-
Medicinal plants have long been recognized for their therapeutic potential due to their rich bioactive compounds, playing a crucial role in traditional medicine and increasingly gaining attention in modern healthcare. The World Health Organization reported that 80% of the population depends on herbal medicine for basic health care[1]. Furthermore, the diversity of medicinal plants globally reflects their varied applications across different cultures. However, despite their significant contributions, the cultivation and utilization of medicinal plants face a major challenge: the need to increase both yield and the content of specialized metabolites, which makes them different from staple crops. This dual challenge is essential to meet the growing global demand for herbal remedies while ensuring sustainable practices in medicinal plant cultivation and utilization.
The growth of medicinal plants and the accumulation of their bioactive compounds, such as flavonoids, flavones, terpenoids, etc., are significantly influenced by the ecological environment[2]. While cultivation of staple crops requires lowered stress levels, a certain amount of stress is usually required for the accumulation of some bioactive compounds in medicinal plants, which is one of the major determinants for their quality. Considering the contradictory effect of stresses on yield and quality in medicinal plants, it is a forever-research topic to use elicitors from abiotic or biotic factors to enhance growth and specialized metabolites[3]. Therefore, it is necessary to review existing research on the application of specific growth regulators in the accumulation of biomass and specialized metabolites to establish a basis for developing efficient and environmentally friendly technologies related to medicinal plant utilization.
The microbiome has gained more and more attention in regulating plant growth, stress responses, and the accumulation of specialized metabolites (SMs). The most commonly used bacteria in green farming are the genus Bacillus, which has a variety of beneficial functions and is commonly categorized as plant growth-promoting bacteria (PGPB). These gram-positive bacteria are known for their spore-forming ability, which allows them to survive and thrive in diverse environments[4]. They enhance nutrient uptake, promote plant growth, synthesize specialized metabolites, and induce systemic resistance against pathogens[5] (Fig. 1). Despite the numerous benefits of using Bacillus spp., existing research has primarily focused on their role in the growth of crops with less attention paid to the relationship between Bacillus and medicinal plants (Table 1). Therefore, significant gaps still exist in the understanding of the full potential and optimization of their application in medicinal plants. Furthermore, while many studies highlight the potential of Bacillus spp, there is still a lack of standardized methods for their incorporation into cultivation practices, particularly for medicinal plants that require specific growth conditions. This review aims to explore the current understanding of utilizing Bacillus species to enhance growth, stress tolerance, and the accumulation of specialized metabolites in medicinal plants. Furthermore, it highlights existing gaps and proposes a way forward to improve the comprehension of the relationship between Bacillus species and medicinal plants.
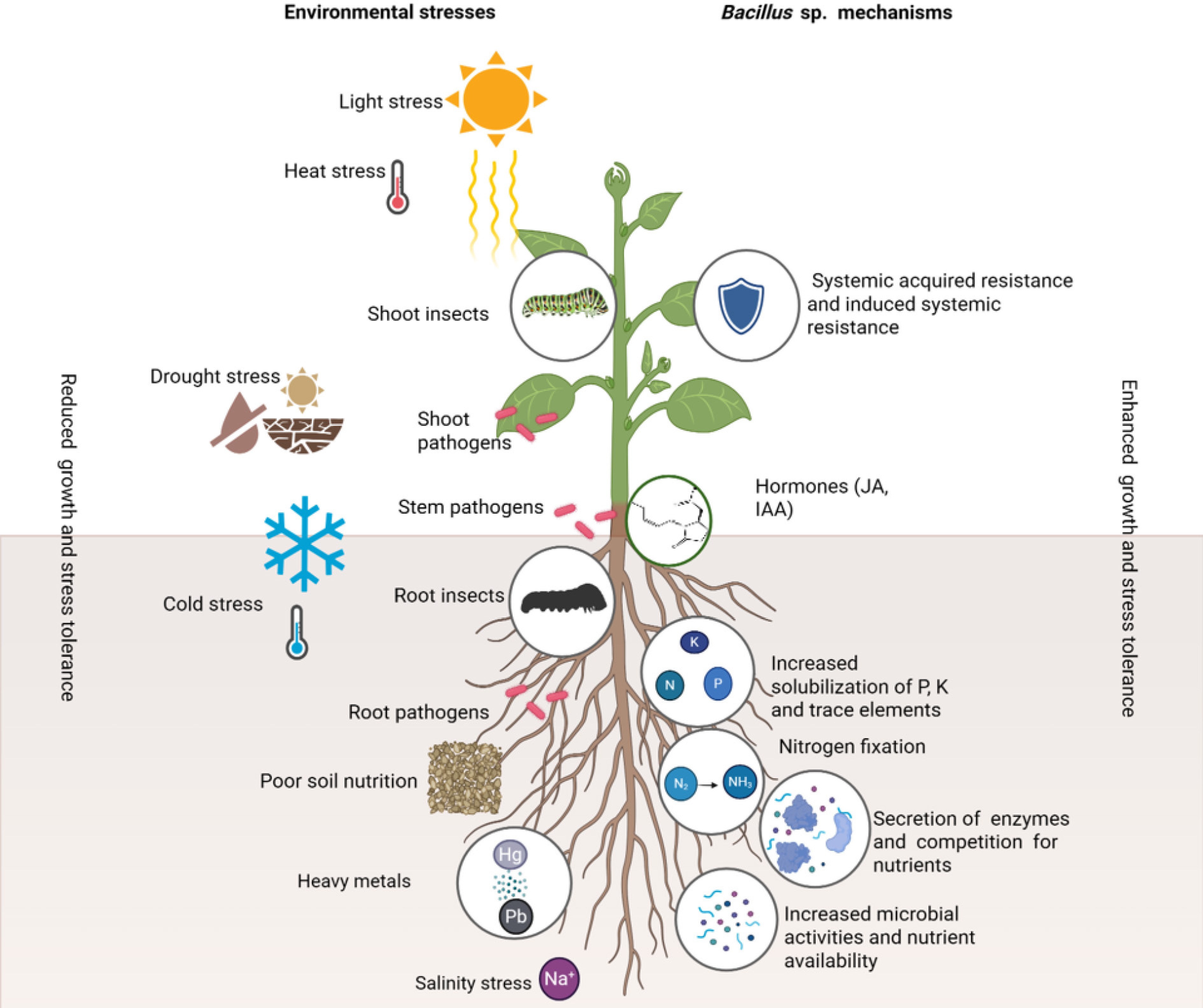
Figure 1.
The proposed model illustrating the role of Bacillus spp. in promoting medicinal plant growth, and enhancing environmental stress tolerance.
Table 1. Some of the medicinal plants associated with Bacillus spp. that result in growth promotion and environmental stress tolerance.
Medicinal plant Bacillus strain Function Ref. Zingiber officinale (Ginger) B. paralichenirfomis, B. velezensis ATR2 Plant growth promotion, biological control [6,7] Ocimum gratissimum L. B. subtilis Tc1 Plant growth promotion, biological control [8] Duranta plumeri B. amyloliquefaciens D41 Plant growth promotion, biological control [8] Lilium leucanthum B. velezensis L1e-9 Plant growth promotion, biological control [9] Camelia sinensis B. megaterium TRS-4 Plant growth promotion, biocontrol, secondary metabolites [10] Abelmoschus esculentus B. thuringiensis Plant growth promotion [11] Solanum surattense B. subtilis SSR21 Biological control [12] Abutilon indicum B. flexus AIKDL Biological control [12] Glycyrrhiza uralensis Fisch B. pumilis G5, Bacillus spp. Plant growth promotion [13,14] Curcuma longa L. B. endophyticus TSH42, B. cereus TSH77 Plant growth promotion [15] Withania somnifera B. amyloliquefaciens (MPE20) Plant growth promotion, secondary metabolism [16] Vinca rosea B. megaterium MCR-8 Plant growth promotion [17] Limonium sinense B. flexus KLBMP 4941 Plant growth promotion, stress alleviation [18,19] Aster tripolium L. Various Bacillus spp. − [20] Fagonia indica B. tequilensis MOSEL-FLS3 Biological control of plant diseases [21] Caralluma tumerculata B. flexus MOSEL-MIC5 Biological control of diseases [21] Solanum surattense B. subtilis SSL2 Biological control of diseases [22] Oxytropis merkensis Bge B. amyloliquefaciens MR4 Plant growth promotion and biological control of diseases [23] Echinacea purpurea (L.) Bacillus sp. EpSL5 Plant growth promotion [24] Origanum heracleoticum L. Various Bacillus strains Biological control [25,26] -
Bacillus spp. have long been used as PGPB in sustainable agriculture. Field and greenhouse studies show that single strains increase the yield of medicinal crops such as Artemisia annua, Cannabis sativa, Stevia rebaudiana, and Mentha arvensis through parallel processes: solubilization of bound P, biological N2 fixation, and secretion of Fe3+-chelating siderophores[27−29]. Genomes of model strains (B. cereus TS4, B. thuringiensis, B. subtilis) harbor complete gene clusters for these functions, ensuring activity even in nutrient-depleted soils[30].
Beyond nutrition, Bacillus spp. colonizes roots, and continuously releases auxin (IAA), cytokinins, gibberellins and volatile organic compounds (VOCs) that enlarge root surface area and activate systemic stress tolerance. Whole-genome scans and in-planta assays confirm the presence of indole-3-pyruvate decarboxylase (ipdC) and related IAA operons in B. megaterium BRN1, B. aryabhattai BRN3, and B. velenzensis CLA178; corresponding treatments raise essential-oil biomass and phytochemical content by 20%–45%[31−33]. VOCs emitted by B. subtilis GB03 further stimulate carbon allocation to oils without extra fertilizer[34]. Root-associated Bacillus spp. also restructure the rhizosphere microbiome, enriching phosphate-mobilising and disease-suppressive taxa[35]. Therefore, a single Bacillus strain simultaneously supplies nutrients, delivers phytohormones, and engineers a beneficial root microbiome, offering an integrated bio-inoculant strategy for low-input cultivation of high-value medicinal crops.
-
Bacillus spp., particularly those classified as plant growth-promoting rhizobacteria, have been shown to confer various benefits to plants under stressful environments, such as drought, salinity, and heavy metal toxicity. Bacillus spp. consistently enhances plant tolerance to drought, salinity, and heavy metal stresses. Under 200 mM NaCl, B. flexus KLBMP4941 increased Limonium sinense survival by 65%[19]; B. amyloliquefaciens augmented Glycyrrhiza uralensis biomass 40% under field drought and restored leaf relative water content (RWC) to 90% of well-watered controls[36]. In cadmium-spiked soils, two B. atrophaeus (WL1210 and CD303) isolates raised perennial ryegrass shoot dry weight 1.8-fold while lowering root Cd up to 34%[37]. Co-inoculation of B. subtilis with arbuscular mycorrhiza further doubled Artemisia annua artemisinin contents under water deficit conditions[38].
Protection is exerted mainly through three complementary mechanisms. First, antioxidant priming: Bacillus-colonized roots rapidly up-regulate SOD, CAT, and the AsA-GSH cycle, suppressing reactive oxygen species (ROS) bursts that typically damage membranes and photosystems[39,40]. Second, osmolyte and hormone tuning: Bacillus strains accumulate proline, sucrose, and glycine-betaine, while secreting ABA to close stomata and ACC-deaminase to cleave stress-induced ethylene, jointly maintaining water status and chlorophyll content[41,42]. Third, molecular signaling: the bacterial σB regulon and plant salt-overly-sensitive module (SOS) and C-repeat binding factor module (CBF) pathways are synchronously activated. Published transcriptome results show that B. amyloliquefaciens SQR9 up-regulates host genes, including RBCS, RBCL, HKT1, NHX1, NHX2, NHX3, etc, for osmo-protectant synthesis, an induced systemic tolerance (IST) response that persists[43].
-
Medicinal plants are susceptible to many diseases that can significantly impact their growth, yield, and medicinal properties. Among sustainable approaches to managing these diseases, the use of Bacillus spp. as biocontrol agents has emerged as a promising strategy for controlling medicinal plant diseases[44]. Bacillus spp. serves as a broad-spectrum biocontrol agents that protect medicinal crops from pathogens and insect pests while preserving yield and bioactive metabolite content. As for pathogen resistance, B. velezensis CLA178 reduced the incidence of crown gall on Rosa multiflora by 70% via induced systemic resistance (ISR)[33], and B. amyloliquefaciens HK34 curtailed Phytophthora root rot in Panax ginseng by 60%[45]. Consortia containing B. subtilis, B. thuringiensis, and B. toyonensis decreased Verticillium wilt severity in Cotinus coggygria from 58% to 12% and elevated leaf terpene content 1.4-fold[46]. As for insect control, B. thuringiensis formulations expressing Cry1/3 proteins cause > 90% mortality of lepidopteran and coleopteran larvae on Salvia miltiorrhiza without affecting pollinators[47], while lipopeptides from B. amyloliquefaciens cut aphid fecundity on Chrysanthemum by 55%[48].
The underlying mechanisms operate simultaneously and function through three mechanisms: antibiosis, ISR, and microbiome engineering for pathogen resistance. Genome mining reveals eight–12 gene clusters for lipopeptides (fengycin, surfactin), polyketides, and lytic enzymes that disrupt pathogen membranes and cell walls[44,49]. Colonization triggers plant NPR1- and MAPK-dependent signalling, up-regulating pathogenesis-related genes within 24 h, resulting in a three-fold increase in phenolics and pathogenesis-related proteins[50]. Bacillus exudates such as bacillibactin and acetoin, restructure the rhizosphere, enriching Streptomyces and Pseudomonas populations that out-compete Fusarium and Alternaria for Fe and niche space[51]. As for insect control, Bacillus spp. generates insect-specific toxins. Alkaline activation of Cry protoxins forms pores in the mid-gut epithelium of target larvae, whereas secreted lipopeptides interfere with aphid feeding and reproduction, providing complementary pest suppression with negligible non-target impact[28]. Collectively, these traits position Bacillus-based biopesticides as effective, low-resistance components of integrated disease and pest management programs for high-value medicinal plants.
-
Bacillus endophytes and rhizosphere strains act as ecologically benign elicitors that raise the concentration of pharmaceutically active terpenoids, flavonoids, and alkaloids in medicinal crops, which is essential for the quality of medicinal crops. Under normal or saline conditions, B. cereus elevates glycyrrhizic acid, liquiritigenin, and total flavonoids in G. uralensis[52], while nitrogen-fixing Bacillus J1 and Bacillus. G4 increases astragaloside, calycosin, and polysaccharide contents in Astragalus mongholicus by a similar margin[29]. Comparable increments have been recorded for ginsenosides in P. ginseng[53], artemisinin in A. annua[54], stevioside in Stevia rebaudiana[55], and withanolides in Withania somnifera[16], demonstrating broad taxonomic efficacy (Fig. 2).
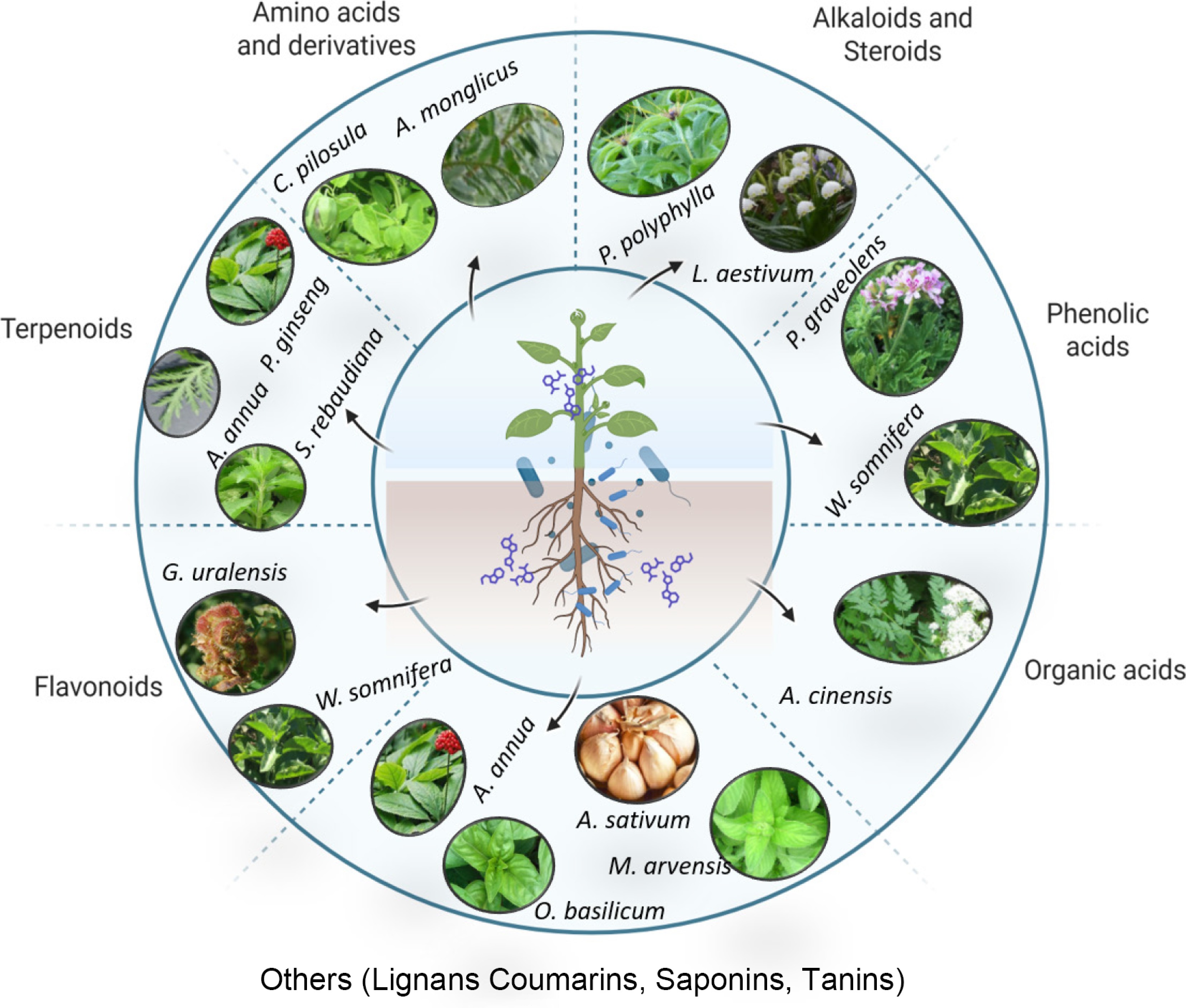
Figure 2.
Representation of some of the medicinal plants reported to interact with Bacillus spp. and lead to the accumulation of specialized metabolites.
The metabolic upshift is triggered by low-molecular signals (surfactins, fengycins, volatiles) secreted by Bacillus spp. that are perceived as microbe-associated molecular patterns. Within hours, mitogen-activated protein-kinase (MAPK) and WRKY/MYB transcriptional cascades are activated, leading to sustained over-expression of gateway genes for secondary metabolism. Examples include up-regulation of HMGR, DXS, CYP7AV1, and bZIP TFs that control sesquiterpene and artemisinin biosynthesis[54]; induction of SQS, CYP88D6, and β-AS governing glycyrrhizic acid formation[52]; and enhanced transcription of VDC for alkamide assembly in Echinacea purpurea[24] (Fig. 3).
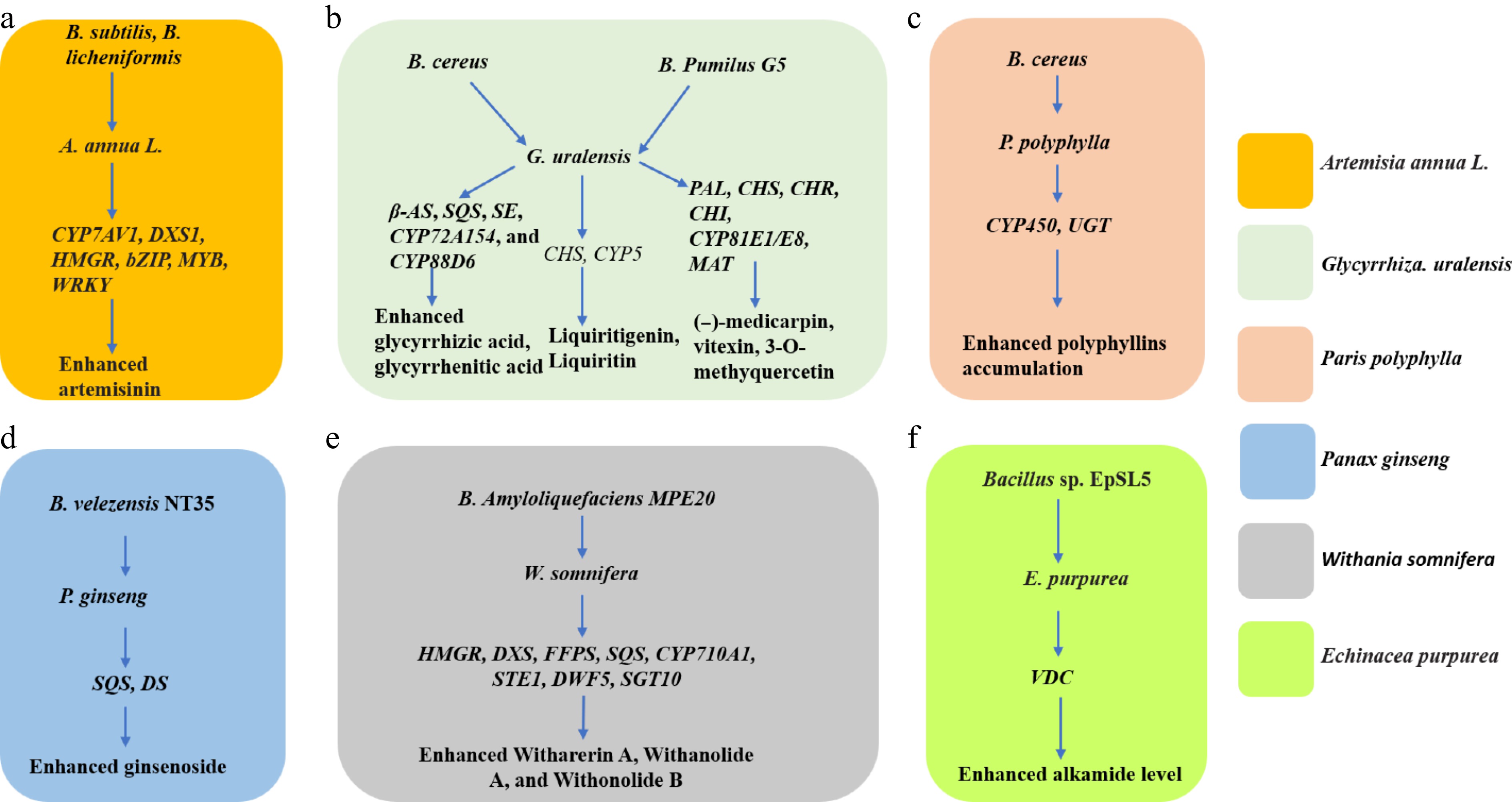
Figure 3.
Genes activated by Bacillus spp. to promote the accumulation of specialized metabolites in medicinal plants. CYP7AV1 (amorphadiene-12-hydrolase), DXS1 (1-deoxy-Dxylulose-5-phosphate synthase,1); HMGR (3-hydroxy-3-methyl-glutaryl coenzyme A reductase); bZIP (basic leucine zipper); MYB (myelobastosis), β-AS (β-amyrin synthase); SQS (squalene synthase); SE (squalene epoxidase); CYP88D6 (P450 monooxygenase ecoding β-amyrin 11-oxidase); CHS (Chalcone synthase); CYP5 (Cytochrome P450 family 5), PAL (phenylalanine ammonia-lyase); CHR (cell cycle homology region); MAT (homotetramer), DS (dammarenediol synthase); CYP450 (Cytochrome P450); UGT (UDP-Glycosyltransferase); DXS (1-deoxy-D-xylose-5-phosphate synthase); FFPS (fluorescent fusion protein), STE (sulfotransferase); DWF (Dwarf); SGT (sterol glycosyltransferase); CYP710A1 (Cytochrome P450 subfamily 710 A1); VDC (Valine decarboxylase).
-
Bacillus spp. reconciles the classical breeding dilemma between biomass and drug quality by synchronously raising yield and the concentrations of pharmaceutically active flavonoids, terpenoids, or alkaloids. Upon root colonization, surfactant and fengycin lipopeptides produced by B. subtilis and B. amyloliquefaciens act as microbe-associated molecular patterns that are perceived by plant pattern reorganization receptors (PRRs); within hours, this triggers a mild ISR response coupled to MAPK-mediated up-regulation of key biosynthetic genes (CYP72A154, β-AS, HMGR) without imposing the severe oxidative burst that normally accompanies pathogen-induced secondary metabolism[56,57]. Concomitantly, Bacillus-derived IAA and transient ABA pulses expand the photosynthetic surface and improve water-use efficiency, ensuring that the extra carbon skeletons required for secondary biosynthesis are supplied through enhanced primary productivity rather than through stress-driven re-allocation[43]. In addition, Bacillus spp. induces systemic resistance mechanisms in plants, which further fortify their defenses through the production of various metabolites, including organic compounds (VOCs), which induce systemic resistance that prepares the plant against future pathogen attacks[28,58]. These metabolites, alongside phytohormones and antimicrobial compounds, synergistically foster a conducive environment for healthy growth amid environmental stressors.
Bacillus spp. are recognized for their ability to produce a diverse array of specialized metabolites that enhance growth and productivity. These metabolites provide additional benefits by enhancing the levels of specialized metabolites and fortifying the plant's resistance to biotic and abiotic stresses[59]. This phenomenon highlights the duality of the metabolites' functional scope, as they serve both the bacteria and their plant hosts.
As part of their molecular mechanisms, Bacillus spp. such as B. subtilis, B. amyloliquefaciens, and B. licheniformis are recognized for their extensive biosynthetic gene clusters (BGCs) that encode for various specialized metabolites, including lipopeptides, polyketides, and non-ribosomal peptides, as previously documented[60]. In addition, environmental factors and plant interactions often regulate the production of specialized metabolites by Bacillus spp. The interaction with plant roots may trigger the expression of these biosynthetic pathways, leading to enhanced production of bioactive compounds. For example, the presence of plant root exudates has been reported to stimulate the expression of genes involved in the biosynthesis of specialized metabolites, leading to an increased production of beneficial compounds[61]. This interaction highlights the dynamic relationship between Bacillus and plants, where the metabolic activities of the bacteria can be influenced by the plant's physiological state and vice versa. Such interactions can enhance the plant's ability to cope with abiotic stresses such as drought and salinity by modulating specialized metabolite pathways.
-
The ecological interactions facilitated by Bacillus spp., especially in the rhizosphere, are crucial for the sustainable production of medicinal plants. For instance, dual inoculation of plant growth-promoting B. endophyticus IGPEB 33 and Funneliformis mosseae improves soil properties for Zingiber officinale (ginger) growth[62]. Moreover, many Bacillus strains secrete extracellular polymeric substances (EPS), including polysaccharides and proteins. These substances act as biological glues, binding soil particles into stable aggregates. Aggregated soil reduces compaction, improves water infiltration, and creates pore spaces that facilitate root growth and oxygen diffusion, critical for both plant and microbial survival[63]. Moreover, Bacillus plays a role in regulating soil pH, thereby restoring degraded soils[64]. In addition, the use of Bacillus in combination with organic amendments such as biochar has been particularly effective in reducing soil contaminants such as cadmium, while enhancing microbial activity and soil fertility[65].
Bacillus spp. have been implicated in the mobilization of essential nutrients in the soil, notably phosphorus, potassium, and micronutrients such as iron (Fe), zinc (Zn), and manganese (Mn), through mechanisms such as solubilization. In addition, many strains exhibit efficient nitrogen fixation. Bacillus spp. SSAU-2 can fix atmospheric nitrogen and synthesize phytohormones, such as IAA, even under harsh conditions, including high salinity and heavy metal stress, thereby directly enhancing soil fertility[66]. Furthermore, Bacillus spp. contribute to soil fertility through the decomposition of organic matter through extracellular enzymes, such as cellulases and proteases, that facilitate the breakdown of complex organic materials, thus releasing nutrients readily available for plant uptake[6]. This microbial activity is essential for the recycling of nutrients and the maintenance of soil structure, which are critical components of soil fertility[67].
The interaction between Bacillus and other soil microbial communities is also crucial for enhancing soil fertility for cultivating medicinal plants. Bacillus modulates the composition of the rhizosphere microbiome, promoting beneficial interactions among various microbiota taxa[68]. This synergistic relationship underscores the importance of microbial diversity in maintaining soil fertility and ecosystem resilience.
-
Machine learning algorithms are increasingly utilized in the analysis of large, heterogeneous datasets derived from genomic, transcriptomic, and metabolomic studies. For instance, supervised machine learning techniques have been effectively applied to identify interaction factors within microbial genomes associated with specific plant behaviors, revealing intricate patterns that drive host-microbe dynamics[69]. These approaches allow researchers to discern nuanced interactions that traditional methods may overlook, effectively correlating specific microbial traits with plant health outcomes[70]. Moreover, machine learning's capacity for pattern recognition is pivotal for addressing the complexity of the multifactorial interactions between Bacillus spp. and various medicinal plants, facilitating the identification of beneficial traits and resistance mechanisms[71].
Combining biological knowledge with computational tools fosters the development of resilient models that guide the design of synthetic ecosystems, emphasizing plant-microbe interactions through machine learning insights[72]. For instance, transfer learning frameworks have shown that taxonomic signatures of Pyrinomonadaceae, Nitrososphaeracea, and Ca. Udaeobacter reliably indicates soil health across various medicinal-crop datasets[73]. In addition, machine learning has been used to improve the prediction of disease-associated and health-associated microbial signatures with high accuracy[72]. The use of systematic CRISPRi repression in screening amino acid biosynthesis in B. subtilis enhances the production of surfactin[74]. Algorithms like random forests and support vector machines analyze trait combinations in ecological networks, enabling predictions of how plant-microbe interactions evolve under different environmental conditions[75,76]. Furthermore, meta-analyses and machine learning research indicate that models such as random forests and deep learning, trained on 16S rRNA or whole-genome data, can forecast Bacillus-mediated disease suppression, plant growth promotion, and phytochemical production[77,78]. Advances in machine learning have also enhanced the modeling and prediction of plant-microbe interactions within more dynamic, real-time frameworks[72]. For example, Muñoz & Carneiro explore how symbiotic relationships influence ecosystem behavior and dynamics, proposing that AI could model these interactions to improve ecosystem services[79]. The potential of AI to deepen understanding of interactions aligns with the rising demand for data-driven methods to optimize medicinal plant–microbe relationships. Furthermore, as ecological models grow more sophisticated through machine learning, better insights are gained into the complex interdependencies that define medicinal plant–microbe interactions.
The integration of AI and machine learning in medicinal plant research also extends to ethnobotanical studies. By examining traditional plant use and chemical profiles, researchers can uncover and validate indigenous knowledge about medicinal plants. Aziz et al. documented these traditional uses based on ethnobiological insights, which can be further validated and explored through AI analysis[80]. Bioinformatics projects, such as the TCM Systems Pharmacology Database and Analysis Platform, compile extensive data on the effects of phytochemicals and their interactions within biological systems[81]. These databases support AI-driven computational methods that enable comprehensive analysis of potential therapeutic uses of plant-derived compounds, considering microbial interactions that influence their bioavailability and efficacy. Proteomics is a key area where machine learning significantly contributes. For example, Khatabi et al.[82] reviewed how ML helps analyze large datasets to identify protein markers linked to successful plant-microbe symbiosis. Therefore, the use of current technologies can accelerate the study of Bacillus-medicinal plant interactions by providing sophisticated analytical frameworks that facilitate the understanding of complex biological systems, enhance data interpretation, and optimize microbial applications for agricultural sustainability. By integrating machine learning and other AI methodologies, researchers can unveil critical insights into plant-microbe interactions, paving the way for innovative practices in agriculture and medicine.
-
Research on Bacillus-plant interactions has been built largely on maize, rice, tomato, and other staple crops, and the resulting mechanistic framework is essentially transferable to medicinal species. Regardless of the host, Bacilli colonize the root surface, export auxins, cytotoxins, and volatile terpenes that enlarge the absorptive area and simultaneously release siderophores, phosphates, and ACC that improve mineral uptake and release ethylene-mediated stress[83]. However, the central difference is the value chain endpoint: In crops, the focus is on yield and shelf stability, thus controlling growth disruptors and improving productivity, with agricultural practices leveraging Bacillus for competition with pathogens and nutrient availability[84]. Conversely, the interaction between Bacillus and medicinal plants extends beyond growth promotion to include the enhancement of phytochemical profiles essential to the therapeutic efficacy. Recent work shows that endophytic Bacillus spp. can satisfy both criteria by coupling the generic growth-promoting circuitry described above to host-specific elicitation of pharmacologically active terpenoids, flavonoids, and alkaloids[85]. However, the number of medicinal species examined remains minuscule compared with the diversity used commercially, and co-culture protocols and harvest timing needed to maximize metabolite content without compromising biomass have been mapped for only a handful of taxa. Consequently, while the vast knowledge base generated for cereals and vegetables offers ready-made entry points, dedicated systematic studies are still required to translate these universal mechanisms into agronomic practices that simultaneously boost yield and therapeutic value in the medicinal plant production system.
Optimizing the use of Bacillus spp. under field conditions remains challenging. Many studies are conducted under controlled laboratory conditions, which may not accurately predict field performance due to differences in microbial community dynamics, inconsistent formulations, dosage, and abiotic factors[86]. Moreover, studies have not explored the comparative efficacy of various beneficial microbial strains across diverse plant species and cultivation systems, which limits the ability to recommend the most suitable Bacillus strains for enhancing the growth of specific medicinal plants[87]. Therefore, there is a pressing need for comprehensive field trials that evaluate the long-term effects of Bacillus treatments on medicinal plants for the universality and stability of their application in different ecological regions[88]. In addition, exploring the colonization laws of Bacillus in different soil types and climatic conditions, clarifying their interaction patterns with indigenous communities and host plants, and providing a basis for the development of regional microbial agents would help to fully address these challenges through continued research and innovation, which is crucial for maximizing the potential of Bacillus spp. in sustainable medicinal plant production[4]. Furthermore, standardizing bioinoculant production, delivery, and resistant management guidelines to ensure reproducible disease suppression and non-target safety would enable the use of high-efficiency strains of Bacillus with high symbiotic activity for specific medicinal plants.
The knowledge gap also remains in the molecular mechanisms that govern Bacillus-plant signaling, transcription factor networks, and epigenetic modulation, which influence plant growth and secondary metabolite production. For example, the signal transduction pathways involved in Bacillus interactions with most medicinal plants, such as hormone regulation and gene expression networks, are not yet fully understood. Therefore, there is a need to explore the complexity of these interactions at the molecular level through genetic and biochemical studies. Future priorities include developing a curated, genotype-level Bacillus library matched to specific medicinal plants and employing multi-omics approaches to map colonization patterns, hormonal crosstalk, and metabolite flow under field stress conditions. Additionally, strain genome editing technologies can be used to selectively modify Bacillus strains and medicinal plants, enhancing enzyme and metabolite production or stress-resistance gene expression, thus helping to turn promising laboratory results into scalable, real-world medicinal plant systems. Recent advancements in traditional and modern plant improvement techniques, such as CRISPR, RNAi, and next-generation sequencing (NGS), have provided valuable insights into genes involved in the biosynthesis of plant specialized metabolites. Notably, CRISPR technologies are seen as promising tools to optimize therapeutic phytochemical production by creating genetically modified medicinal plants with targeted metabolite profiles[89]. Furthermore, NGS enables the identification of genetic variations among plant species, shedding light on the metabolic pathways that increase yields of bioactive compounds[90].
The other notable challenge is the public concern over the use of beneficial microbes such as Bacillus to enhance plant production. Despite the numerous benefits of using Bacillus spp., the environmental impact of Bacillus spp. is still a critical aspect of their role in plant production and protection. For instance, studies have shown that the application of B. thuringiensis does not adversely affect non-target organisms, including beneficial insects and soil microorganisms, making it a safer alternative to conventional insecticides[91]. However, concerns regarding the potential development of resistance in target pest populations have been raised, necessitating the implementation of resistance management strategies[88]. Furthermore, although the biocontrol of plant diseases by Bacillus spp. represents a multifaceted approach to improve medicinal plant cultivation, the complexities involved in ensuring consistency and efficacy in disease suppression in various plant systems represent a barrier, making it unacceptable to some growers worldwide[92]. Moreover, the high variability in commercially available products often leads to confusion in the application and effectiveness of Bacillus strains. For instance, Bueno et al. have shown that the concentration and formulation of Bacillus inoculants significantly impact their colonization and consequent efficacy in plants[93]. However, inconsistent product application protocols can lead to underwhelming results in practical applications, signaling a need for standardized practices and more reliable product formulations, as mentioned previously in this section.
Artificial intelligence offers significant opportunities to advance the study of Bacillus-medicinal plant interactions. It enables the collection and analysis of large datasets on plant-microbe relationships through crowd-sourcing and remote sensing, complementing lab results and revealing ecosystem-level factors affecting plant health and pathogen resistance. Remote sensing coupled with AI allows real-time monitoring of medicinal plant health and growth, helping maintain optimal conditions for Bacillus species[94]. Looking ahead, three key AI-driven innovations will be essential: (1) Environment-specific strain design: reinforcement learning agents, using soil multi-omics and climate data, will suggest targeted gene edits or promoter swaps to customize Bacillus genomes. (2) Optimized microbial communities: graph neural networks will analyze interaction scores, metabolite complementarity, and niche overlap to create three- to five-member Bacillus consortia with > 90% rhizosphere persistence and enhanced secondary metabolite production. (3) Soil-ecosystem prediction: digital twin models integrating metagenomic data and geospatial factors will predict the long-term effects of introduced Bacillus strains on microbiota diversity, antibiotic resistance gene flow, and soil health, providing regulators with quantitative risk assessments before field deployment. This forecasting enables better optimization of Bacillus and medicinal plant interactions to maximize therapeutic benefits. AI models that incorporate climate, plant, and microbial genomics data can forecast which bacterial and plant combinations yield the greatest medicinal advantages, guiding cultivation strategies[95].
-
Bacillus spp. plays a versatile role in supporting medicinal plant growth through its various biochemical and ecological activities. They can improve plant nutrition, promote growth and metabolite synthesis, and defend against pathogens, offering a sustainable approach to medicinal plant cultivation that balances yield, quality, and environmental responsibility. This highlights their importance in modern agriculture. To realize this potential, a shift is needed from traditional inoculation methods to precise, system-level strategies supported by strong ecological and regulatory measures. Ongoing research on the molecular interactions between Bacillus and medicinal plants will reveal how these microbes can be better utilized to boost plant resilience and increase bioactive compound production. As research advances, identifying more Bacillus strains and understanding their genomes will aid in developing effective bioinoculants for sustainable medicinal plant farming. Importantly, integrating artificial intelligence offers the predictive power and speed necessary to match specific Bacillus strains or synthetic communities to particular crops, climates, and soils, while also evaluating their long-term impact on soil health. This review provides a broad overview and a basis for future research into how Bacillus strains can improve plant growth and specialized metabolite production. Future studies are expected to deepen the understanding of these beneficial interactions, fostering innovative agricultural practices focused on plant health and medicinal product yield.
This work was supported by the Biological Resources Programme, Chinese Academy of Sciences (Grant No. KFJ-BRP-007-017), the Guangdong Provincial Key Laboratory of Applied Botany (Grant No. 2023B1212060046), and the Shanghai Key Laboratory of Plant Functional Genomics and Resources, Shanghai Chenshan Botanical Garden (Grant No. PFGR202502).
-
The authors confirm their contributions to the paper as follows: study conception and design: Medison RG, Xing Y, Wang Y, Li Y; methodology, visualization: Medison RG, Xing Y; investigation, funding acquisition: Li Y; resources: Yang X, Wang Y, Li Y; draft manuscript preparation: Medison RG, Xing Y, Medison MB, Liu H, Chai M, Li Y; writing – review & editing: Medison RG, Xing Y, Li Y, Khitov B, Liu H, Chai M, Yang X, Li Y; supervision: Li Y, Yang X, Wang Y, Li Y; project administration: Wang Y, Li Y. All authors reviewed the results and approved the final version of the manuscript.
-
Data sharing is not applicable to this article, as no datasets were generated or analyzed in the current study.
-
The authors declare that they have no conflict of interest.
-
# Authors contributed equally: Rudoviko Galileya Medison, Yusi Xing
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Medison RG, Xing Y, Medison MB, Li Y, Khitov B, et al. 2026. Harnessing Bacillus spp. for sustainable production of medicinal plants: from growth promotion and stress protection to specialized-metabolite enhancement. Medicinal Plant Biology 5: e007 doi: 10.48130/mpb-0026-0001
Harnessing Bacillus spp. for sustainable production of medicinal plants: from growth promotion and stress protection to specialized-metabolite enhancement
- Received: 26 September 2025
- Revised: 10 December 2025
- Accepted: 26 December 2025
- Published online: 19 March 2026
Abstract: Plant growth-promoting bacteria (PGPB) play a crucial role in enhancing plant health and productivity. Among PGPBs, the genus Bacillus has become a research hotspot due to its diverse beneficial functions. Existing research has predominantly focused on their role in crop growth and stress tolerance, with limited attention given to their relationship with medicinal plants. Unlike general crops, accumulation of specialized metabolites (SMs) is essential for medicinal plant cultivation, and the quality of medicinal plant products is a combined output of both internal and external factors. This review aims to provide a comprehensive overview of recent advancements in understanding the role of Bacillus spp. in promoting the growth of medicinal plants, enhancing tolerance to environmental stresses, promoting the accumulation of bioactive compounds, and improving soil fertility. By synthesizing the existing knowledge, this review provides a further background for exploring the potential of Bacillus in optimizing growth and specialized metabolism in medicinal plants. It also suggests future research directions to encourage in-depth investigations into the interaction between Bacillus spp. and medicinal plants in the artificial intelligence (AI) era, ultimately leading to innovative strategies that prioritize plant health and the yield of medicinal products. Expanding understanding of beneficial interactions will further pave the way for sustainable and efficient medicinal plant production and agricultural practices.
-
Key words:
- Bacillus /
- Medicinal plant /
- Plant growth-promoting bacteria /
- Soil health /
- Specialized metabolites












