, 6 cm diameter by 35 cm depth; Stuewe and Sons, Tangent, OR). The pots contained sandy loam (69.5% sand, 14.9% silt, 19.2% clay) soil. Seeds were placed at a shallow depth of 1.27 cm or planted deeply at 6.37 cm below the soil surface. These planting depths were manually implemented with a ruler and a line marked on a dibble for precise seed placement. The genotypes used were Dyna-Gro 9242W and Whitetail. Dyna-Gro 9242W contains a dwarfing gene allele called reduced height 1 (Rht-1), which is associated with a longer coleoptile. Whitetail contains the Rht-2 allele resulting in a shorter coleoptile. All pots were treated with a fungicide treatment of mefenoxam (Subdue Maxx; Corteva Agriscience, Indianapolis, IN) as a preventative measure 2 d after seed planting (18 Dec 2023, for experiment one and 18 Apr 2024 for experiment two). Pots were then transferred to a vernalization room (LED 500 lumen light and an 8 h photoperiod) for eight weeks at 10 °C with removal on 16 Jan 2024 for experiment 1 and 13 May 2024 for experiment two. Upon removal from vernalization, they were transferred to a low-temperature growth chamber (LTCB-19; Biochambers, Winnipeg, Manitoba, Canada) to stimulate cold acclimation. The growth chamber conditions were stepped down from 25 °C for two weeks at 350 µmol·m−2·s−1 light level and 10/14 h photoperiod,15 °C for two weeks at 350 µmol·m−2·s−1 light level and 10/14 h photoperiod, 10 °C for two weeks (350 µmol·m−2·s−1 light level and 10/14 h photoperiod), 5 °C for one week (300 µmol·m−2·s−1 with 12/12 h photoperiod), 0 °C for one week at 300 µmol·m−2·s−1 light level and 8/16 h photoperiod, and then –1 °C for remainder of the winter treatment period with a light intensity of 300 µmol·m−2·s−1 8/16 h photoperiod.
Winter treatments
-
Winter treatments included temperatures set to −1 °C with either ice encasement or no ice encasement treatment. The ice encasement treatment was implemented by dipping the bottom of the pots into paraffin wax until a solid wax layer was formed and completely covered the bottom drainage portion of the pot to at least one inch up the side of the pot. An acetate sheet was wrapped around the top of the pot to serve as a structure to form and hold an ice layer that was two inches deep for each pot. Ice layers were imposed by misting deionized water at −1 °C for 5 min every 30 min for 2 d, or until an ice layer was formed. The acetate wrap allowed for inspection of ice layers, and misting was used as needed to replace ice lost to potential sublimation in the chamber. Plant leaves were visibly sticking out above the ice, so it is not ice encasement of the entire plant but of the lower leaves, stem, crown, and roots. The ice or no ice treatment periods were for 0, 7, or 14 d and occurred on the following dates: day zero on 13 Mar 2024 (experiment one) and 18 June 2024 (experiment two), day seven was on 20 Mar 2024 (experiment one) and 25 June 2024 (experiment two), and day 14 was on 27 Mar 2024 (experiment one) and 2 July 2024 (experiment two). On each respective duration of treatment, a randomly selected half of the plants (four replicates) from each treatment were taken and destructively sampled by removing acetate wraps right after ice encasement treatments and separating tissues into leaf, crown, and root prior to storing in the freezer on the same day. Samples were stored as separate leaf, crown, and root tissues in a low-temperature freezer (−80 °C) for further analysis of malondialdehyde content, while the other half of the plants (four replicates) went through de-acclimation and were moved to a greenhouse for growth and yield measurements.
OJIP transients and measurements
-
Plants were subjected to overnight dark adaptation to ensure that all photosynthetic reaction centers were in a relaxed state. The most mature leaf on the main stem was selected for measurements. A dark adaptation leaf clip was used to ensure darkness during the time of measurement. Fluorescence measurements were taken weekly during the cold acclimation process of temperature adjustments from 25, 15, 10, 5, to 0 °C (Table 1). During experiment one, measurements were taken on 23 Jan 2024, 30 Jan 2024, 20 Feb 2024, 1 Mar 2024, and 8 Mar 2024, respectively. Second-year measurements were taken on 17 May 2024, 24 May 2024, 31 May 2024, 7 June 2024, and 14 June 2024 during the same cold acclimation treatment process. Fluorescence measurements were conducted using a pulse-modulated chlorophyll fluorometer (OS5p+; Opti-Sciences, Inc., Hudson, NH, USA). OJIP fluorescence transients were recorded based on the rapid rise of in vivo chlorophyll fluorescence quantum yield following the initiation of actinic light. OJIP parameters and calculations were performed based on the methods and formulas outlined by Strasser et al.[14] and Kalaji et al.[17].
Table 1. OJIP parameters with calculations and descriptions.
Parameter Calculation Description Fv/Fm (Fm − Fo)/Fm Ratio of variable fluorescence (Fv) to maximal fluorescence (Fm) indicating the maximum quantum yield of primary photosystem II (PSII) photochemistry. F0 is the minimal fluorescence. Fv/Fo (Fm − Fo)/Fo Proportional to the activity of the water-splitting complex on the donor side of the PSII. VJ (Fj − Fo)/(Fm − Fo) Relative variable fluorescence at 2 ms (Vj). For unconnected PSII units, this equals the fraction of closed RCs expressed as proportion of the total number of RCs (QA reduction). VI (Fi − Fo)/(Fm − Fo) Represents probability or efficiency of electron transport from specific electron carrier to a subsequent carrier leading to the reduction at the end of electron acceptor (QA turnovers). ET/TR (φ0) 1 − VJ Electron transport (ET) to trapping flux (TR) ratio indicating the probability that an electron residing on QA will enter the electron transport chain. ET/ABS (φE0) [(1 − Fo/Fm)](1 − VJ) Quantum yield for electron transport – how effectively absorbed light is utilized for electron transport. ABS is the absorption flux. RE/ET(δR0) (1 − VI)/(1 − VJ) Measures the efficiency with which an electron is transferred from the reduced intersystem electron acceptors to the final Photosystem I (PSI) end acceptors Do (φDo) 1 − Fv/Fm The fraction of light energy absorbed by chlorophyll molecule that is not used for photochemistry. Recovery and yield
-
Prior to recovery assessment in the greenhouse, all plants went through step-up temperature increases for de-acclimation processes to occur in the plants. This included adjusting chamber temperatures every two weeks with temperature steps of –1 to 4 to 10 to 15 °C with a light level of 300 μmol·m−2·s−1 and then to greenhouse conditions (23 °C, a nighttime temperature of 16 °C, a 14-h photoperiod, and an average light intensity of 400 µmol·m−2·s−1). The greenhouse was supplemented with LED light fixtures (Phillips GreenPower Toplights DRW_MP). Recovery from the winter treatments was analyzed for plants with and without ice encasement weekly following each respective treatment period (day zero, seven, or 14) across both years. Digital images were taken in a light box weekly for six weeks during the de-acclimation and greenhouse recovery period to determine leaf area regrowth over time. Recovery analysis took place from 14 Mar 2024 through 1 May 2024, for experiment one, and 18 June 2024 through 7 Aug 2024, for experiment two. Leaf area (cm2) was estimated using digital image analysis software (ImageJ, version 1.8.0)[18]. Biomass and yield were assessed upon reaching full maturity (Feekes 11.4), with plant organs separated and weighed on a mass balance for spikes, tillers, and roots. Subsequently, spikes were harvested to determine grain weight for yield determination.
Malondialdehyde content
-
Lipid peroxidation level was determined on plant parts harvested after winter treatments and based on malondialdehyde (MDA) content by using the method of Dhindsa et al.[19] and Zhang et al.[20]. The extraction solution was prepared by making a 50 mM phosphate buffer with pH 7.0 and 1% polyvinylpolypyrrolidone. Leaf, crown, and root samples were separated from plants and submerged in liquid nitrogen. Approximately 0.250 g of tissue was ground with a homogenizer (1,600 MiniG; SPEX SamplePrep LLC, Metuchen, NJ, USA) to a fine powder. The ground tissues were then centrifuged for 20 min at 15,000 g at 4 °C. The Supernatant was saved by decanting into a new centrifuge tube. A total of 100 mL of enzyme solution containing 20% trichloroacetic acid (TCA) and 0.5% thiobarbituric acid (TBA) was prepared. Next, 2.0 mL of enzyme solution was added to centrifuge tubes with tissues. The mixture was heated in a water bath at 95 °C for 30 min. The mixture was quickly cooled in an ice-water bath to room temperature. Then, it was centrifuged at 10,000 g for 10 min. A 1mL aliquot of supernatant was taken and measured at 562 and 600 nm wavelengths to determine the absorbance using a spectrometer (Genesys 10S ultraviolet-VIS; Thermo Fisher Scientific Inc., Waltham, MA, USA). The concentration of MDA was calculated using its extinction coefficient of 155 mM−1·cm−1[19].
Experimental design and statistical analysis
-
The experimental design was a completely randomized design with four replications. Fixed effects were genotype, depth (1.27 or 6.37 cm), treatment (ice or no ice), duration under treatment (0, 7, 10, or 13 d), and week of recovery. The experiment was repeated twice in the same growth chamber, where pots were randomly placed in the growth chamber. There was no interaction with experiments, so the data were pooled from both years and presented collectively. Normality was assessed using visual analysis of histograms and residual plots. Homogeneity of error was assessed using Levene's test. Data was analyzed using analysis of variance (ANOVA, RStudio ver. 4.2.1, Boston, MA, USA) with a linear mixed model to determine the main and interacting effects of the experimental factors. Mean separations were performed using Fisher's protected least significant difference test at the p ≤ 0.05 level.
-
Temperature influenced all parameters measured using the OJIP test (Table 2). Fluorescence values for maximum quantum yield of PSII (Fv/Fm), potential photochemical efficiency (Fv/Fo), efficiency of reduced electron acceptors (RE/ET), probability of electrons entering the transport chain (ET/TR), and quantum yield for electron transport (ET/ABS) all exhibited a decline with decreasing temperatures of 25, 15, 10, 5, and 0 °C (Table 3). Both the maximum quantum yield of PSII (Fv/Fm) and the activity of the water splitting complex (Fv/Fo) showed a significant decline as the temperature decreased from 25 to 15 °C/10 °C, with the lowest fluorescence values observed at 5 °C and 0 °C. This indicates impaired efficiency in converting light into chemical energy. Similarly, from decreasing temperatures of 25, 15, 10, 5, and 0 °C, ET/TR and ET/ABS diminished, indicating reduced energy transfer to the electron transport chain during cold acclimation. ET/TR demonstrated no difference between 25, 15, and 10 °C, but significantly declined at 5 and 0 °C. For ET/ABS, the highest fluorescence values were observed at 25 °C, with a significant decrease at 15 and 10 °C, with the lowest values recorded at 5 °C and 0 °C. Additionally, the efficiency of electron movement to reduce end electron acceptors (RE/ET) declined as temperatures dropped. The lowest significant values for RE/ET were at 5 °C and 0 °C.
Table 2. Analysis of variance for OJIP parameters including Fv/Fm, Fv/Fo, VI, VJ, RE/ET, ET/TR, ET/ABS, ET/TR, and Do in response to genotype, depth, and acclimation temperatures (25, 15, 10, 5, and 0 °C) in growth chamber conditions.
Effect Fv/Fm Fv/Fo Vi Vj RE/ET ET/TR ET/ABS Do Genotype (G) NS NS NS NS NS NS NS NS Depth (D) NS NS ** *** NS *** * NS Temperature (T) ** ** *** *** *** *** *** ** G × D NS NS NS NS NS NS NS NS G × T NS NS NS NS NS NS NS NS D × T NS NS NS * NS * NS NS G × D × T NS NS NS NS NS NS NS NS NS = non-significant (p ≤ 0.05); *, **, *** significant at p ≤ 0.05, p ≤ 0.01, p ≤ 0.001, respectively. Table 3. Relative fluorescence responses from parameters Fv/Fm, Fv/Fo, VI, VJ, RE/ET, ET/TR, ET/ABS, ET/TR, and Do across acclimation temperatures 25, 15, 10, 5, and 0 °C in growth chamber conditions. Letters are based on Fisher's least significant difference values (p ≤ 0.05).
Temperature
(°C)Fv/Fm Fv/Fo VI VJ RE/ET ET/TR ET/ABS Do 25 0.75 a 3.17 a 0.51 b 0.30 b 0.70 ab 0.70 a 0.92 a 0.25 c 15 0.69 b 2.68 b 0.49 b 0.34 b 0.77 a 0.66 a 0.90 b 0.30 b 10 0.69 b 2.64 b 0.50 b 0.33 b 0.75 a 0.67 a 0.89 b 0.31 b 5 0.68 bc 2.40 bc 0.65 a 0.43 a 0.60 c 0.57 b 0.86 c 0.32 b 0 0.64 c 2.14 c 0.63 a 0.43 a 0.64 bc 0.57 b 0.84 c 0.35 a Relative parameters at the I-step (VI) and at the J-step (VJ) increased as temperatures increased to 25, 15, and 10 °C, to 5 and 0 °C (Table 3), suggesting enhanced trapping of energy due to the temperature change, without the ability to move them down the electron transport chain. Both VI and VJ had significantly higher fluorescence at 5 and 0 °C when compared to 25, 15, and 10 °C. Do additionally increased with temperature (Table 3), indicating an enhanced level of non-photochemical energy dissipated under conditions of acclimation. Do at 25 °C had the lowest fluorescence, and at 15, 10, and 5 °C there were no significant changes. At 0 °C, Do was significantly higher when compared to all other temperatures during acclimation.
The seeding depth of either 1.27 or 6.35 cm resulted in significant differences in relative fluorescence parameters, specifically in ET/ABS, ET/TR, VI, and VJ (Fig. 1). The 6.35 cm seeding depth exhibited higher response values for ET/ABS and ET/TR when compared to 1.27 cm. This enhancement may be attributed to the later emergence associated with this depth, allowing newly emerged leaves to function at a higher level of photosynthesis during early acclimation. Conversely, plants seeded at the 1.27 cm depth exhibited higher VI and VJ values than the 6.35 cm, suggesting that at a shallower depth, the reaction centers were more effectively closed and able to trap energy (Fig. 1). However, this did not translate into greater energy utilization, as ET/ABS and ET/TR were higher in plants seeded at the deeper depth of 6.35 cm. This means that deeper-seeded (6.35 cm) plants were more photosynthetically active during acclimation compared to the shallow-seeded (1.27 cm) plants.
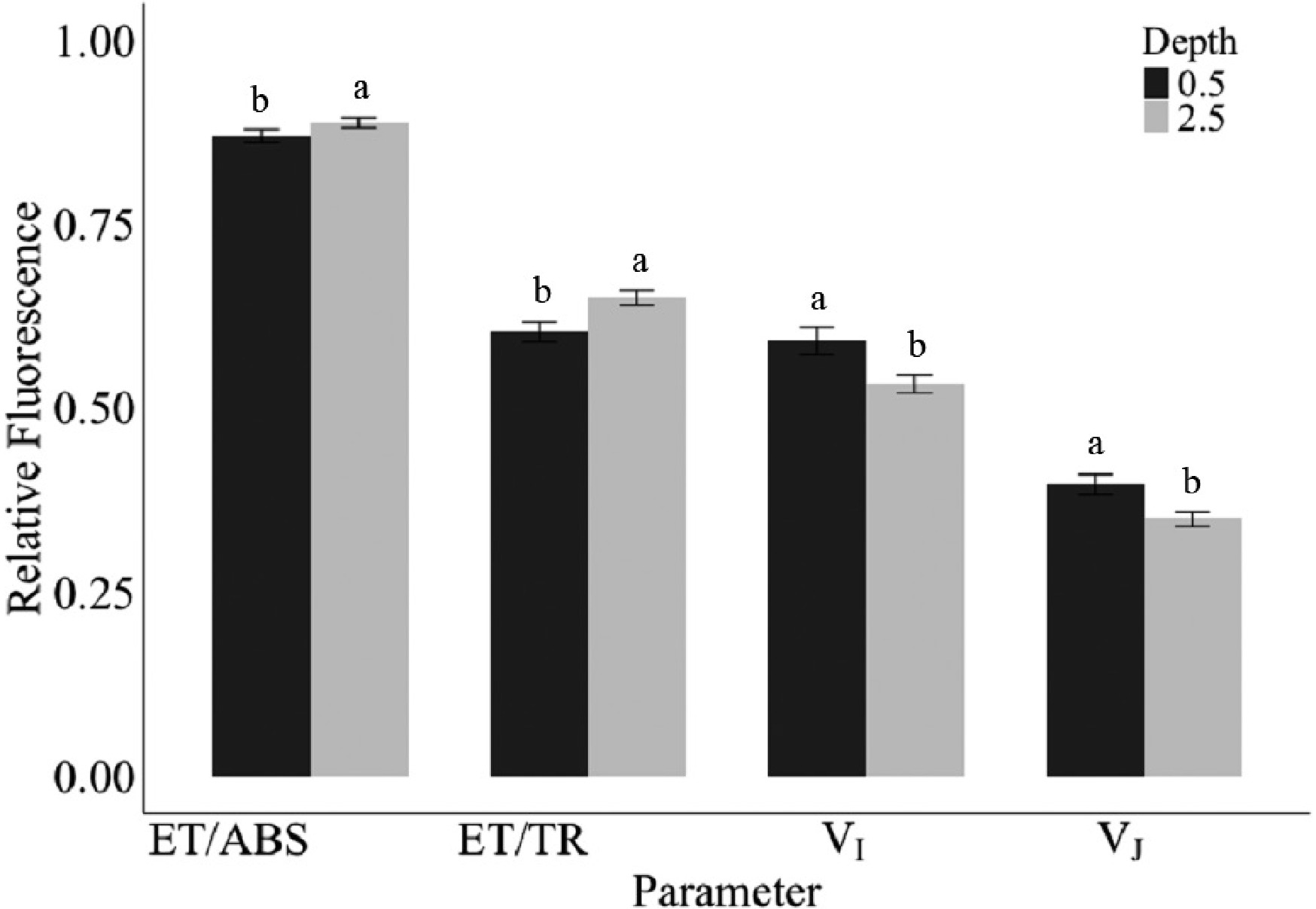
Figure 1.
Average relative fluorescence responses from parameters ET/ABS, ET/TR, VI, and VJ at seeding depth of 0.5-inch (black) and 2.5-inch (grey) across acclimation temperatures 25, 15, 10, 5, and 0 °C in growth chamber conditions. Error bars represent standard error. Letters are based on Fisher's least significant differences between depths on each parameter (p ≤ 0.05).
Leaf area
-
Ice encasement treatment had a negative effect on leaf area during each duration of treatment when compared to the control (day zero; cold acclimated only). Plants subjected to low temperatures (no ice) on day seven and day 14 of treatment consistently displayed larger leaf area when compared to ice encasement treated plants on the same respective days (Fig. 2a). After 7 d of ice encasement, the leaf area was the smallest compared to day zero and day 14. The plants that did not have ice encasement had no significant differences between sampling days. Additionally, on day seven, the interaction between genotype and treatment was significant (Fig. 2b), with DynaGro and WhiteTail displaying larger leaf areas when not under ice encasement.
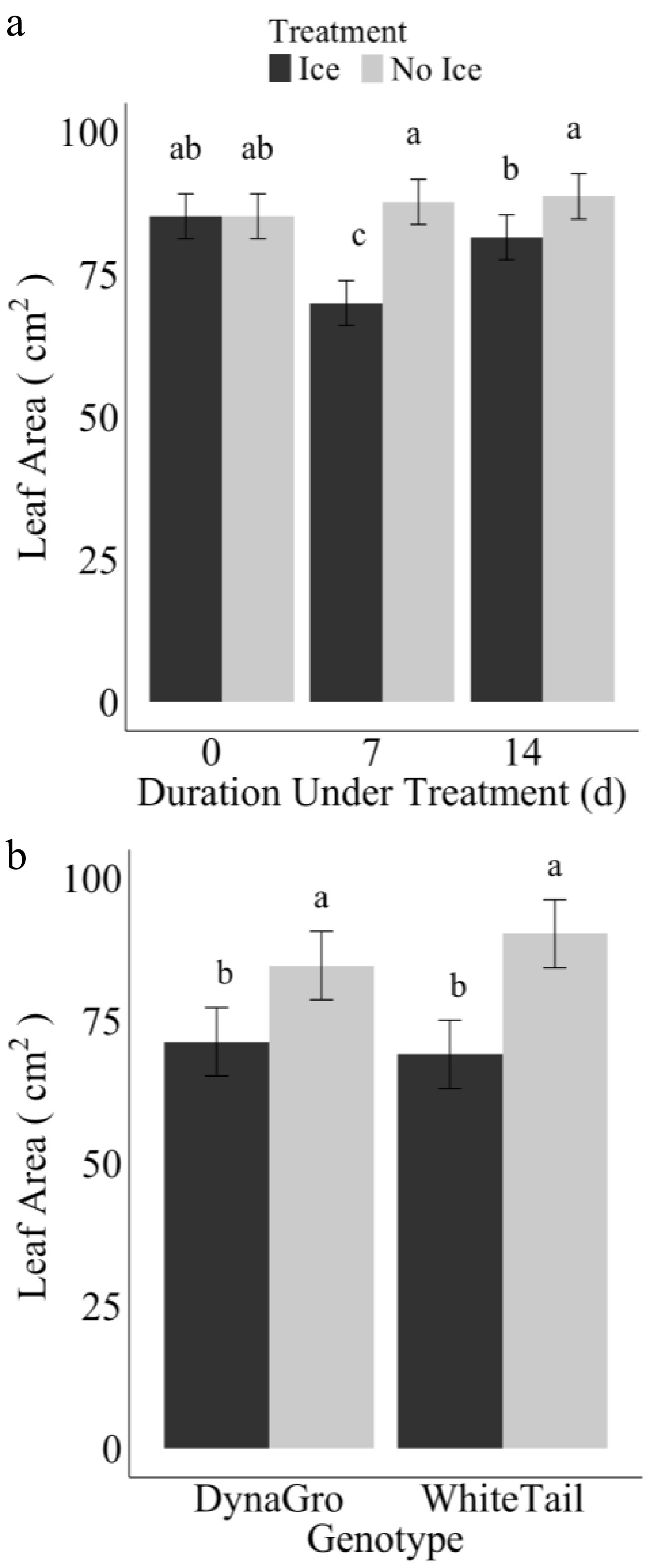
Figure 2.
Average leaf area (cm2) of winter wheat plants after ice or no ice treatment (−1 °C) for duration of (a) 7 or 14 d, and (b) the interaction of genotypes DynaGro and WhiteTail and treatment of ice (dark grey) and no ice (light grey) on day seven after treatment during de-acclimation (4, 10, and 15 °C) in growth chamber and greenhouse conditions (24 °C). Error bars represent standard error. Letters are based on Fisher's protected least significant difference for the comparison of means between treatment and genotype (p ≤ 0.05).
The main effect of seeding depth influenced leaf area throughout all treatment durations (Fig. 3), with plants seeded at 1.27 cm depth consistently exhibiting larger leaf areas than the plants that were seeded at 6.35 cm depth on all treatment durations. Genotype additionally influenced leaf area. On day seven, WhiteTail at the 1.27 cm depth yielded larger leaf area when compared to WhiteTail at 6.35 cm depth, but not significantly different than DynaGro at either seeding depth (1.27 or 6.25 cm). However, by day 14, DynaGro genotype had numerically the highest leaf area compared to WhiteTail when planted at 1.27 cm, although not significantly larger. Both genotypes (DynaGro and WhiteTail) planted at a seeding depth of 1.27 cm continued to exhibit larger leaf area compared to the 6.35 cm depth. This shift suggests that over prolonged durations of ice encasement, the choice of variety plays a role in determining growth outcomes.
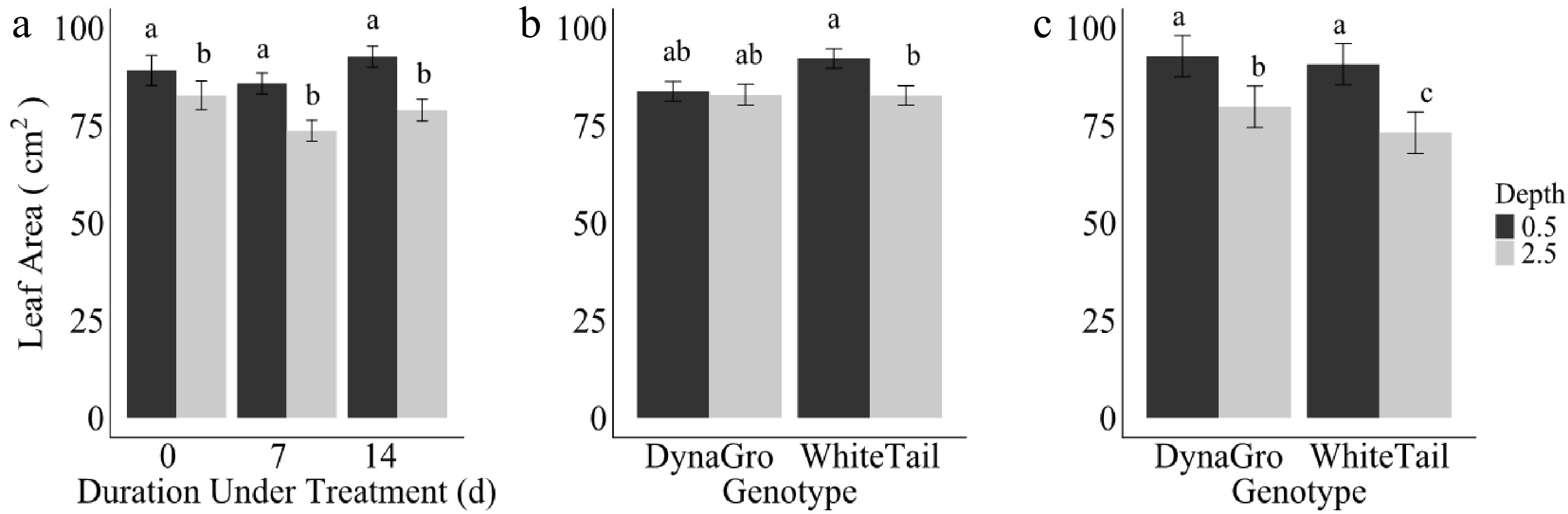
Figure 3.
Average leaf area (cm2) for (a) main effect of depth at 1.27 cm (dark grey) or 6.35 cm (light grey) for durations 0, 7, and 14 d, (b) interaction between genotypes DynaGro and WhiteTail recovery after treatments (−1 °C) during de-acclimation (4, 10, and 15 °C) in growth chamber and greenhouse conditions (24 °C) of winter wheat plants on day zero, and (c) day 14. Error bars represent standard error. Letters are based on Fisher's least significant difference (LSD) mean comparisons (p ≤ 0.05).
Plant biomass and yield
-
Biomass was influenced by tissue type between spikes, tillers, or roots in genotype, treatment, and seeding depth (Table 4). Tiller and root tissues were significantly influenced by genotype, with DynaGro genotype yielding significantly higher biomass for both tissue types compared to WhiteTail (Fig. 4). In both genotypes, tillers had this higher biomass compared to the root tissues. Seeding depth of either 1.27 or 6.35 cm influenced biomass in tissue types, particularly in spikes and tillers, with a 0.5-inch depth producing a larger biomass than 6.25 cm. The tiller tissues displayed higher biomass compared to spikes (Fig. 4b). Furthermore, ice encasement influenced spike and tiller biomass, with plants under ice encasement having significantly lower biomass compared to the no ice encasement treated plants (Fig. 4c). Under both no ice and ice treatments, tillers yielded higher biomass than spikes. Overall, the tillers were the tissue type that had the highest biomass when compared to spikes and roots. Duration under treatment additionally had an impact on biomass, with longer treatment durations of 7 or 14 d yielding lower biomass for both DynaGro and WhiteTail genotypes (Fig. 5). However, DynaGro displayed the highest biomass overall. In all tissue types, except for spikes in DynaGro, where there was no difference in biomass, the duration of stress had a negative influence on biomass, with day 14 yielding the lowest biomass in all. This indicates that extended ice encasement durations generally lead to decreased biomass.
Table 4. Analysis of variance for leaf area, malondialdehyde (MDA) content, biomass, and yield for main treatment factors of Genotype (G), Depth (D), Recovery period (R) and Ice Treatment (T) on day zero, seven, or 14 during growth recovery for winter wheat plants during de-acclimation (4, 10, and 15 °C) in growth chamber and greenhouse conditions (24 °C). Recovery rate analysis of variance for Genotype (G), Duration Under Treatment (R), Ice Treatment (T), and Depth (D) under same conditions. Using mixed model procedure.
Effect Leaf area MDA Biomass Yield Day zero Day seven Day 14 Leaf Crown Root Tillers Spikes Root Genotype (G) NS NS * NS NS NS *** NS *** * Depth (D) * *** *** NS NS NS ** ** NS ** Ice Treatment (T) NS *** *** NS NS NS * *** NS *** Duration Under Treatment (R) *** *** *** *** ** *** *** * *** NS G × D NS NS NS NS NS NS NS NS NS NS G × T NS NS NS NS NS NS NS NS NS NS D × T NS NS NS NS NS NS NS NS NS NS G × R NS NS NS NS NS NS ** NS * NS D × R NS NS NS NS ** NS NS NS * * T × R NS NS ** NS NS NS NS ** NS * G × D × T NS NS NS NS NS NS NS NS NS NS G × D × R NS NS ** NS NS NS NS * * * G × T × R NS NS NS NS NS NS NS NS NS NS D × T × R NS NS NS NS NS NS NS NS NS NS G × D × T × R NS NS NS NS NS NS NS NS NS NS NS = non-significant (p > 0.05); *, **, *** significant at p ≤ 0.05, p ≤ 0.01, p ≤ 0.001, respectively. 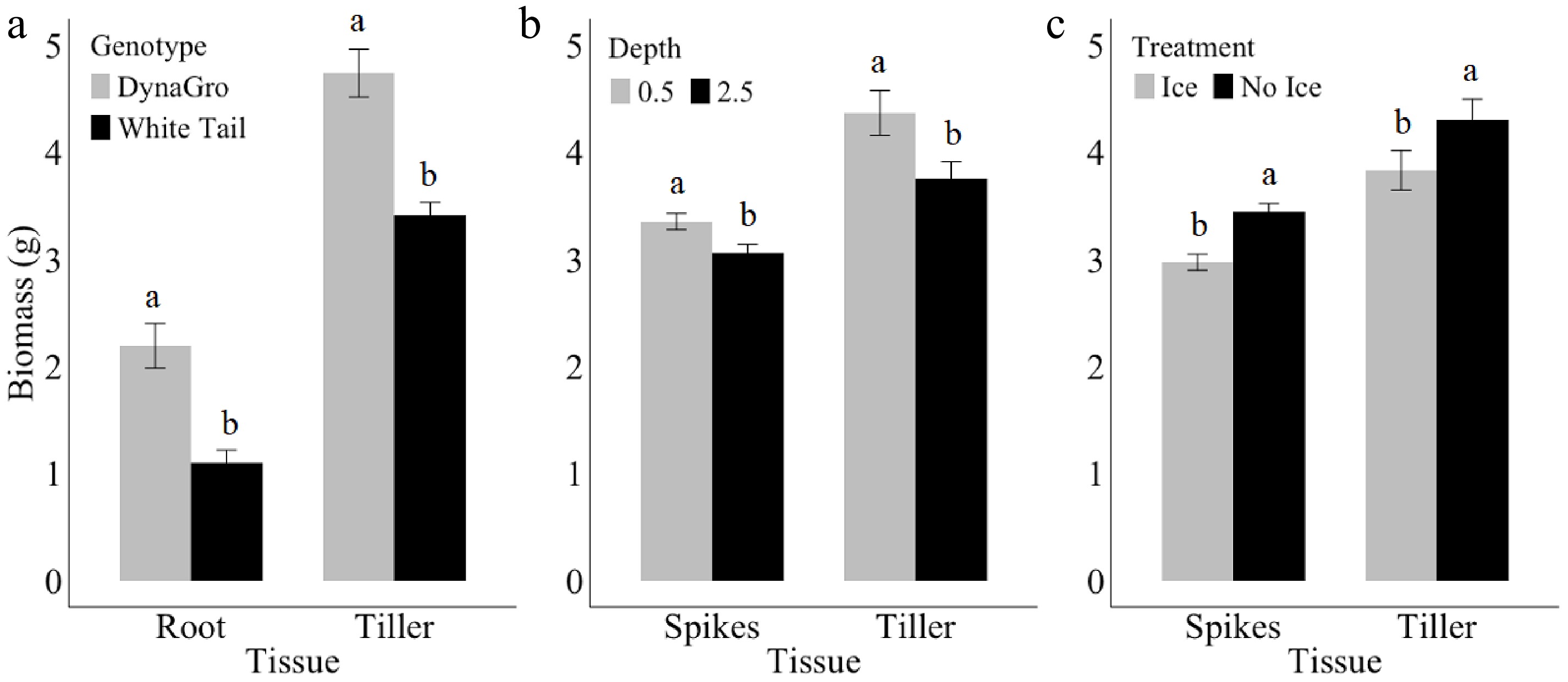
Figure 4.
Biomass (g) of (a) roots and tillers in DynaGro (grey) and WhiteTail (black), (b) spikes and tillers in 1.27 or 6.37 cm seeding depth, and (c) between ice (grey) and no ice (black) treatment in spikes and tillers. Error bars represent standard error. Letters are based on Fisher's least significant difference (LSD) mean comparisons (p ≤ 0.05).
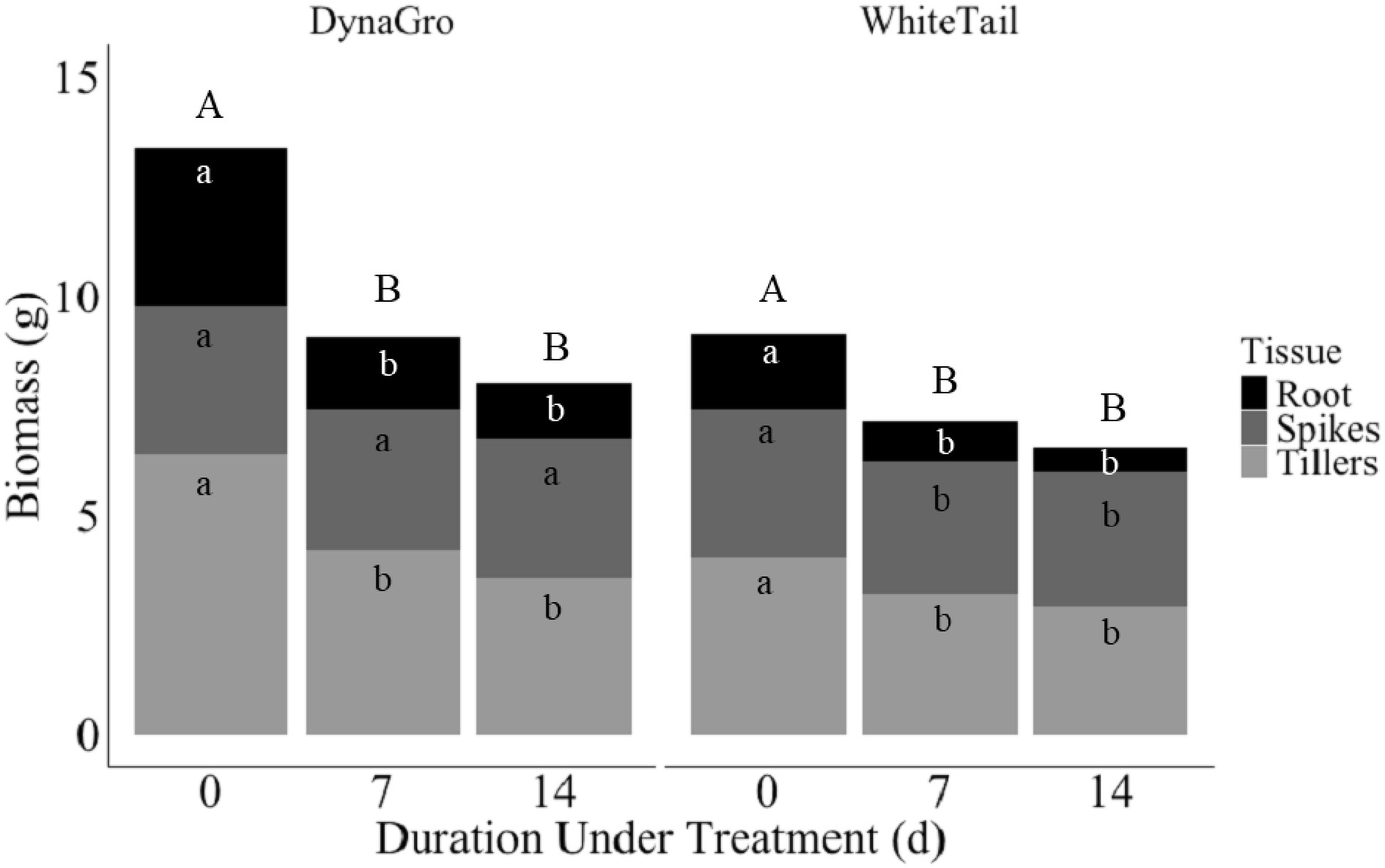
Figure 5.
Biomass (g) in DynaGro (left) and WhiteTail (right) across duration under treatment of 0, 7, or 14 d in root (black), spikes (dark grey), and tillers (light grey). Uppercase letters indicate Fisher's least significant difference between duration of treatment, white lowercase letters represent Fisher's least significant difference between root, and grey lowercase letters represent Fisher's least significant differences between spikes or tillers at p ≤ 0.05. Mean of two sowing depths.
Seven days of ice encasement impacted yield at the 1.27 cm depth, but not at the 6.35 cm depth (Fig. 6). After 14 d under treatment, the yield regained and was not significantly different than the control plants (Fig. 6a). In 6.35 cm seeding depth plants, there were no significant differences in yield between durations 0, 7, or 14 d under treatment. While there was more variation between the duration of treatments in yield for 1.27 cm compared to 6.35 cm, overall, the 1.27 cm depth seeded plants did have the numerically highest yield. Yield was additionally impacted by treatment of either ice or no ice encasement over treatment duration (Fig. 6b). Ice encasement treatment had a negative impact on yield compared to the no ice treatment with longer durations of treatment. On day zero, there were no differences between the yield for ice and no ice treatments, but by day seven and 14, no ice treatments had significantly higher yield compared to the ice treatments.
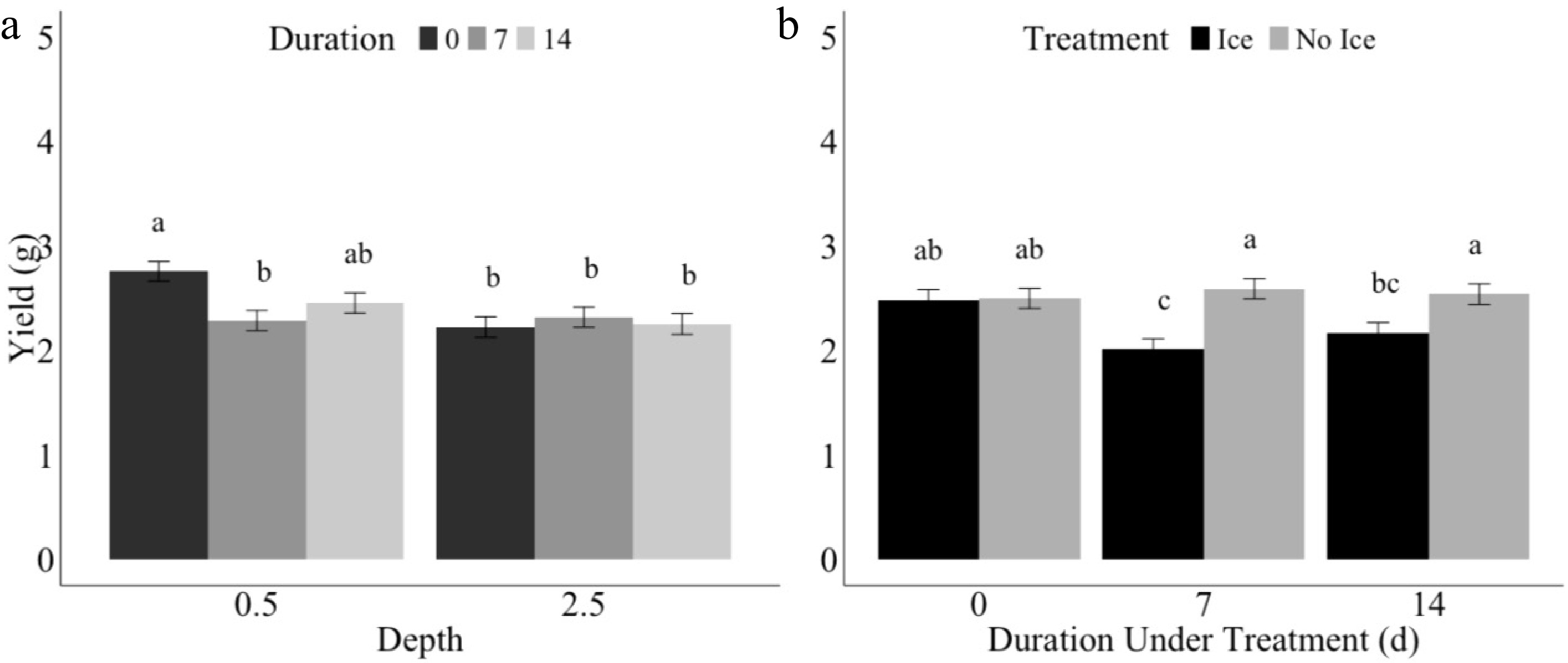
Figure 6.
Yield (g) of winter wheat plants sown at (a) 1.27 or 6.35 cm after 0 (black), 7 (dark grey), or 14 (light grey) d under ice treatment, and (b) yield (g) in ice (black) and or no ice (dark grey) treatments under 0, 7, or 14 d under treatment. Error bars represent standard error. Letters indicate Fisher's least significant difference (LSD) for comparison of means at p ≤ 0.05.
Lipid peroxidation
-
Malondialdehyde (MDA) content, a measure of lipid peroxidation, increased under prolonged durations of ice encasement across all tissue types of leaf, crown, and root (Table 4, Fig. 7a). After 14 d of treatment, MDA levels were elevated across all tissue types when compared to earlier sampling days (days zero and seven). In crown tissues, MDA levels on day 14 were significantly elevated when compared to day zero. In leaf tissues, day 14 indicated the greatest damage. In the root tissues, MDA levels were significantly elevated on prolonged durations of 7 and 14 d when compared to control plants. Overall, root tissue had the lowest MDA content, followed by leaf tissues, and the crown tissues had the overall most elevated amount of MDA across all time points.
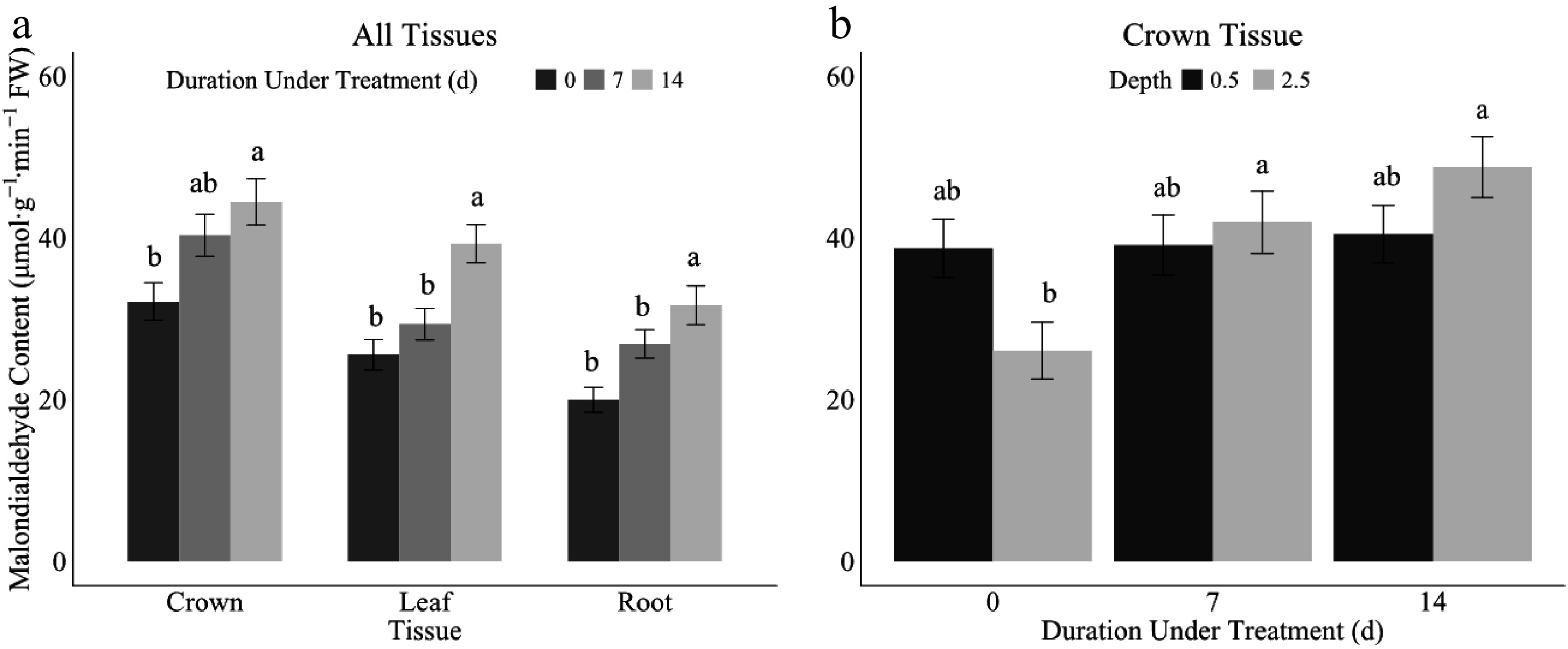
Figure 7.
Malondialdehyde content (MDA) in crown, leaf, and root tissues under 0 (dark grey), 7 (mid-grey), or 14 (dark grey) d under ice encasement (a) and crown tissue 1.27 (black) and 6.37 cm (light grey) depth under 0, 7, or 14 d under treatment. (b) Error bars represent standard error. Letters indicate Fisher's least significant difference (LSD) for comparison of means at p ≤ 0.05.
In crown tissues, the seeding depth and duration of exposure to ice encasement interaction were significant (Fig. 7b). On day zero, the 0.5-inch seeding depth exhibited higher MDA levels than the 2.5-inch depth. However, with prolonged durations of ice encasement, days seven and 14, the 6.37 cm seeding depth had elevated MDA levels when compared to the 1.27 cm depth. There were no differences in the crown tissue between duration under treatment for 1.27 cm seeding depth alone, and for 6.37 cm seeding depth, the prolonged durations of 7 and 14 d caused more stress and damage to internal plant structures when compared to only cold acclimated plants (day zero).
-
The seeding depth of winter wheat could play a significant role in plant productivity[21]. Seeding depth is a critical agronomic factor that influences germination and early growth[22,23]. Seeding depth may affect the plants' ability to endure extreme environmental stresses, such as how the crop responds to winter stress, such as ice encasement. The depth at which winter wheat is seeded in the Fall season may influence the plant's ability to survive ice encasement damage since oxygen availability is a critical factor in this stress. Stress at any stage of a plant's development, particularly the critical seedling stage, can influence physiological responses and plant vigor[24].
Prior to evaluating coleoptile length and plant health in response to seeding depth following winter, measuring if there was a photosynthetic advantage in relation to these factors during cold acclimation was critical. Fall conditions and cold acclimation can influence the resiliency of plants to winter stresses. Temperature played a role in influencing chlorophyll-related parameters during plant cold acclimation. Photosynthesis is a highly sensitive process to temperature fluctuations, since a balance between light energy absorbed by photosystems and energy consumed by metabolic sinks is important[25]. As winter wheat adapts to colder conditions, its photosynthetic systems undergo significant adjustments, and low temperature depresses photosynthesis, mainly due to the negative effects on the photosynthetic electron transport system[24,26], which gradually reduces their efficiency in preparation for winter dormancy. When temperatures drop, the ability of the chlorophyll antenna complexes to process absorbed light energy may be compromised, leading to disturbances in electron transport efficiency and potentially causing an accumulation of excess energy in the system[27]. This aligns with what was found during OJIP measurements during acclimation with parameters Fv/Fm, Fv/Fo, RE/ET, ET/ABS, and ET/TR decreasing with lowering temperatures, and parameters VI, VJ, and Do increasing with decreasing temperatures. This means that as the temperatures drop during acclimation, the electron transport becomes less efficient, while the trapping of energy becomes greater. The declines in Fv/Fm, Fv/Fo, ET/TR, ET/ABS, and RE/ET are likely due to lower temperatures, which increase membrane viscosity and restrict the diffusion of plastoquinone (PQ) to inhibit thylakoid electron transport[28].
Regarding the seeding depth influence on photosynthetic parameters during cold acclimation, the wheat plants that were seeded at a greater depth displayed enhanced values of ET/ABS and ET/TR when compared to the shallow-seeded plants. This suggests a more efficient conversion of absorbed light energy into the electron transport chain early in the cold acclimation process. The deeper-seeded plants additionally had lower VI and VJ values than the shallow-seeded plants, indicating that the deeper-seeded plants had less energy trapping. Consequently, the deeper-seeded plants were able to perform at a higher photosynthetic activity and enhanced electron transfer compared to the shallow-seeded plants. This may be attributed to the later emergence from the deeper-seeded plants, which allowed their leaves to achieve higher photosynthetic performance compared to the plants at shallower-seeded depths. Emergence plays a critical role in the thermal acclimation of photosynthetic systems in winter wheat[13], with deeper-seeded plants exhibiting a more active photosynthetic response in early stages of acclimation, possibly due to later emergence. However, as temperatures decline further and ice encasement becomes more prominent, deeper-seeded plants may face limitations in processing light energy effectively. Further research is required to determine whether emergence contributes to enhanced photosynthetic activity during acclimation, as well as its role in electron transport following ice encasement stress during the recovery phase.
Planting at a shallower depth may offer certain advantages for winter wheat, particularly in terms of leaf area, biomass accumulation, and MDA levels following ice encasement stress. Shallow-sown plants consistently exhibited larger leaf area and greater biomass in spikes and tillers compared to those wheat plants sown deeper. This difference could be attributed to the delayed emergence observed in the deeper-seeded plants, which potentially hindered their ability to establish and grow at a comparable rate. Shallow seeding depths are known to promote quicker emergence and better early growth, providing a head start for plants, but can be susceptible to soil drying[28]. Emergence from deep sowing depth commonly results in fewer, later emerging plants[29]. Deep sowing is advantageous if the soil is dry, but seedlings can have smaller relative growth rates and slower leaf area development, observed as reduced plant biomass[30]. Increasing sowing depths can enhance wheat establishment, resulting in better germination and emergence of seedlings[31]. The timing and emergence, and the subsequent growth stages at which ice encasement stress occurs, may have important implications for plant resilience.
Under prolonged durations of ice encasement, the shallow seeded plants exhibited improved cellular integrity as evidenced by reduced MDA content. Plants experiencing prolonged stress, like ice encasement, tend to accumulate higher MDA levels because of cell membrane peroxidation, induced by a drop in stability and an increase in membrane permeability[32]. This suggests that plants seeded at shallower depths may have been less stressed and more efficient in managing oxidative stress, potentially due to enhanced antioxidant defense mechanisms or improved cellular maintenance under stress. In contrast, elevated MDA content in deeper-seeded plants could be related to expenditure of energy reserves to reach the surface and oxidative damage associated with that metabolism. Oxygen availability would be lower in deeper conditions[29]. Stress after emergence would reduce abiotic stress tolerance. Additional literature is needed on seeding depth, seedling emergence, and development associated with lipid peroxidation. This information could be incorporated into crop models for winter survival[33].
The pronounced effects of seeding depth on yield under prolonged ice encasement stress further highlight the critical role of planting depth in influencing plant resilience. While no significant differences in yield were observed between seeding depths after two weeks of ice encasement compared to the control plants, those seeded shallowly still demonstrated higher yield. These findings are consistent with Loeppky et al.[9] who reported that shallower planting depths enhanced yields and winter survival, particularly under no-till practices. Furthermore, deeper seeding may introduce additional stressors, such as increased vulnerability to soil pathogens[34]. Additional research is needed to identify the ramifications of nonlethal winter stresses on yield potential.
Among the two genotypes tested, DynaGro with the elongated coleoptile consistently exhibited higher leaf area, biomass, and yield compared to the genotype with a shorter coleoptile, WhiteTail. These findings suggest that DynaGro possesses enhanced growth characteristics that contribute to improved performance under stress, potentially making it a more suitable cultivar for regions prone to ice encasement or other winter stresses. It is crucial to investigate cultivar-specific differences, particularly under prolonged exposure to lower temperatures and ice encasement stress. The elongated coleoptile of DynaGro likely facilitates quicker emergence and better seeding vigor. The results of this study are consistent with a previous one, which found that coleoptile length showed a positive overall correlation with final plant height[11]. Coleoptile length was related to emergence ability and is more desirable because rain after sowing can cause soil crusting, and the emerging coleoptile or first leaf often cannot penetrate such crusts[35]. Coleoptile length was significantly correlated with seedling emergence, but that only accounted for 28% of the variability for emergence on 10–15 d after planting (DAP)[11]. In contrast to genotypes with shorter coleoptiles, which have been linked to reduced competitiveness against weeds and leading to smaller biomass and reduced grain yield[36,37]. These traits make short coleoptile genotypes less effective at overcoming environmental stressors, especially in cold, competitive environments.
Additional research should connect the photosynthetic results and varietal results found here to carbohydrate accumulation and other important factors during cold acclimation, de-acclimation, and dormancy. Trishuk et al.[38] found major differences in wheat and canola (Brassica napus) carbohydrate responses during re-acclimation to cold temperatures once exposed to higher temperatures. Winter wheat carbohydrate accumulation was less apparent during that re-acclimation period and was associated with reduced ability to regain freezing tolerance. They also found certain proteins to play an important role in the ability to regain freezing tolerance. Whether this type of carbohydrate or amino acid metabolism could relate to recovery from ice encasement conditions warrants further investigation.
In conclusion, both seeding depth and coleoptile length potentially play important roles in determining winter wheat's capacity to withstand ice encasement stress. For farmers facing ice encasement challenges, planting at a shallower depth may offer a distinct advantage in certain regions or field areas prone to ice encasement, as these plants demonstrated larger leaf area, greater biomass, and improved cellular integrity under prolonged ice encasement. While deeper-seeded plants at 2.5 inches may initially support more efficient photosynthesis during early acclimation, they may hinder resilience after prolonged stress conditions. Additionally, selecting a winter wheat genotype with an elongated coleoptile, such as DynaGro, can further improve performance under stress, as it consistently exhibited superior growth traits. Field research and additional experiments are needed to validate these recommendations and refine guidelines for managing winterkill risks for winter wheat crops. Future research could evaluate carbohydrates or other important winter survival factors associated with planting depth and winter wheat variety.
-
The authors confirm contribution to the paper as follows: study conception and design: Merewitz E; experiment execution, data collection and analysis: Miller KM and Merewitz E; genotype physiological analysis and maintenance, draft manuscript preparation: Merewitz E and Miller KM; funding acquisition: Merewitz E. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
-
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Miller KM, Merewitz E. 2026. Seeding depth and coleoptile length effects on cold acclimation and ice encasement responses of winter wheat. Grass Research 6: e007 doi: 10.48130/grares-0026-0001
Seeding depth and coleoptile length effects on cold acclimation and ice encasement responses of winter wheat
- Received: 19 August 2025
- Revised: 03 December 2025
- Accepted: 16 December 2025
- Published online: 25 March 2026
Abstract: Winter wheat is susceptible to winterkill stresses, and management practices that target specific field problems, such as ice encasement, are needed. Therefore, a 2-year growth chamber study was conducted to assess the impact of two seeding depths (shallow: planted at 1.27 cm and deep: planted at 6.35 cm) on two winter wheat genotypes' survival following 0, 7, or 14 d of ice encasement. The two winter wheat genotypes used differed in coleoptiles length: 'DynaGro 9242W' (long coleoptile) and 'WhiteTail' (short coleoptile). This study measured chlorophyll fluorescence during acclimation, lipid peroxidation (as measured by malondialdehyde content) in leaf, root, and crown tissues, leaf area during recovery, biomass at harvest, and yield. Chlorophyll fluorescence parameters Fv/Fo, Fv/Fm, ET/ABS, ET/TR, VI, VJ, and Do were measured during acclimation and found to be significantly influenced by decreasing temperatures. Winter wheat sown deeper (6.35 cm) exhibited higher ET/ABS and ET/TR values, while the VI and VJ values were higher in plants sown at shallow depths, suggesting differences in the rate of stress-induced changes in the photosynthetic processes. Shallow seeding depth also appeared to offer advantages for plant growth. Plants sown at 1.27 cm had greater leaf area, biomass, and less lipid peroxidation in crown tissues under prolonged stress conditions. Additionally, DynaGro, with the longer coleoptile, demonstrated enhanced performance and resilience compared to WhiteTail, particularly under prolonged ice encasement stress. Results should be confirmed in field conditions to validate these findings.
-
Key words:
- Winterkill /
- Winter wheat /
- Seeding depth /
- Wheat variety /
- Ice encasement /
- Chlorophyll a fluorescence /
- OJIP transient /
- Energy flux












