-
Nephotettix nigropictus (Stål) (Hemiptera: Cicadellidae)[1], commonly known as rice green leafhopper, is found in lowland rainfed and irrigated wetlands more than in upland rice areas of Asia, Africa, Oceania, Australia, Fiji, Guam, Palau, and Papua New Guinea[2]. This causes 'hopperburn', a yellowing or browning and death of rice plants, wild grasses, and sedges, mostly done by nymphs and adults sucking directly on the sap of plants and, in large numbers, causes plant wilting and death[2,3]. Also, N. nigropictus can transmit pathogens such as rice yellow dwarf mycoplasma-like organisms, rice dwarf viruses, rice transitory yellowing viruses, and rice tungro viruses[4]. N. nigropictus is among the six species of leafhoppers (five species under Nephotettix and the Recilia dorsalis [Motschulsky] [Hemiptera: Cicadellidae]) that are the main potent carriers of the rice tungro viruses[5]. Rice tungro disease (RTD) caused by infection of two distinct virus species, namely Rice tungro spherical virus (RTSV) and Rice tungro bacilliform virus (RTBV), is often linked to stunted growth, yellow-green to orange discoloration of the leaves, and reduced tillering (with small and/or sterile panicles) of rice[6]. RTSV has a single-stranded RNA and belongs to the genus Waikavirus of the family Secoviridae, while RTBV has a double-stranded DNA and belongs to the genus Tungrovirus of the family Caulimoviridae. RTBV mainly drives the severity of RTD symptoms, while RTSV plays a role as a helper virus in transmitting RTBV[7]. If rice plants are infected only with RTBV, they develop mild stunted growth with yellowing and reddening of leaves. While with RTSV, they only develop mild stunted growth and no occurrence of leaf discoloration[8]. The two viruses have synergistic interactions involving partial dependence, i.e., RTBV depends on RTSV, which encodes an insect transmission factor for transmission. Both viruses are transmitted by nymphs and adults of the rice green leafhopper through phloem-feeding on rice plants. Furthermore, N. nigropictus can also feed on weeds and sedges, which serve as an additional source of viral inoculum[6,9].
Rice is a staple food for over half of the global population as it is an excellent source of carbohydrates[10]. Rice production, however, is confronted with many challenges, including the diversity of pests (viruses, pathogens, insects, weeds), harsh environmental conditions as effects of climate change, and post-harvest management[11]. The significant loss in crop yield and food production due to insect infestation is a serious problem for the agriculture sector globally and a threat to food security. Various control measures have been employed to control the population of N. nigropictus and minimize the spread of pathogens that affect rice. These include (1) transplanting seedlings to avoid early infection; (2) good weed management; (3) crop rotation; and (4) the use of biological control[3,12]. Pests that inflict damage between 5% and 10% are categorized as minor pests, while those that cause more than 10% harm are considered major pests[13]. Various chemicals, such as pesticides including phoxim, pyrethroid, emamectin benzoate, fipronil, farnesyl acetate, neonicotinoids, cyantraniliprole, and novaluron, are used to safeguard crops from damaging pests. Some juvenile hormone analogs used as pesticides are methoprene, fenoxycarb, and pyriproxyfen. However, these pesticides harm the ecosystem, which also leads to the development of insecticide-resistant pests[14].
Biological controls, including entomopathogenic fungi (EPF), offer an environmentally friendly, precise, and safe-to-use approach to controlling insect pests. EPF infects and kills insects indirectly via mechanisms involving secretions of enzymes and secondary metabolites, as well as direct contact with insect exoskeleton through spores or hyphae, allowing penetration of the cuticle of the host insect and fungal proliferation within the host, causing insecticidal effects without requiring ingestion[15]. Additionally, EPF adapts to host antimicrobial responses by modifying components of its cell wall, toxin production, and metabolite secretion to evade insect immune responses[16,17]. Once the insect cuticle is penetrated, the inner layers are colonized, which can lead to the death of the insect[18,19].
EPF can infect all stages of insect development in over 18 orders of insects. However, the virulence of EPF varies significantly among species[16]. Generally, the stages of fungal infection to susceptible host include: (1) precontact of fungal spore or mycelium to insect host exoskeleton; (2) cuticle contact of EPF and subsequently attach, germinate, and penetrate the host exoskeleton through secretions of various metabolites, enzymes or formation of appressoria; (3) internal growth of fungi within the host hemocoel and other tissues in the form of hyphal filaments, blastospore cells or protoplast and infection beyond hemocoel leads to death of insect; and, (4) outward growth penetration (from inside of insect growing externally) followed by sporulation of fungus externally on the surface of the insect cadaver[19]. Yamini et al.[20] demonstrated the successful penetration of Metarhizium anisopliae (Metschn.) Sorokin (Hypocreales: Clavicipitaceae) against the tomato fruit borer (Helicoverpa armigera [Hübner] [Lepidoptera: Noctuidae]). Mycelial adherence and penetration of Metarhizium spp. were also observed by Monisha et al.[21] in the insect pest–ash weevil (Myllocerus subfasciatus [Guérin-Méneville] [Coleoptera: Curculionidae]). Moreover, the virulence of Metarhizium rileyi ([Farl.] Kepler, Rehner, and Humber) (Hypocreales: Clavicipitaceae) against pupae of Spodoptera litura (Fabricius) (Lepidoptera: Noctuidae) and Spodoptera frugiperda (J. E. Smith) (Lepidoptera: Noctuidae) under soil conditions was demonstrated by Vivekanandhan et al.[22]. Interestingly, Vivekanandhan & Waheeb[15] showed the capability of Beauveria bassiana (Bals.-Criv.) Vuill. (Hypocreales: Cordycipitaceae) to infect different life stages of the mealworm beetle (Tenebrio molitor [Linnaeus] [Coleoptera: Tenebrionidae]), a major threat to global grain and flour production.
EPF is the first type of microorganism employed as a biocontrol agent against pests. Beauveria bassiana was used as a biocontrol agent in 1956 to combat the sweet potato leaf weevil Cylas formicarius (Fabricius) (Coleoptera: Brentidae)[19]. EPFs are categorized into six major phyla: Ascomycota, Basidiomycota, Chytridiomycota, Entomophtoromycota, Microsporidia, and Oomycetes. From less speciose orders like Pleosporales, Myriangiales, and Ascosphaerales to incredibly varied subgroups within the comparatively well-known order Hypocreales[14]. The most well-known EPFs that are capable of killing a wide variety of insects from the order Hypocreales include Aschersonia, Beauveria, Cordyceps, Culicinomyces, Hirsutella, Hypocrella, Lecanicillium, Metarhizium, Paecilomyces, Samuelsia, Tolypocladium, Torrubiella, and Trichoderma[14].
Bats (Order: Chiroptera) are a diverse group of mammals with more than 1,400 different species, with the ability to inhabit various ecological niches and environments, such as caves[23]. A substantial number of bat populations roost in caves; therefore, they produce excreta deposits called bat guano. Bat guano deposits in caves are a rich source of major and trace elements. The high nutrient levels of bat guano promote the growth of diverse groups of organisms, from small arthropods and insects to microorganisms such as bacteria and fungi, creating a small community. Bat guano deposits may also harbor various microorganisms with pathogenic or zoonotic potential to humans. Interestingly, they may also harbor diverse fungal species with a wide array of biological activities, including potential sources of entomopathogens or EPF[24].
Since bat guano deposits on cave floors come from bats with varying dietary preferences, such as insectivorous, nectarivorous, or frugivorous, various coprophilous fungi (strict sense) or fimiculous fungi (broader sense), both of which grow on dung, from these guano deposits may harbor potential entomopathogenic activities. Many of these dung-inhabiting fungi have been studied and explored for sources of secondary metabolites or biotechnological processes for use in agriculture, like biocontrol agents such as Chaetomium and Trichoderma, as reviewed by Sarrocco[25] and Calaça et al.[26] However, limited studies have been made in dung-inhabiting fungi for entomopathogenic potential. This study, then, aimed to determine the entomopathogenic activities of several fungal isolates from bat guano deposits in Palale Cave, Tayabas, Quezon, Philippines, against N. nigropictus third instar larvae. Moreover, the percent mortality of N. nigropictus was assessed, and microscopic observation of dead larvae, as well as Koch's postulate approach, was performed. Isolation and purification of the fungal isolates from bat guano, as well as morpho-molecular characterization and phylogenetic identification of the fungal isolates, were also conducted. Furthermore, SEM and histopathological analyses of the mycelial-infected samples were employed to determine the extent of infection.
-
The fungi in this study were isolated from the bat guano collected by the researchers and curators of the University of the Philippines Los Baños Museum of Natural History (UPLB MNH) in Palale Cave, Tayabas, Quezon, Philippines. Isolation procedures in the present study were based on the serial dilution plating method of Aziz & Zainol[27] with modifications. In this study, the guano solution (50 g guano dissolved in 450 mL 1% peptone water) was serially diluted (0.85% saline solution as diluent) up to three times (10−1 to 10−3), then a volume of 0.1 mL from each dilution (10−1, 10−2, and 10−3) was aseptically spread-plated in potato dextrose agar (PDA) (Conda, Spain) plates in triplicates. The PDA medium was autoclaved (CL-40L:810544, ALP Co., Ltd) at 121 °C, 15 psi for 15 min. After sterilization, it was cooled down to 40 °C and aseptically supplemented with streptomycin antibiotic (100 ppm final concentration in PDA medium) from a filter-sterilized 50,000 ppm streptomycin stock solution. The culture plates sealed with cling wraps were incubated at room temperature (ca. 28 °C) for 3–7 d to allow fungal growth. Each distinct fungal morphotype was isolated and subcultured in PDA plates through a single-point inoculation technique, i.e., an advancing hyphal tip was inoculated at the center of a PDA plate.
In assessing the purity of the isolates, the methods of Lopez[28] were adopted. A three-point inoculation technique was employed, in this manner, by seeding the fungal isolates at three points (triangular) in Petri plates with solidified PDA medium. After incubation at ambient room temperature (ca. 28 °C) for several days, the similar cultural characteristics of the three growing mycelial colonies indicate the purity of the culture. Once pure culture was obtained, PDA slants and water stocks of each isolate were prepared and stored in the refrigerator (ca. 4 °C), serving as stock cultures of the fungal isolates. The pure cultures of fungal isolates were deposited in the Microbial Culture Collection (MCC) of the UPLB MNH.
Morpho-molecular characterization and phylogenetic identification
-
The selected fungal isolates were initially characterized and identified through the classical technique based on the method of Lopez[28], in which the cultural and morphological characteristics were observed. In assessing the cultural characteristics, isolates were grown as one-point and three-point colonies in PDA. After 7 d of incubation, the cultural features of the obverse/reverse side of the colonies were described and recorded. On the other hand, in assessing the morphological features, the slide culture method was employed to observe their conidiophores/sporangiophores using an electric binocular microscope (Motic, BA310E, China). With the help of the Illustrated Genera of Imperfect Fungi, 4th Edition by Barnett & Hunter[29], the isolates were identified at the genus level. To confirm this, molecular techniques were carried out using the internal transcribed spacer (ITS) marker. DNA extraction was performed using the Fungus DNA Extraction Kit (Vivantis GF-1, Malaysia). Extracted genomic DNAs were run in a 1% agarose gel (Vivantis, Malaysia) at 100 volts for 30 min using an electrophoresis (Mupid-One 080872 Submarine Electrophoresis System, Japan) and viewed using a gel documentation system (Azure Biosystems C280 Gel Documentation System, USA) for DNA quality check. The genomic DNAs were sent to MacroGen, Inc. in South Korea for purification, cleaning, amplification, and sequencing of the ITS region (ITS1-5.8S-ITS2) using forward primer ITS1 (5′TCCGTAGGTGAACCTGCGG3′) and the reverse primer ITS4 (5′TCCTCCGCTTATTGATATGC3′). The sequence results were matched in the National Center for Biotechnology Information (NCBI) database using BLASTn for preliminary identification of the isolates. The ITS sequences of the isolates in this study were submitted to the NCBI GenBank database.
In phylogenetic analysis, the nucleotide sequences for ITS1–5.8S–ITS2 of reference strains corresponding to the fungal isolates in the present study were downloaded from the NCBI GenBank database. Respective related sequences were aligned using MUSCLE in MEGA-X. All phylogenetic trees were created using IQ-TREE multicore version 2.4.0 (command line) with 1,000 ultrafast bootstrap replicates (-bb 1,000), Shimodaira-Hasegawa approximate likelihood ratio test (-alrt 1,000), approximate Bayes test (-abayes), and ModelFinder (-m TEST) functions. The generated phylogenetic trees thus include three support values (SH-aLRT, aBayes, and UFBoot) on each branch. FigTree version 1.4.4 was used for visualizing the phylogenetic trees.
Entomopathogenicity assay
The source of the third instar larvae of N. nigropictus
-
In this study, the 700 third instar larvae of N. nigropictus were provided by the Institute of Weed Science, Entomology, and Plant Pathology (IWEP) of the College of Agriculture and Food Science (CAFS) at the University of the Philippines, Los Baños.
Preparation of the experimental setup for the entomopathogenicity assay
-
The experimental setup (Supplementary Fig. S1) was carried out using a glass bottle containing 30 mL compounded Hoagland's plant culture agar medium (components: 1% agar, 6 mL/L 1 M KNO3, 4 mL/L 1 M Ca(NO3)2·4H2O, 2 mL/L 1 M MgSO4·7H2O, 1 mL/L (NH4)H2PO4, 1 mL/L of 5,000 ppm Fe-EDTA, and 1 mL/L from 1 L micronutrient solution containing 2.86 g H3BO3, 1.81 g MnCl2, 0.22 g ZnSO4, 0.02 g MoO3·H2O, and 0.08 g CuSO4). A total of 15 experimental setups were prepared since there were five treatments corresponding to the four fungal isolates and a negative control group, where each treatment had three replicates. The glass bottles containing the medium were sealed with filter paper and rubber bands and sterilized at 121 °C, 15 psi for 15 min.
Meanwhile, around 500 seeds of Taichung Native-1 (TN1) rice variety (a pathogen-susceptible reference rice variety) provided by IWEP were surface-sterilized by immersing them in a 3% sodium hypochlorite solution in a sterile well-sealed bottle container for 5 min, followed by two washes in distilled water. The surface-sterilized seeds were germinated in Petri plates containing sterile solidified 1% water agar to further screen out possible bacterial/fungal-contaminated seeds. After several days of germination, 20 healthy rice seedlings of approximately the same size were transplanted in each experimental setup (sterilized bottle container with Hoagland's plant culture agar medium) and allowed to grow in a laboratory area with proper lighting and ventilation. Physical parameters such as temperature and humidity were monitored daily for the entire course of the experiment. When the two-leaf stage of the rice seedling was observed, 20 third instar larvae of N. nigropictus were aseptically introduced into each setup and were subjected to 2-d acclimatization before exposure to the entomopathogenicity test.
Assessment of the percent mortality of N. nigropictus
-
The assessment of the percent mortality of N. nigropictus third instar larvae treated with the four fungal isolates was carried out following the methods of Nilamudeen & Sudharma[30] with modifications. A single-dose screening assay composed of two parts, namely, (1) preparation of conidial suspension and (2) entomopathogenicity test to third instar larvae of N. nigropictus, was employed. In the preparation of spore suspension, fungal isolates were cultured in PDA plates incubated at room temperature (ca. 28 °C) for 7 d. Spores of each fungal isolate were harvested by pouring sterile 2 mL distilled water into a culture plate, and the 2 mL spore suspension was aseptically transferred into a sterile tube using a sterile 5 mL needle. Using a Neubauer hemocytometer and an electric binocular microscope (Motic, BA310E, China), the initial concentration of the spores was determined and adjusted to ~106 spores/mL. For the entomopathogenicity test, a 3 mL spore suspension (ca. 106 spores/mL) was applied topically to the 2-d acclimatized third instar larvae inside each bottle setup using a pre-sterilized atomizer. The experiment was laid out in a completely randomized design. The setups were watered every other day with 5 mL of sterile distilled water using a sterile needle injected through the filter paper seal. Five days after exposure to spore suspension, the number of dead larvae was counted to determine the percent mortality using the formula:
$ {\rm{ PM}} = {\rm{N/TP}} \times 100 $ PM is the percent mortality, N is the number of dead larvae, and TP is the total larvae population in each setup, i.e., 20 instar larvae. In the present study, the dead larval samples were collected aseptically and subjected to microscopic observation using a dissecting microscope. The Koch postulate analysis approach was also employed for larval samples observed to be infected with fungal mycelia.
SEM of mycelial-infected samples
-
Mycelial-infected samples were subjected to SEM. Dried samples were sent to the Integrated Electron Microscopy Laboratory, Central Instrumentation Facility, De La Salle University – Laguna Campus, Sta. Rosa, Laguna, Philippines, for SEM analysis. The dehydrated insect samples were attached to the sample stub via a double-sided carbon tape and carbon paste and then sputter-coated with Pt using a sputter coater equipment (JEC-3000FC, USA) (current of 30 mA and coating time of 50 s) before being loaded and imaged using a SEM (JEOL JSM IT500HR/LA Schottky Field Emission SEM, USA).
Histopathological analysis of mycelial-infected samples
-
The histopathological analysis of mycelial-infected samples was conducted at the Histopathological Laboratory of the College of Veterinary Medicine, University of the Philippines Los Baños. It was based on the methods of Toledo et al.[31] with modifications. The samples were fixed in 10% formaldehyde phosphate buffer (pH 6.8) and then dehydrated by immersing the samples for a few minutes in a series of 30%, 50%, 75%, and 95% ethanol, followed by 100% xylene. Samples were air-dried for a few minutes. In paraffin sectioning, the fixed and dehydrated samples were embedded in melted paraffin wax inside a molder. The samples were then cut (coronal and sagittal sections of the head, thorax, and abdomen) at 5–6 µm thickness using a microtome (Thermo Scientific, Microm HM 325, USA). Cut sections were placed in a tissue floatation processor and subsequently placed on clean slides lined with adhesives. Then, the slides with cut sections were deparaffinized using two series of 100% xylene for 5 min each, followed by air drying for a few minutes, and then stained using modified Grocott methenamine-silver nitrate staining with modification, i.e., amoxicillin was used to prevent artifacts associated with bacterial cell walls. The cut sections in the slide were added with drops of 2% sulfuric acid and allowed to stand for 15 s before washing with distilled water. Then, drops of 1% sodium sulfite were added and allowed to stand for 1 min. Subsequently, drops of amoxicillin-silver nitrate were added to the cut sections and allowed to stand for 1 min before gently heating the slide over a flame using an alcohol lamp for 5 s and washing with distilled water. Immediately, drops of 5% sodium thiosulfate were added and allowed to stand for 2 min before gently washing with distilled water. Then, drops of 0.2% fast green stain were added to the slides and allowed to stand for 1 min before gently washing with distilled water. The slides were then air-dried for a few minutes before mounting with Canada balsam and covered with a cover slip. The slides were observed using an electric binocular microscope (Motic, BA310E, China). Fungal tissues appear black or metallic silver over a green background (insect tissues). However plausible, this method needs comparison with other staining methods, such as Periodic Acid-Schiff (PAS) and Calcofluor white staining, for consideration by future researchers.
Statistical analyses of data
-
The Minitab ver. 21 statistical software was used for statistical analyses of data. The experiment was laid out in a completely randomized design (CRD). The Shapiro-Wilk test was used to determine the normality of the data. One-way analysis of variance (ANOVA) was used to analyze the data on mean percent mortality, and treatments declared significant by ANOVA were further compared using Tukey's comparison of means.
-
Generally, 38 different fungal morphotypes were isolated and purified from the bat guano deposits of Palale Cave, including the four fungal isolates in the present study. These four fungal isolates were selected for the entomopathogenicity assay based on their protease activity (Supplementary Fig. S2)[32], which was among the enzymatic assays conducted as the preliminary assessment for screening the potential biofunctional activities of the different morphotypes. With their biofunctional potentials, the four fungal isolates in this study were the earliest among the different morphotypes to be characterized and preliminarily identified. Nevertheless, the morpho-molecular characterization and phylogenetic analysis of the mycoflora of Palale Cave is ongoing and will be published shortly.
In this study, the selected fungi isolated from bat guano were characterized at the genus level in terms of cultural and morphological features with reference to the illustrated guide by Barnett & Hunter[29]. For confirmation, a molecular technique using the ITS1-5.8S-ITS2 sequence was conducted for preliminary identification and further confirmed through phylogenetic analysis. Table 1 shows the observed cultural and morphological features of the selected fungal isolates and the summary of the BLASTn analysis of the selected fungal isolates using the ITS marker. On the other hand, Fig. 1 shows the single-point, three-point, and asexual fruiting structures of the selected fungal isolates. Moreover, Figs. 2−5 show the phylogenetic tree for each fungal isolate.
Table 1. Preliminary identification of fungal isolates based on classical characterization and ITS nucleotide BLAST searches in GenBank.
Culture collection code GenBank Accession no. Cultural/morphological
featuresSequence length (bp) GenBank Accession no. of the closest match Closest match Identity (%) MCC-UPLB MNH-70156 PX139108 Cottony aerial, scattered powdery green (obverse), creamy white (reverse), conidiophores present 502 NR_130668.1 Trichoderma asperellum (Samuels, Lieckf. & Nirenberg) 98.07 MCC-UPLB MNH-70157 PX139107 Powdery, yellow-green (obverse), yellow (reverse), conidiophores present 540 NR_135325.1 Aspergillus tamarii (Kita) 99.43 MCC-UPLB MNH-70158 PX139105 Cottony, pinkish-white (obverse), cream-orange (reverse), conidiophores present 540 NR_121529.1 Talaromyces purpureogenus ([Stoll] Samson, N. Yilmaz, Frisvad & Seifert) 98.49 MCC-UPLB MNH-70160 PX139106 Compact velvety, white (obverse-reverse), sporangiophores present 625 MW412762.1 Actinomortierella ambigua ([B.S. Mehrotra] Vandepol & Bonito) 99.00 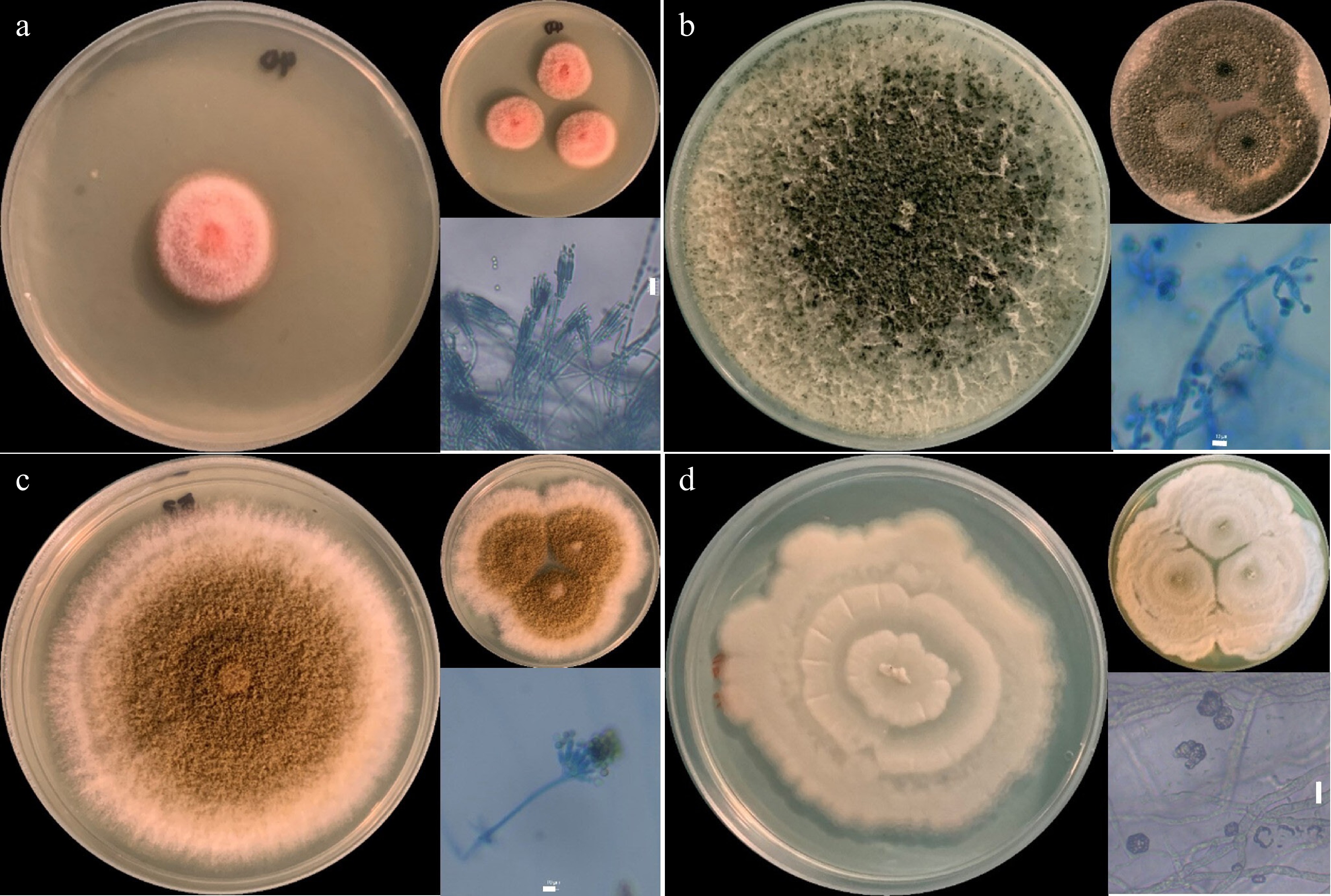
Figure 1.
Fungal isolates. (a) Talaromyces sp., (b) Trichoderma sp., (c) Aspergillus sp., and (d) Actinomortierella sp., showing one-point and three-point cultures in PDA after 7 d of incubation. Microscopic asexual fruiting structures are also shown. Scale bar = 10 µm.
The phylogenetic position of Trichoderma sp. MCC-UPLB MNH-70156, as shown in Fig. 2, is closely related to the Trichoderma asperellum (Samuels, Lieckf. & Nirenberg) (Hypocreales: Hypocreaceae) with a strong branch support of 90.7% SH-aLRT, 0.99 posterior probability, and 99% bootstrap values. The phylogenetic relationship (Fig. 3) of Aspergillus sp. MCC-UPLB MNH-70157, with the reference strains within the Aspergillus Section Flavi, shows that it is closely associated with Aspergillus tamarii (Kita) (Eurotiales: Aspergillaceae), having a branch support of 0.99 posterior probability and 96% bootstrap values. Meanwhile, Talaromyces sp. MCC-UPLB MNH-70158 is found within the clade of reference strains and the type strain of Talaromyces purpureogenus ([Stoll] Samson, N. Yilmaz, Frisvad & Seifert) (Eurotiales: Trichocomaceae) within the Section Talaromyces, with a strong branch support of 91.8% SH-aLRT and 0.98 posterior probability values, as shown in Fig. 4. Actinomortierella sp. MCC-UPLB MNH-70160 belongs to the Actinomortierella clade with highly strong branch support of 100% SH-aLRT, 1 posterior probability, and 100% bootstrap values, as shown in Fig. 5. Specifically, Actinomortierella sp. MCC-UPLB MNH-70160 is found within the group of Actinomortierella ambigua ([B.S. Mehrotra] Vandepol & Bonito) (Mortierellales: Mortierellaceae) reference strains and type strain with a strong branch support of 97.6% SH-aLRT, 1 posterior probability, 99% bootstrap values.
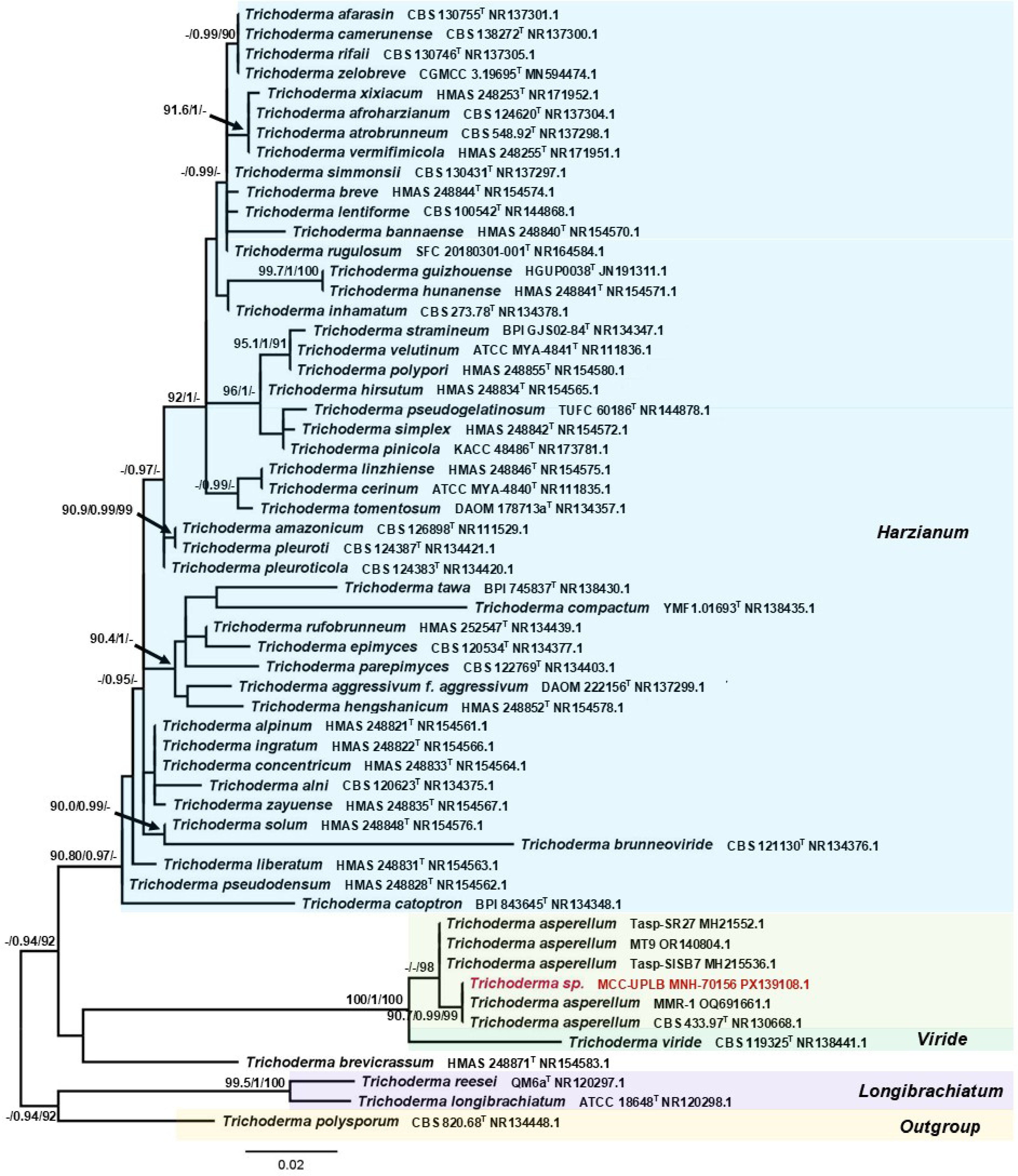
Figure 2.
Phylogenetic tree inferred using the ITS1-5.8S-ITS2 sequence showing the relationships of Trichoderma sp. MCC-UPLB MNH-70156 with representative reference strains (type strains are denoted byT) under the genus Trichoderma. Support in nodes is provided by SH-aLRT, aBayes, and UFBoot, where values ≥ 90%, ≥ 0.90, and ≥ 90%, respectively, are shown (a dashed line indicates lower values). Tip labels are scientific names followed by strain, then Accession number. The bar indicates the number of substitutions per site.
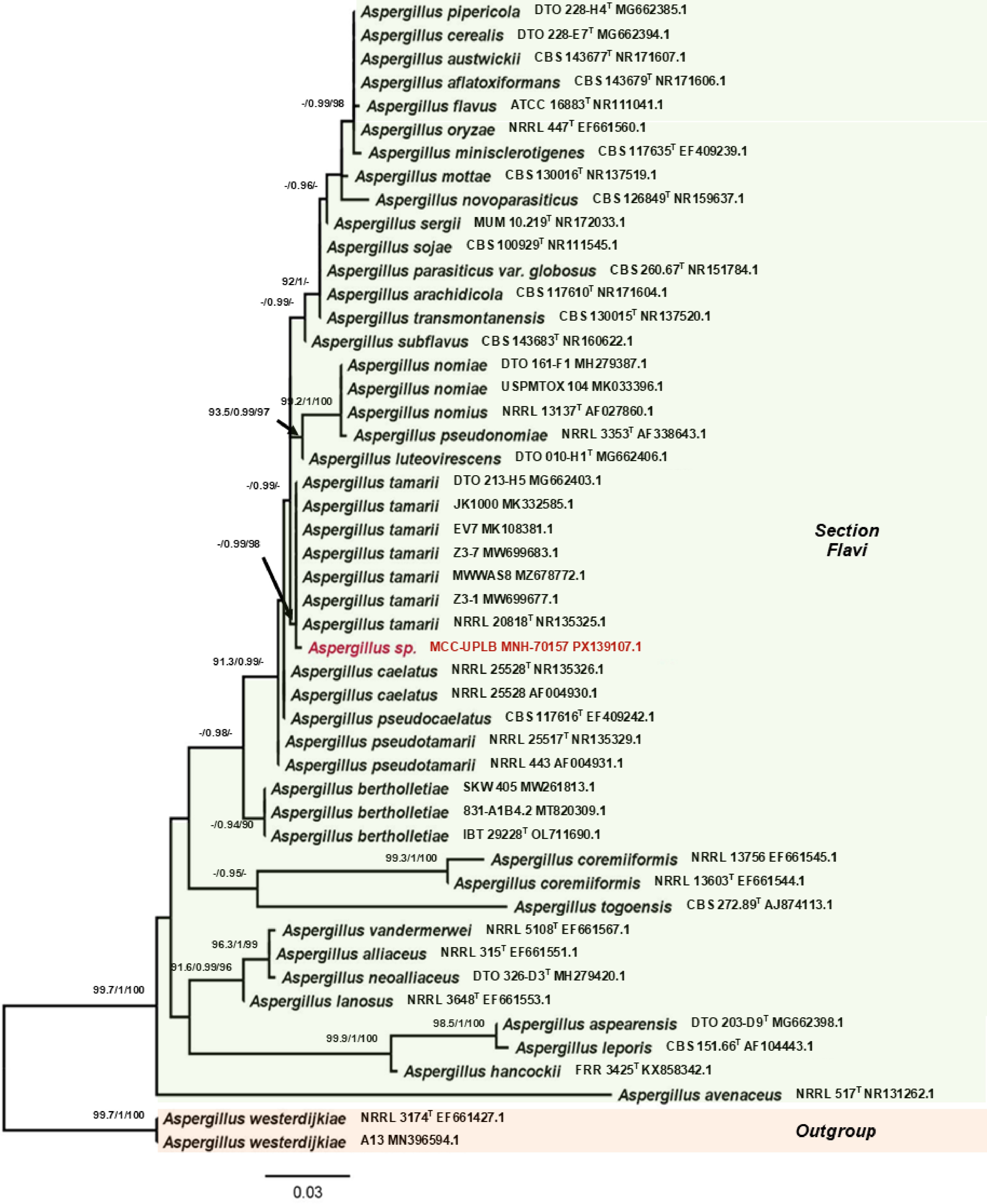
Figure 3.
Phylogenetic tree inferred using the ITS1-5.8S-ITS2 sequence showing the relationships of Aspergillus sp. MCC-UPLB MNH-70157 with representative reference strains (type strains are denoted byT) under the genus Aspergillus section Flavi. Support in nodes is provided by SH-aLRT, aBayes, and UFBoot, where values ≥ 90%, ≥ 0.90, and ≥ 90%, respectively, are shown (a dashed line indicates lower values). Tip labels are scientific names followed by strain, then Accession number. The bar indicates the number of substitutions per site.
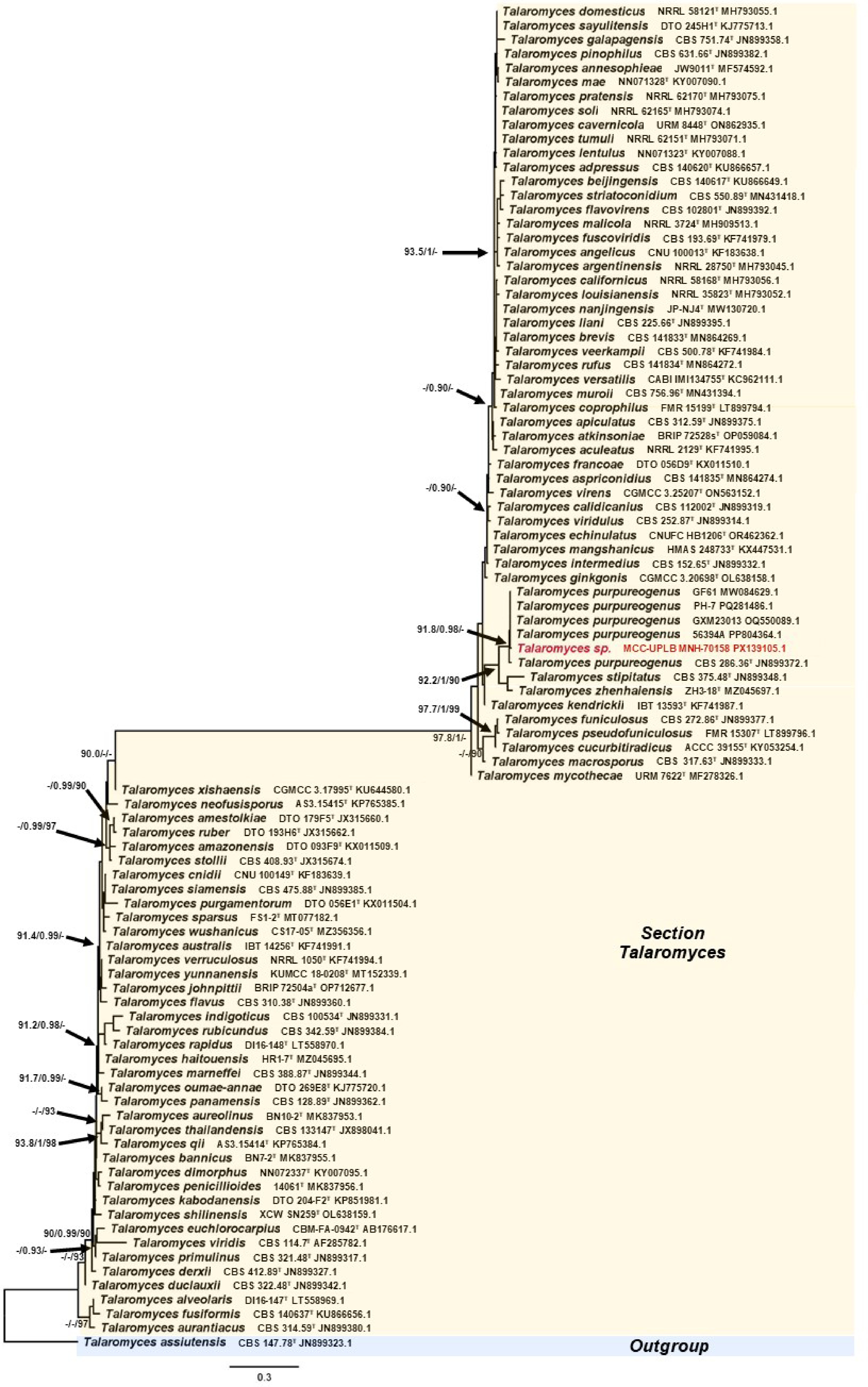
Figure 4.
Phylogenetic tree inferred using the ITS1-5.8S-ITS2 sequence showing the relationships of Talaromyces sp. MCC-UPLB MNH-70158 with reference strains (type strains are denoted byT) under the genus Talaromyces. Support in nodes is provided by SH-aLRT, aBayes, and UFBoot, where values ≥ 90%, ≥ 0.90, and ≥ 90%, respectively, are shown (a dashed line indicates lower values). Tip labels are scientific names followed by strain, then Accession number. The bar indicates the number of substitutions per site.
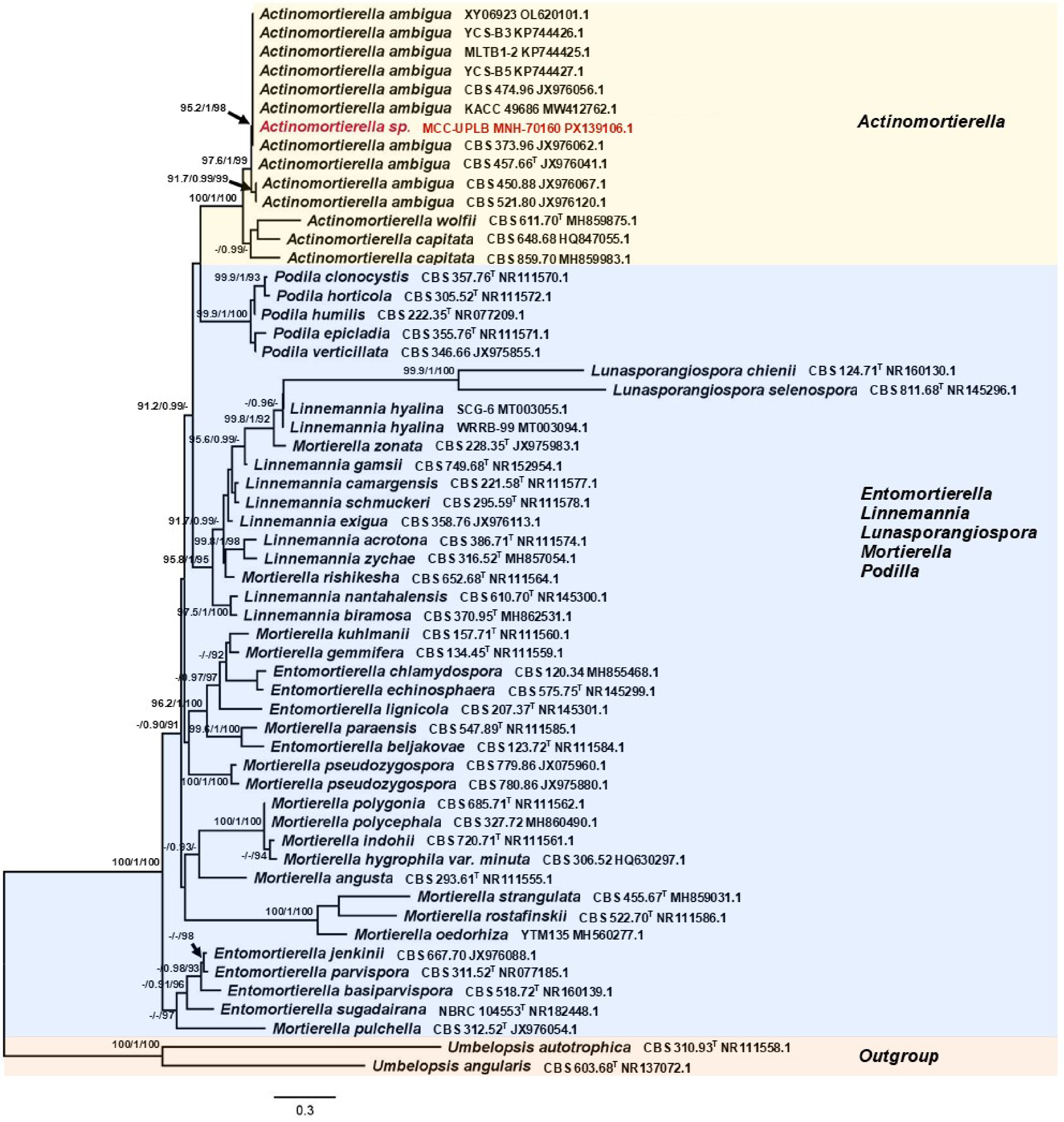
Figure 5.
Phylogenetic tree inferred using the ITS1-5.8S-ITS2 sequence showing the relationships of Actinomortierella sp. MCC-UPLB MNH-70160 with representative reference strains (type strains are denoted byT) under the genus Actinomortierella and closely related genera. Support in nodes is provided by SH-aLRT, aBayes, and UFBoot, where values ≥ 90%, ≥ 0.90, and ≥ 90%, respectively, are shown (a dashed line indicates lower values). Tip labels are scientific names followed by strain, then Accession number. The bar indicates the number of substitutions per site.
Percent mortality of N. nigropictus, microscopic observations, and Koch's postulate
-
The entomopathogenic activities of the fungal isolate Talaromyces sp. Trichoderma sp., Aspergillus sp., and Actinomortierella sp. were assessed against the third instar larvae (characterized with a wedge-shaped body, i.e., head is wider than the abdomen and around 2 mm in size) of green leaf hopper (N. nigropictus) in terms of assessing the mean percent mortality of N. nigropictus, microscopic observations of dead larvae, and Koch's postulate analysis approach on infected larvae. Table 2 presents the mean percent mortality of N. nigropictus third instar larvae 5 d after exposure to conidia (~106) of the four fungi. Meanwhile, Fig. 6 shows the microscopic observations and the Koch postulate analysis of the dead third instar larvae of N. nigropictus.
Table 2. Mean percent mortality of N. nigropictus third instar larvae 5 d after exposure to spores (~106) of the selected fungal isolates.
Culture collection code Putative fungal isolates/control Mean percent mortality of N. nigropictus MCC-UPLB MNH-70158 Talaromyces sp. 43.33 (± 2.89)A MCC-UPLB MNH-70156 Trichoderma sp. 38.33 (± 7.64)AB MCC-UPLB MNH-70157 Aspergillus sp. 26.67 (± 2.89)BC MCC-UPLB MNH-70160 Actinomortierella sp. 18.33 (± 5.77)C Negative Control 1.67 (± 2.89)D *Means (± SD) that do not share a letter are significantly different. ANOVA (Shapiro–Wilk normality); Tukey's Comparison of Means, p < 0.05. Average temperature: am (30.47 °C), noon (33.15 °C), pm (31.72 °C), and humidity: am (72.33%), noon (58.33%), pm (65.17%) of the setup. 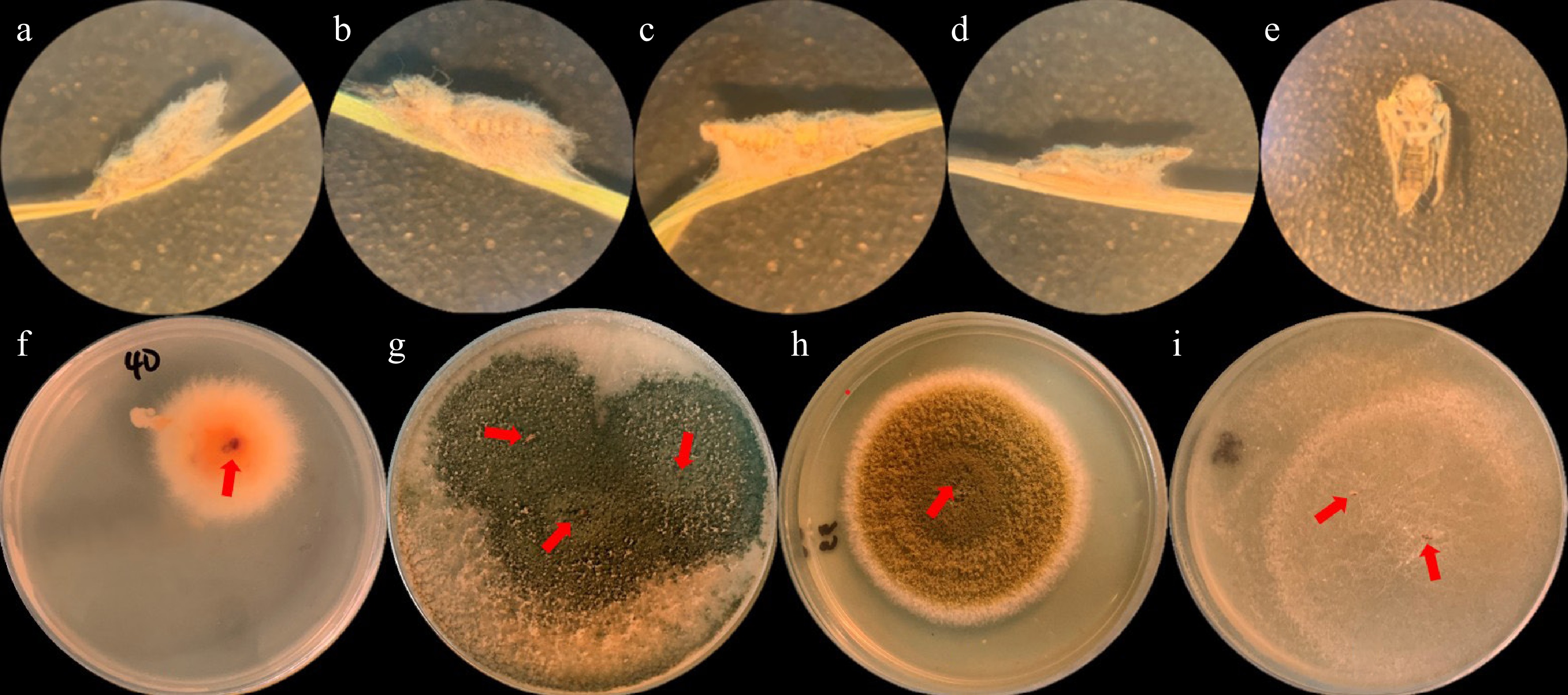
Figure 6.
Microscopic observation of dead N. nigropictus larvae treated with (a) Talaromyces sp., (b) Trichoderma sp., (c) Aspergillus sp., (d) Actinomortierella sp., and (e) Negative control (distilled H2O) using a dissecting microscope (400x magnification). Note the mycelia growing in fungi-treated larvae. Results of Koch postulate analysis of the dead mycelial-infected N. nigropictus (pointed by red arrows): (f) Talaromyces sp., (g) Trichoderma sp., (h) Aspergillus sp., and (i) Actinomortierella sp. after 7 d of incubation at room temperature.
It can be inferred from Table 2 that Talaromyces sp. has the highest mean percent mortality (43.33 ± 2.89) against N. nigropictus and is statistically comparable with Trichoderma sp. (38.33 ± 7.64). Compared to Talaromyces sp., Aspergillus sp. (26.67 ± 7.64) and Actinomortierella sp. (18.33 ± 5.77) have significantly lower mean percent mortality against N. nigropictus but are not statistically comparable with the negative control (1.67 ± 2.89). The microscopic observations of the dead third instar larvae, as shown in Fig. 6a–e, show that the four selected fungi can infect the larvae through prolific mycelial growth. Koch's postulate approach was performed to confirm the fungal mycelia that grew in the larvae where the isolates were introduced. Fig. 6f–i shows Koch's postulate analysis of the mycelial-infected dead larvae grown in PDA after 7 d of incubation. The results confirmed that the isolates introduced were the infecting fungi. With these findings, Talaromyces sp., Trichoderma sp., Aspergillus sp., and Actinomortierella sp. showed entomopathogenic activity against the third instar larva of N. nigropictus.
SEM and histopathological analysis of mycelial-infected N. nigropictus
-
SEM and histopathological analysis of mycelial-infected N. nigropictus were conducted to determine the extent of mycelial infection. Figure 7 shows the result of SEM of the mycelial-infected larval samples. The observed mycelial-covered head (Fig. 7a) and abdominal region (Fig. 7b) of the third instar larvae of N. nigropictus are remarkable. Colonization of structures like the seta was also observed, and observable fissures/slits of the endoskeleton were also seen. Although various factors may have contributed to the cuticular damage observed, the present study does not fully disregard the possibility of fungal enzymatic secretions. However, more studies are necessary to further prove this finding. The results of histopathological analyses of the mycelial-infected samples showed that the selected fungi do not only colonize N. nigropictus third instar larvae superficially, but they also penetrate and invade underlying insect tissues (Fig. 8). Mycelial colonization of internal insect tissue is more prominent in the ovipositor region and the compound eye, as shown by the dark masses in Fig. 8a, d, e. The observed mycelial tissue in between tergites (dorsal body segments) and sternites (ventral body segments), as shown in Fig. 8b–d, indicates fungal mycelial penetration in these sites, resulting in internal colonization of insect tissue with the invading fungal mycelia.
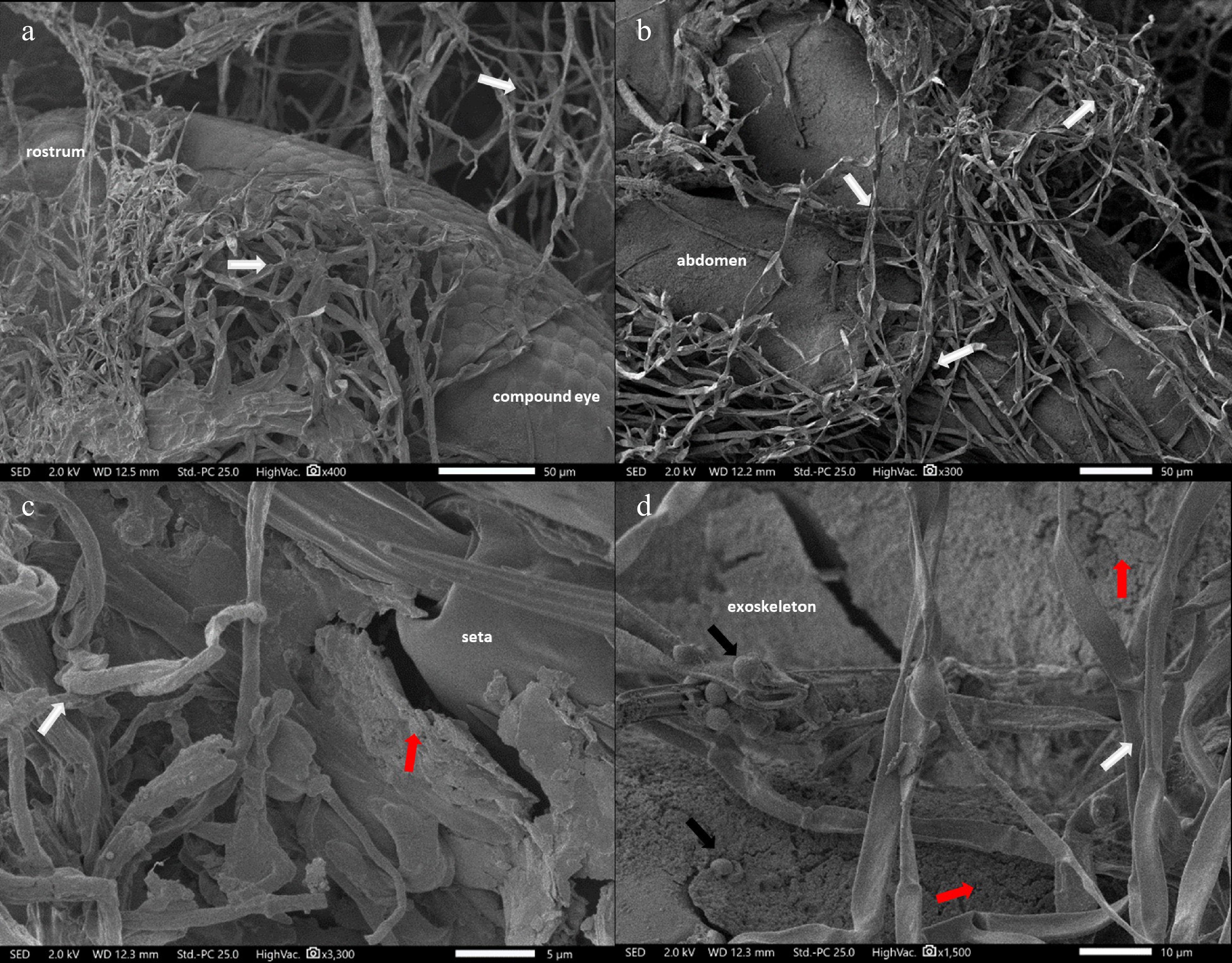
Figure 7.
SEM micrograph of mycelia-infected (indicated by white arrows) N. nigropictus showing: (a) Actinomortierella sp. mycelia-colonized ventral head region. (b) Talaromyces sp. mycelia-colonized ventral abdomen, (c) Aspergillus sp. mycelia-colonized seta, (d) Trichoderma sp. mycelia-colonized exoskeleton with fissures/slits (red arrows). Trichoderma conidia (black arrows). Scale bar: (a), (b) 50 μm, (c) 5 μm, (d) 10 μm.
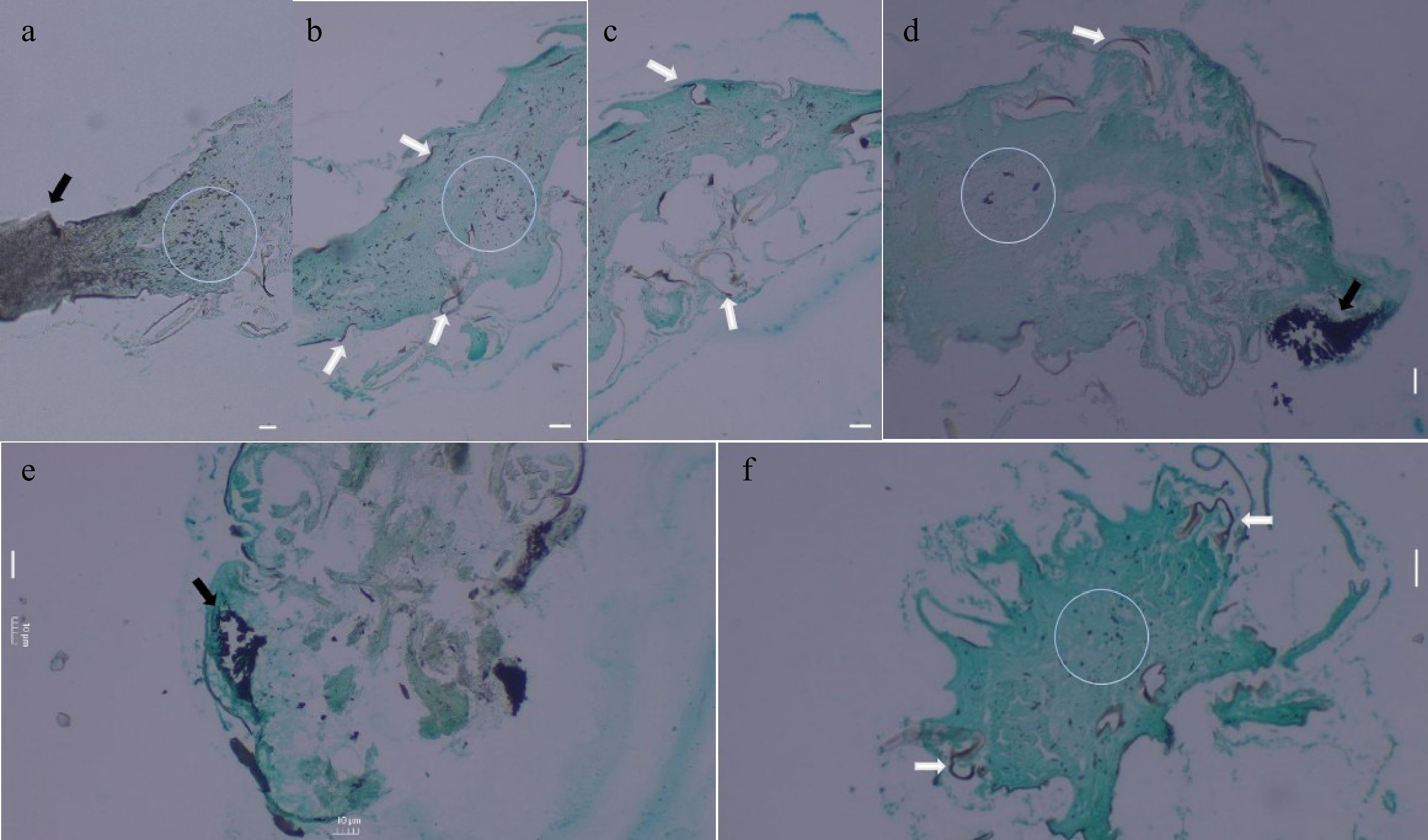
Figure 8.
Results of histopathological analysis of mycelial-infected N. nigropictus third instar larvae (fungal tissues appear black over green insect tissue). (a) Sagittal section showing the densely infected ovipositor region (indicated by black arrow) and portions of the abdomen, (b) Sagittal section showing the abdomen. (c) Sagittal section showing the thorax region. (d) Sagittal section of the head with a densely infected compound eye (indicated by black arrow). Coronal section of the (e) head, and (f) the thorax. (a) Trichoderma sp., (b), (c) Aspergillus sp., (d) Talaromyces sp., and (e), (f) Actinomortierella sp. White arrows indicate penetrating fungal mycelial tissue in between tergites and sternites, while fungal mycelial tissues colonizing the insect's internal tissues are encircled. Scale bar = 10 μm.
-
In the present study, the selected fungal isolates, characterized and preliminarily identified fungal genera from bat guano deposits in Palale Cave, belong to Aspergillus, Talaromyces, Trichoderma, and Actinomortierella. Phylogenetic analysis using the ITS1-5.8S-ITS2 sequence further revealed that Trichoderma sp. MCC-UPLB MNH-70156, Aspergillus sp. MCC-UPLB MNH-70157, Talaromyces sp. MCC-UPLB MNH-70158, and Actinomortierella sp. MCC-UPLB MNH-70160 are closely related to T. asperellum, A. tamarii, T. purpureogenus, and A. ambigua, respectively, with strong branch support values. These are provided by likelihood ratios (SH-aLRT), probabilistic inference (aBayes), and resampling (UFBoot). Reporting all three metrics provides a comprehensive and complementary picture of branch reliability, which has been extensively analyzed by previous studies[33−35]. For confirmation, however, multigene phylogenetic analysis using additional markers can be employed to corroborate this finding.
The result of the present study coincides with the findings of previous research such that Aspergillus, Talaromyces, and Penicillium were among the most frequently reported genera in the studies of Brazilian cave mycobiota[36] and Caatinga dry forest bat caves of Brazil[37], while Penicillium and Aspergillus are dominant fungal strains from bat guano in limestone caves of Borneo along with Trichoderma and Cladosporium[38]. The GenBank closest match of the isolate MCC-UPLB MNH-70160 of the present study is originally to Mortierella ambigua; however, it has been reclassified to the genus Actinomortierella by Vandepol et al.[39] based on molecular analyses using ribosomal and non-ribosomal DNA markers. Nonetheless, novel species of Mortierella were isolated by Karunarathna et al.[40] from bat carcasses in the caves of Yunnan Province, China, while Jurado et al.[41] recently reported the presence of Mortierella in Gypsum Cave biofilm communities as strongly influenced by bats and arthropod-related fungi. Notably, the first reported Actinomortierella isolated from a cave environment, particularly from a soil sample in Le Stegodon Cave of Satun UNESCO Geopark in Thailand, is the novel species Actinomortierella caverna (C. Srihom, Preedanon, S. Saengkaewsuk & Somrith) (Mortierellales: Mortierellaceae)[42]. Insofar as bat guano from a cave environment is concerned, the present study is the first to report an isolated Actinomortierella species. Considering the possible arthropod-fungi association of fungal isolates from bat guano, their potential entomopathogenicity was assessed.
The fungal isolates in this study, namely, Talaromyces sp., Trichoderma sp., and Aspergillus sp., belong to Ascomycota, while Actinomortierella sp. belongs to Mucoromycota, which are among the phyla in which entomopathogenic fungi are present[14,43]. Nicoletti & Becchimanzi[44] reviewed the growing reports of the association of many Talaromyces species as antagonists or mutualists with insects belonging to nine orders, such as Blattodea, Coleoptera, Diptera, Hemiptera, Hymenoptera, Lepidoptera, Orthoptera, Thysanoptera, and Trichoptera. N. nigropictus belongs to the order Hemiptera. In the present study, Talaromyces sp. shows entomopathogenic activity against the third instar larva of N. nigropictus. Third instar larvae are often used in biocontrol studies since they are typically large enough for observation and are characterized by active feeding and growth. While susceptibility can vary between instars, the third instar often represents a key period in the life cycle of an insect where it is vulnerable to attack by natural enemies[45−48].
Meanwhile, Trichoderma is widely studied as a biocontrol agent against pathogenic fungi in agriculture. It showed biocontrol capabilities against insect pests via mechanisms including parasitism or the production of antifeedant compounds and secondary metabolites with insecticidal and repellent properties[49]. The entomopathogenic activity of Trichoderma, along with other fungal species, such as Aspergillus, against brown plant hopper [Nilaparvata lugens (Stål, 1854) (Hemiptera: Delphacidae)] was demonstrated by Trizella et al.[50]. Zhang et al.[19] recently reported the initial record of Aspergillus nomiae (Kurtzman, B.W. Horn & Hesselt) (Eurotiales: Aspergillaceae) as a broad-spectrum entomopathogenic fungus providing resistance against insect pests. As close relatives of Actinomortierella, Edgington et al.[51] reported the entomopathogenic activities of Mortierella alpina (Peyronel) (Mortierellales: Mortierellaceae) and Mortierella signyensis (K. Voigt, P.M. Kirk & Bridge) (Mortierellales: Mortierellaceae), which caused significant mortality (p < 0.05) against wax moths and houseflies. Yoder et al.[52] also reported the significant virulence (p < 0.05) of M. alpina against winter tick, Dermacentor albipictus (Packard) (Ixodida: Ixodidae). Limited studies show the entomopathogenicity in Actinomortierella, and the present study is the first to show entomopathogenic activity of an Actinomortierella sp., isolated from bat guano deposits, against the third instar larvae of N. nigropictus.
Several studies have demonstrated the potential application of these isolates as promising biocontrol agents. Afandhi et al.[53] showed higher larvicidal activity of Trichoderma asperellum compared to Beauveria bassiana against the fall armyworm, Spodoptera frugiperda (J.E. Smith) (Lepidoptera: Noctuidae), which affects maize and causes huge economic losses. Baskar et al.[54] demonstrated the mosquitocidal potential of Aspergillus tamarii against Aedes aegypti (Linnaeus) (Diptera: Culicidae) and Culex quinquefasciatus (Say) (Diptera: Culicidae), while Yue et al.[55] reported the insecticidal activity of Talaromyces purpureogenus BS5 against the Locusta migratoria manilensis (Meyen) (Orthoptera: Acrididae), which is a threat to crop production due to massive locust invasion.
The findings of the present study demonstrate positive entomopathogenic activities of the selected fungi from bat guano against N. nigropictus third instar larvae. The highest mean percent mortality of N. nigropictus third instar larvae exposed to conidia of Talaromyces sp. MCC-UPLB MNH-70158 suggests the potential of this isolate for further efficacy studies, i.e., involving dose-response and time-course mortality studies. Of note, the activity of this isolate against N. nigropictus is statistically comparable to that of Trichoderma sp. MCC-UPLB MNH-70156, a genus often used as a biocontrol agent in agriculture[56]. While a lower mean percent mortality of N. nigropictus third instar larvae has been observed in Aspergillus sp. MCC-UPLB MNH-70157 and Actinomortierella sp. MCC-UPLB MNH-70160 compared to Talaromyces sp. MCC-UPLB MNH-70158, their entomopathogenic potential against N. nigropictus third instar larvae is undeniable, just like the other isolates, as further confirmed by the stereomicroscopic, ultrastructural, and histopathological analyses of the mycelial-infected larvae in this study.
The prolific mycelial growth observed throughout the body (particularly in the head and abdominal region) of the larvae via stereomicroscopic observations and ultrastructural analysis using SEM indicates pathogenic infection and proliferation of the isolates, a feature common to entomopathogenic fungi[19]. Moreover, the re-isolation of the infecting isolates in this study from the cadaver of N. nigropictus third instar larvae, as described in Koch's postulates[57], strengthens the pathogenic nature of these fungal isolates against N. nigropictus third instar larvae. The observed fissures and slits in the exoskeleton of the mycelial-infected larvae, as revealed by the SEM analysis, give insights into the mechanistic action of their infection, possibly involving enzymatic secretions that weaken the integrity of the exoskeleton of the larvae[19]; however, more studies are necessary to further prove this finding. Of note, the hyphal penetration that is also observed in-between the tergites and sternites of the larvae demonstrates internal colonization in the insect tissues, causing the death of the larvae due to several mechanisms involving secretions of toxins or fungal metabolites and successful evasion of antimicrobial responses of the host insect[17,18].
Globally, there has been an increasing trend in the use of synthetic insecticides in agricultural pest management over the years. Inadvertently, these synthetic insecticides bring negative impacts to the environment and the well-being of many living organisms in the long run due to their immutable nature[58−60]. This study, therefore, is relevant to the pursuit of sustainable, effective, and eco-friendly alternatives to synthetic insecticides. Since EPFs are naturally derived, they do not harm the environment and work well as natural enemies of agricultural pests. Moreover, entomopathogenic compounds from EPFs can be extracted and studied for the possible development of sustainable, more efficient, and specific bioinsecticides against significant insect pests in agriculture. The mycoflora from bat guano deposits can be a potential source of naturally-derived entomopathogens. While the entomopathogenic activity of the isolates in this study offers great potential, some limitations need to be addressed, such as field validation, assessment of host specificity, and potential non-target effects on beneficial insects. The researchers seek to address these limitations in the future and are also interested in profiling the entomopathogenic compounds present in the EPF isolates in an attempt to develop an effective and sustainable bioinsecticide for use in agriculture.
-
In conclusion, Talaromyces sp. MCC-UPLB MNH-70158 and Trichoderma sp. MCC-UPLB MNH-70156 have significantly higher mean percent mortality against Nephotettix nigropictus third instar larvae, compared to Aspergillus sp. MCC-UPLB MNH-70157, and Actinomortierella sp. MCC-UPLB MNH-70160. Moreover, microscopic observations showed that all fungi are capable of infecting the larvae with prolific mycelial growth, which was further confirmed and validated by the Koch postulate approach. Furthermore, SEM showed surface fungal mycelial colonization of the larvae, while histopathological analysis showed that internal colonization of fungal mycelia also happens predominantly in the ovipositor and compound eye regions. Hence, Talaromyces sp., Trichoderma sp., Aspergillus sp., and Actinomortierella sp., in this study, are entomopathogenic to N. nigropictus third instar larvae. The findings of this study have practical value for the agricultural sector, particularly in rice production. To fully assess their effectiveness, future studies (benchmarking with positive control EPFs), such as assessment of the dose-response curve and time-course mortality, can be investigated. Other staining methods, such as Periodic Acid-Schiff (PAS) and Calcofluor white staining methods, can also be explored for comparison. Moreover, further studies, such as entomopathogenicity tests on other insect pests, such as armyworms, stem borers, thrips, and similar pests, which have significant impacts on agriculture, can be studied for cross-host efficacy testing. The formulation of bionano-pesticide making use of secondary metabolites, enzymes, or entomopathogenic substances from these fungi can also be explored and offers promising potential. In general, the findings of this study give insights into bat guano as a potential source of entomopathogens.
The researchers extend their sincere gratitude to the University of the Philippines Los Baños Museum of Natural History (UPLB MNH) Niche Center for Research and Development in the Region (NICER) Center for Cave Ecosystems Research (CAVES) and Microbial Culture Collections (MCC). The researchers thank the Microbiology Division of the Institute of Biological Sciences (IBS), College of Arts and Science (CAS), and the Institute of Weed Science, Entomology, and Plant Pathology (IWEP) of the College of Agriculture and Food Science (CAFS) at the University of the Philippines Los Baños (UPLB) for allowing the researchers to conduct the laboratory experiments for this project. Also, appreciation and thanks are given to the Veterinary Histopathology Laboratory (Mr. Maurell M. Navasero), Veterinary Paraclinical Sciences of the College of Veterinary Medicine (CVM), UPLB, and the Integrated Electron Microscopy Laboratory (Mr. Reuben Miguel S. Felix), Central Instrumentation Facility, De La Salle University – Laguna Campus. Special thanks to Dr. Connie C. Gibas, a clinical mycologist at the University of Texas Health San Antonio, Texas, USA. Profound gratitude is accorded to the Department of Science and Technology (DOST) Grants-in-Aid and the Philippine Council for Agriculture, Aquatic and Natural Resources Research and Development (PCAARRD) for the financial support given to the project. This project was funded by the Department of Science and Technology, Republic of the Philippines, under the Grant No.: QMSR-FERD-IED-BIODIVE-643-4300.
-
The authors confirm their contributions to the paper as follows: study conception and design: Lopez MKS, De Leon MP, Sabino NG, Lantican NB; data collection, draft manuscript preparation: Lopez MKS; analysis and interpretation of results: Lopez MKS, Sabino NG. All authors reviewed the results and approved the final version of the manuscript.
-
All the data generated and analyzed in this study are available as a Supplementary File S1. DNA sequence data are available in the GenBank database, and the Accession numbers are provided in Table 1, while the information of the different reference strains used in phylogenetic analysis is provided as Supplementary File S2.
-
The authors declare that they have no conflict of interest.
-
accompanies this paper online at: https://doi.org/10.48130/sif-0026-0010.
- Supplementary Fig. S1 The experimental setup for the entomopathogenicity test in this study.
- Supplementary Fig. S2 Protease activity of the isolates.
- Supplementary File S1 Entomopathogenicity setup.
- Supplementary File S2 Protease enzymatic screening.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Lopez MKS, Sabino NG, Lantican NB, De Leon MP. 2026. Entomopathogenicity of fungal isolates from bat guano deposits in Palale Cave, Tayabas, Quezon Province, Philippines against rice green leafhopper (Nephotettix nigropictus Stål). Studies in Fungi 11: e012 doi: 10.48130/sif-0026-0010
Entomopathogenicity of fungal isolates from bat guano deposits in Palale Cave, Tayabas, Quezon Province, Philippines against rice green leafhopper (Nephotettix nigropictus Stål)
- Received: 16 March 2025
- Revised: 01 February 2026
- Accepted: 05 March 2026
- Published online: 30 April 2026
Abstract: Nephotettix nigropictus Stål transmits rice pathogens such as yellow dwarf mycoplasma-like organisms, rice dwarf viruses, rice transitory yellowing viruses, and rice tungro viruses; hence, it requires effective control measures. Biological control strategies, such as the use of entomopathogenic fungi, are eco-friendly, targeted, and safer alternatives to chemically synthesized insecticides. This study aimed to investigate the entomopathogenic activities of several fungal isolates from bat guano deposits in Palale Cave, Tayabas, Quezon, Philippines, against the third-instar larvae of N. nigropictus. The fungi were characterized and identified using phenotypic (cultural and morphological features) and genotypic (ITS marker) methods. Entomopathogenicity was assessed through the mean percentage mortality of N. nigropictus third instar larvae exposed to conidia of Talaromyces sp., Trichoderma sp., Aspergillus sp., and Actinomortierella sp. Microscopic observation showed that prolific mycelial growth covered the third instar larvae, and the Koch postulate approach confirmed that the selected fungi are the infecting mycelia. Scanning electron microscopy confirmed the presence of the fungal mycelia covering the head and abdominal region of the larvae. Histopathological analysis using modified Grocott silver nitrate staining showed internal colonization and dominance of the fungal mycelial tissue on the ovipositor region and the compound eyes. In the abdomen of the larvae, fungal mycelial penetration was also observed between tergites and sternites. The promising findings of this study can provide avenues for the development of bioinsecticide agents against N. nigropictus and potentially other insect pests, such as armyworms, stem borers, and thrips that affect crops, particularly rice, the most important staple crop in the Philippines.
-
Key words:
- Bat guano /
- Entomopathogenicity /
- Fungi /
- Histopathology /
- Nephotettix /
- Scanning electron microscopy













