-
Seed production in sugar beet represents a major bottleneck in India, due to the crop's strict requirement for vernalization—a prolonged exposure to cool temperatures necessary for floral induction. The absence of such climatic conditions in subtropical plains severely restricts local seed multiplication and self-sufficiency. Unlike sugar production, which prioritizes root biomass accumulation, seed production necessitates distinct physiological and environmental conditions to enhance reproductive success[1]. Sugar beet is a temperate, biennial crop that requires a prolonged period of cool temperatures (vernalization) to induce bolting and flowering[2]. Vernalization for sugar beet typically requires exposure to cool temperatures in a band roughly between ~0–12 °C for several weeks (often 6–14 weeks depending on genotype)[3]. However, most of India's plains and subtropical regions rarely provide these prolonged, suitably low temperatures, resulting in weak or absent natural vernalization. Consequently, reliable bolting and seed formation are difficult to achieve under such field conditions. To address this, seed production is often limited to hill stations and temperate pockets, or relies on imported seed, which is both costly and logistically demanding[1]. The limited number of suitable sites and narrow climate windows further constrain national seed self-sufficiency and pose challenges under climatic conditions[1].
To overcome vernalization constraints, different planting methods such as steckling transplantation, direct seeding, and pit method planting have been developed as strategic approaches to facilitate reproductive induction under suboptimal environments. The steckling planting method is an efficient approach for sugar beet seed production under such environments. This method involves growing small vegetative plants—known as steckling—from harvested roots[4,5]. These steckling are produced during the first season and are then transplanted the following season into seed production fields at the onset of favorable climatic conditions. This two-phase approach not only ensures reliable flowering and seed setting, but also allows for early selection of superior plants, enabling breeders to screen for desired traits such as disease resistance, vigor, and uniformity before transplantation. Furthermore, the steckling method significantly reduces the total duration of the seed production cycle to approximately 9–10 months, saving nearly 5–6 months compared to the traditional direct-seeded method. The process enhances synchronization of flowering between male and female lines, resulting in improved pollination efficiency, and higher seed yield[4–6]. In contrast, the direct-seeded method involves simultaneous sowing of male and female parental lines for natural pollination. Seeds are harvested exclusively from female plants after pollination, and the male parent is subsequently eliminated to maintain genetic purity[7]. The pit planting method is an experimental adaptation designed for high-altitude locations that experience snowfall during the transplantation period. In this technique, stecklings are placed within soil pits to protect them from extreme cold and snow accumulation. The pits create a relatively insulated microenvironment, preventing frost damage and ensuring better establishment and survival of the stecklings under adverse climatic conditions.
While planting methods address the physiological constraints of vernalization, achieving high seed yield and quality also depends on optimizing nutrient availability, particularly nitrogen, phosphorus, and potassium. The quality of sugar beet seeds is heavily influenced by environmental conditions and the agricultural practices employed during production[7]. Optimizing planting techniques, selecting appropriate varieties, and applying the right fertilizer regimes are critical factors that affect seed yield, viability, and economic feasibility. Recent advancements in agronomic practices emphasize the need for precise seed production methodologies tailored to specific crop varieties and environmental conditions. Fertilizer application strongly influences reproductive success, seed viability, and overall productivity in sugar beet[8]. Nitrogen (N), phosphorus (P), and potassium (K), are key macronutrients essential for crop development and significantly influence the yield and quality of field crops[9]. Sugar beet is particularly responsive to fertilization and highly sensitive to N, P, and K availability[10]. Studies indicate that deficiencies in N and P can reduce stomatal conductance, limit photosynthesis, and constrain the plant's hydraulic conductivity, ultimately affecting growth and productivity[11]. While N is crucial for vegetative growth and chlorophyll synthesis, excessive application can lead to excessive foliage development at the expense of reproductive structures. Over-fertilization with N can be particularly detrimental to sugar beet as it promotes vegetative growth, reduces sugar accumulation, and negatively impacts overall quality and economic returns[12]. Proper N management is therefore essential to maintain a balance between vegetative and reproductive growth while minimizing environmental impacts[13]. Research has confirmed that both N and K contribute to improved yield and quality in sugar beet by enhancing organic metabolite biosynthesis and overall plant nutritional status[14]. K plays a multifaceted role in sugar beet physiology, particularly in enzyme activation, leaf photosynthesis, sugar metabolism, and protein synthesis, while also improving stress tolerance[10]. Several studies have established a strong positive correlation between K fertilization and increased dry matter accumulation, root tuber yield, sugar content, and overall sugar yield in sugar beet[15].
Despite several international studies exploring fertilizer effects and seed production physiology in sugar beet, there remains a clear gap in region-specific optimization for subtropical India. The present study represents the systematic attempt to integrate the steckling method with an optimized NPK regime under Indian agro-climatic conditions. This combined approach aims to establish a reproducible, climate-adapted seed production model capable of enhancing bolting response, flowering synchronization, and seed viability in environments lacking natural vernalization.
Sugar beet seed production success relies on high-performing varieties, proper spring field establishment, and sound agronomic practices for optimal seed viability and germination potential[7]. Therefore, this study uniquely evaluates the synergistic effect of planting methods and nutrient optimization in Indian subtropical conditions, where such integrated investigations have not been conducted previously. The present study aims to: (1) evaluate the comparative effectiveness of different planting methods (steckling, direct seeding, and pit planting) for enhancing bolting, flowering synchronization, and seed yield of sugar beet under Indian agro-climatic conditions; and (2) assess the impact of two NPK fertilizer regimes—conventional (120:60:60 kg ha−1) and optimized (120:80:80 kg ha−1)—on reproductive performance, seed viability, and germination rate.
The central hypothesis of this study posits that an optimized nutrient regime (120:80:80 kg ha−1 of N:P:K) will markedly enhance the reproductive efficiency of sugar beet—reflected through higher bolting frequency, improved flowering synchronization, and superior germination performance—compared to the conventional 120:60:60 kg ha−1 regime under subtropical conditions. Additionally, it is hypothesized that the steckling planting method, owing to its controlled establishment phase and enhanced vernalization response will exhibit superior performance over direct seeding and pit planting methods, resulting in greater seed yield, quality, and overall reproductive success.
-
The investigation took place over three consecutive years with three replications at both the Lucknow farm of ICAR-Indian Sugarcane Research Institute (Uttar Pradesh, India), considered the first experimental site, and the ICAR-ISRI Sugar beet Breeding Outpost in Mukteshwar (Nainital, India), considered as the second experimental site. The first experimental site is located at 26°56' N, 26°52' E, and 111 m above mean sea level with a semi-arid subtropical climate having dry hot summers and cold winters. The soil was sandy loam (13.3% clay, 24.5% silt, and 62.2% sand) of Indo-Gangetic alluvial origin, very deep (> 2 m), well drained, flat, and classified as non-calcareous mixed hyperthermic Udic Ustochrept, with soil pH of 7.4. The second experimental site is at 29°23' N, 79°30' E with an altitude of 2,286 m above mean sea level, located on a ridge on the southern edge of the North-Western Himalaya. The average annual minimum temperature was 8.8 °C and average annual maximum temperature was 19 °C. The total rainfall was 1,286 mm, squall was zero. Number of rainy days was 71.6, hail 6.7 d/year; thunder 51.4 d/year, and fog 61.2 d/year. The soil is Typic Haploudoll (USDA Classification) which is slightly acidic (pH 5.6–5.9) and sandy loam to loamy sand in texture (clay 12%–16%). Prior to planting, the experimental field underwent thorough tillage to achieve optimal soil conditions.
Variety, treatments, experimental design, and procedures
-
The three factors in this experiment were sugar beet varieties, planting methods, and method of NPK fertilizer application, each having different levels. Six ICAR-ISRI, Lucknow developed genotypes were utilized (Table 1) including four indigenous genotypes (LKC 2006, LKC 2010, LKC 2007, and LKC 2020) and two commercially released varieties (IISR Comp 1 and LS 6). Three planting methods were tested, including Steckling (E1), Direct seeding (E2; D-Seeded), and Pit method (E3) for cold treatment. To maintain the plant population, the row spacing was made constant at 50 cm, whereas the spacing between plants or hills was 20 cm for Steckling, 40 cm for D-Seeded and 15 cm for the Pit method. Sugar beet was planted in raised ridges measuring 10−12 cm in height and 20 cm in width. The first fertilizer composition (F1) consists of 120 kg N, 60 kg P2O5, and 60 kg K2O per ha, while the second fertilizer composition (F2) consists of 120 kg N, 80 kg P2O5, and 80 kg K2O per ha. In the F2 dosage, P and K are given as basal doses, with a split application of N at critical growth stages (sowing, thinning, and during earthing up), supported by need-based irrigation. In the F1 dosage, K and P were applied at three distinct stages: initially at sowing, then at thinning, and finally immediately after earthing-up in December, based on soil test values. The three-factor experiment was then conducted in a 3 × 2 × 6 factorial arrangement using a randomized complete block design (RCBD) with three replications.
Table 1. Characteristics of ICAR-ISRI identified sugar beet genotypes used in the study.
Genotypes Characteristics LKC 2020 High ethanol potential even under drought stress, tolerance to Spodoptera litura (Armyworm) LKC 2010 Drought tolerance, elevated sucrose content, and robust bioethanol recovery LKC 2006 High temperature tolerant LKC 2007 Water deficit stress tolerant; high root yields and superior juice quality; high temperature tolerant IISR Comp 1 High temperature tolerant; salinity tolerant LS 6 High sugar content The Steckling (E1) method is a specialized approach utilized in the production of sugar beet seeds, involving a two-stage process across distinct fields (Table 2). These young seedlings, typically aged eight weeks and exhibiting a growth stage of four to six leaves, are carefully selected based on their robustness and root characteristics, with a desired root length falling within the range of 6−14 cm. Following minor root selection, the chosen stecklings are transplanted directly into designated fields at the ICAR-ISRI Sugar beet Breeding Outpost, Mukteshwar, after undergoing meticulous trimming of leaves and petioles to optimize transplantation conditions, with petiole sizes standardized at 4−5 cm. Over subsequent months, these transplanted stecklings are nurtured to maturity, culminating in the harvesting of seeds in September (Fig. 1).
Table 2. Description of different planting methods.
Planting/sowing method Steckling (E1) Pit method (E3) Direct seeded (E2) Location Lucknow (sowing) Mukteshwar (transplanting) Mukteshwar Sowing 1st fortnight of October May/June Transplanting 2nd fortnight of December 1st fortnight of March Not applicable Seed harvesting Aug (2nd fortnight) to September (1st fortnight) Key activities Sowing and steckling preparation at Lucknow (October–December); selection, trimming, and transport of healthy stecklings to Mukteshwar and transplantation, crop care, flowering, and seed setting (December–September). Sowing at ICAR-ISRI, Lucknow (October); selection and transport of stecklings to Mukteshwar; placement of roots in pre-dug pits (60 cm × 100 cm) before snowfall; natural cold treatment for vernalization during winter; retrieval and field transplantation after snowmelt and crop growth, flowering, and seed harvest (July–September). Direct sowing of sugar beet seeds at Mukteshwar (May–June); continuous crop growth without transplantation; regular irrigation, nutrient, and pest management; flowering, seed maturation, and harvesting (September next year). The D-seeded (E2) method is a streamlined approach employed in the production of sugar beet seeds, characterized by uninterrupted cultivation from seed to seed within a single field. The process involves sowing sugar beet seeds directly into the designated field. This method ensures minimal disturbance to the crop's growth trajectory, allowing a seamless transition from seedling to mature plant. Sugar beet plants are nurtured under optimal conditions, with careful attention to irrigation, pest control, and soil management practices. The direct-seeded method's peak occurs in September of the following year when mature seeds are harvested from sugar beet plants (Table 2).
The Pit method (E3) represents a specialized technique utilized in the production of sugar beet seeds, particularly effective in regions of higher altitudes where snowy conditions prevail. This method is employed for genotypes that exhibit delayed germination, leading to late steckling production or in cases where stecklings are not properly developed. To address this, a pit is dug at Mukteshwar, and once the stecklings are ready for transplantation, they are sent to this location. Sugar beet roots are placed in pits for cold treatment during snowy seasons to prevent root deterioration and enhance seed quality. Gradual cold treatment stimulates dormancy and promotes seed development. After the snow melts, the vernalized stecklings are removed from the pit and transplanted into the field to continue their subsequent growth phases. The harvest of mature seeds from the cultivated sugar beet plants occurs in the month of September.
Data collection and analysis
-
To evaluate the planting type's effectiveness on sugar beet seed production, various parameters after seed sowing were monitored, including germination percentage, days to bolting, days to stalk formation, days to flowering, and days to seed harvesting. Germination percentage refers to the proportion of seeds that successfully sprout. Days to bolting (DB) indicate the duration until the plants start producing flower stalks. DB were recorded when 70% of the plants in a plot showed bolting. Bolting percentage represents the total number of plants with bolting, out of the total number of plants per plot[16]. Plot-based analysis is also used to determine other characters, such as stalk percentage, days to flowering (DF), and days to seed harvesting (DSH). In assessing the impact of fertilizer application on yield, test weight of 100 seeds was calculated and yield per plant was observed. The statistical analysis followed a randomized complete block design (RCBD) with a factorial arrangement, as outlined by Gomez & Gomez[17]. When significant differences were observed, mean separations were performed using Duncan's New Multiple Range Test.
-
The experiment revealed significant effects (p < 0.05) of planting methods, fertilizer doses, and genotypic variation on the number of days to bolting (DB), and bolting percentage (BP) in sugar beet (Fig. 2, Supplementary Table S1). Among the planting methods, the in situ (E2) method consistently exhibited the highest DB values across all varieties, indicating a pronounced delay in bolting and an extended vegetative phase compared to E1 (direct sowing) and E3 (pit planting). This delayed bolting under E2 suggests its potential for maintaining vegetative growth for longer periods, which can be advantageous for root development and yield. In contrast, the E1 method resulted in significantly earlier bolting (lower DB values) across varieties, reflecting faster phenological progression. This pattern indicates that E1 favors rapid establishment and early flowering, which may be desirable under short-season or early planting conditions, but could limit root biomass accumulation. The E3 method produced intermediate DB values, suggesting it can partially compensate for delayed or suboptimal E1 establishment by inducing moderate bolting responses. Varietal differences were also evident (p < 0.05), with LKC 2020 and LKC 2010 exhibiting higher bolting tendencies across planting methods, while LKC 2006 consistently showed delayed bolting, confirming its relatively low flowering propensity.
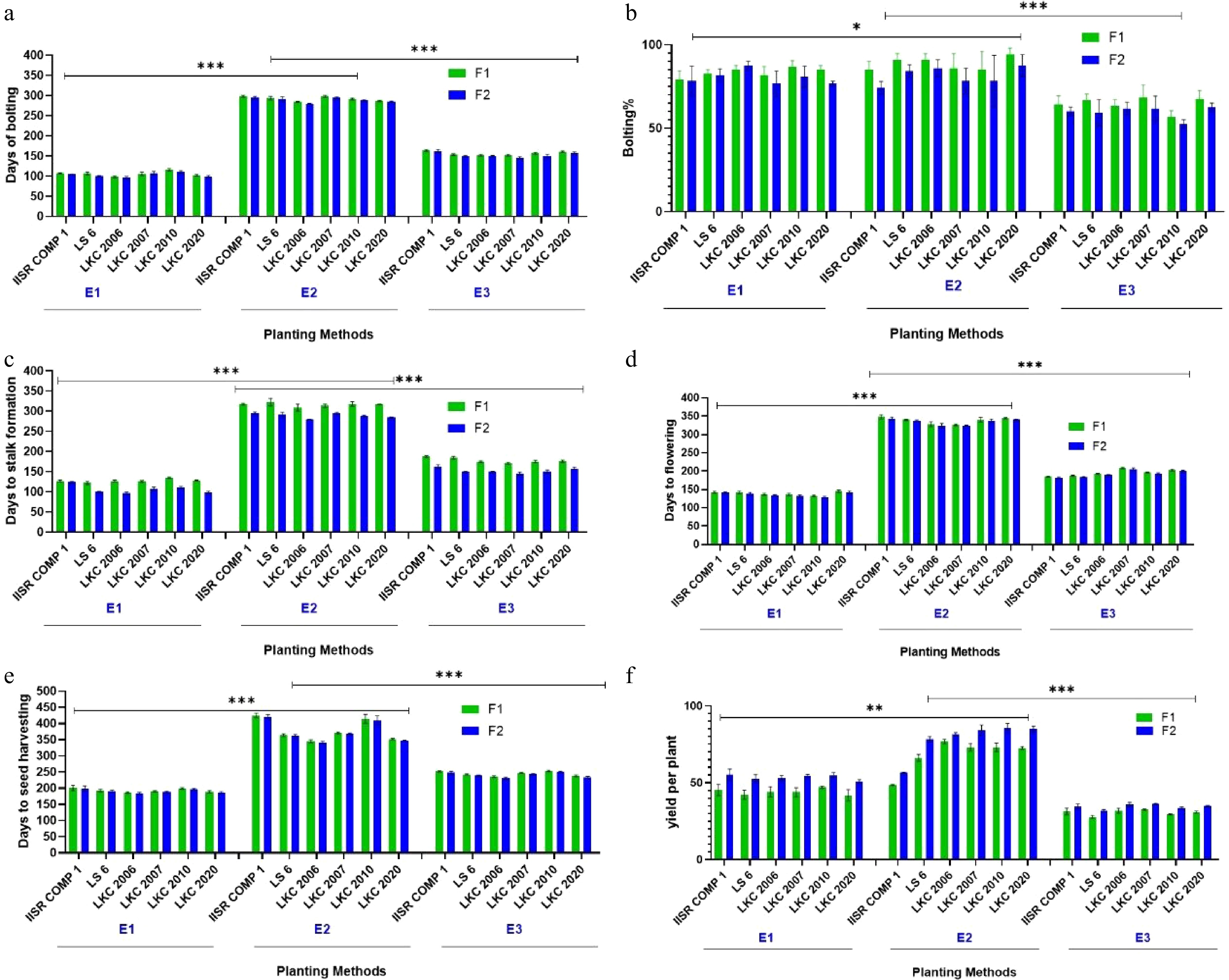
Figure 2.
Comparative assessment of planting methods and fertilizer doses based on recorded observations (a) Days to bolting. (b) Bolting per cent. (c) Days to stalk formation. (d) Days to flowering. (e) Days to seed harvesting. (f) Yield per plant. *** Highly significant (p < 0.001), and ** or * denotes significant differences (p < 0.05).
Fertilizer application further influenced bolting behavior. An increased nutrient supply under F2 (higher P and K doses) generally led to a reduction in DB, particularly in E1 and E3, implying that enhanced nutrient availability may accelerate physiological maturity and induce earlier bolting. However, under E2, fertilizer effects were minimal, suggesting that environmental or microclimatic conditions associated with in situ growth exerted a stronger influence than nutrient availability on bolting dynamics. Bolting percentage (BP) also varied significantly (p < 0.05) among treatments (Supplementary Table S2). The E2 method consistently recorded the highest BP, reaching nearly 97% in LKC 2020, confirming its effectiveness in promoting uniform bolting. The E1 method showed moderate BP across genotypes, while E3 consistently exhibited the lowest BP, indicating possible suppression of floral induction under restricted pit conditions. Among genotypes, LKC 2020 demonstrated the highest bolting potential across all conditions, whereas LKC 2010 showed marked variability, depending on planting method and nutrient regime.
Overall, the results highlight that the E2 planting method significantly delays bolting, while E1 accelerates it, and E3 provides an intermediate response. These interactions between planting method, nutrient supply, and genotype underline the need for tailored agronomic and genetic strategies to optimize sugar beet bolting behavior for specific production goals.
Effect of planting method on days to stalk formation (DSF), and stalk percentage
-
The days to stalk formation (DSF) was significantly influenced by the interaction of planting methods, fertilizer levels, and varieties (Fig. 2, Supplementary Table S3). Among the planting methods, E2 consistently led to the longest duration for stalk initiation, followed by E3, whereas E1 resulted in the shortest DSF across all genotypes and fertilizer levels. This pattern was statistically significant (p < 0.05), indicating that planting methods have a dominant effect on stalk development timing. Across fertilizer treatments, the higher NPK dose (F2) generally promoted slightly earlier stalk formation compared to F1, particularly under E1, where DSF decreased by an average of 2–5 d. However, this effect was less pronounced under E2, suggesting that nutrient supply can enhance developmental rate, but the planting method exerts greater control over vegetative phase duration. Genotypic differences were also evident. LS 6 and IISR COMP 1 exhibited relatively early stalk formation, especially under E1 and E3, while LKC 2010 consistently displayed delayed DSF, irrespective of treatment. These results imply that LS 6 has a strong adaptability for quicker stalk initiation under favorable conditions, making it suitable for early-maturing production systems. The stalk percentage varied significantly (p < 0.05) across planting methods, fertilizer levels, and genotypes (Fig. 3). The E2 planting method produced the highest stalk percentages across most germplasm, with LKC 2010 and LKC 2007 performing particularly well. E1 showed moderate stalk percentages, whereas E3 recorded the lowest values, suggesting that restricted root space in this method adversely affects stalk formation. Notably, the positive effect of E2 on stalk percentage was consistent under both fertilizer levels, highlighting its potential for maximizing stalk productivity. Overall, the findings demonstrate that while fertilizer dose has a secondary but supportive role, the planting method is the primary determinant of stalk initiation and development, with E2 being most favorable for maximizing stalk formation, and LS 6 showing superior adaptability for early stalk emergence.
Effect of planting method on days to flowering (DF), and flowering percentage
-
The analysis revealed significant effects of environmental conditions (E1–E3), fertilizer treatments (F1, F2), and their interaction on days to flowering (DF) and flowering percentage across the evaluated sugar beet germplasm (p < 0.05) (Supplementary Table S4). Across environments, E1 facilitated the earliest flowering, followed by E3, while E2 significantly delayed flowering in most genotypes. The fertilizer treatment F2 slightly accelerated flowering compared to F1, though the difference was not statistically significant in several cases. Among genotypes, LKC 2010 and LKC 2007 consistently exhibited early flowering, whereas IISR COMP 1 showed a prolonged flowering period, particularly under E2, indicating its sensitivity to delayed environmental conditions. Overall, E2 conditions resulted in the highest DF values, reflecting delayed floral initiation, whereas E1 promoted faster transition to flowering, suggesting a favorable combination of temperature and photoperiod for floral induction. In contrast, E3 displayed intermediate responses, highlighting the environmental influence on reproductive phase progression.
Flowering percentage also varied significantly among environments and fertilizer regimes (p < 0.05). E2 recorded the highest flowering percentages, notably in LS 6, suggesting that this environment supports better root establishment and nutrient utilization. E1 produced moderate flowering, whereas E3 exhibited the lowest flowering percentages, indicating suboptimal conditions for reproductive development. Comparative analysis of fertilizer treatments revealed that the higher P and K levels in F2 did not consistently improve flowering percentage. While some genotypes (e.g., LS 6) responded positively under F2, others (e.g., LKC 2010, LKC 2020) showed a decline, implying genotype-specific nutrient responses. The results highlight that environmental conditions exerted the strongest influence on flowering behavior, followed by genotype and fertilizer treatment. The findings underscore the potential of E1 conditions and early-flowering genotypes (LKC 2010, LKC 2007) for achieving optimized flowering in sugar beet, while emphasizing the need for genotype-specific nutrient management strategies.
Effect of planting method and fertilizer doses on days to seed harvesting (DSH)
-
Significant variations in DSH were observed across planting styles, germplasm, and fertilizer levels (Table 3). Among the planting styles, E1 consistently recorded the lowest DSH values, indicating faster seed maturation, whereas E2 significantly prolonged the harvesting period (p < 0.05), likely due to extended vegetative growth (Supplementary Table S5). The E3 planting style showed intermediate values, suggesting a balanced growth-to-reproduction transition. Across fertilizer levels, the F2 treatment slightly reduced DSH compared to F1 in most cases, though the effect was statistically marginal in E2. This indicates that while higher fertilizer input can promote earlier maturity, its impact is context-dependent and less pronounced under conditions that favor prolonged vegetative growth. Among germplasm, LKC 2006 and LS 6 matured significantly earlier, whereas IISR COMP 1 and LKC 2010 exhibited delayed harvesting, particularly under E2 conditions (p < 0.05). Notably, IISR COMP 1 consistently showed the longest DSH across environments, highlighting its relatively slow maturation pattern. Overall, the interaction between planting style and germplasm was significant (p < 0.05), demonstrating that genotype performance varied with planting configuration. The trend suggests that E1 planting coupled with moderate fertilization (F2) can effectively optimize seed maturation in sugar beet, offering a practical strategy to balance productivity and timely harvesting.
Table 3. Mean ± standard deviation of agro-morphological traits under different planting methods and NPK fertilizer levels.
Planting style Germplasm Stalk percentage
(mean ± SD)Flowering percentage
(mean ± SD)Germination percentage
(mean ± SD)Test weight
(mean ± SD with rank)F1 F2 F1 F2 F1 F2 F1 F2 E1 IISR COMP 1 69.17 ± 3.75 65.00 ± 5.00 61.67 ± 8.75 58.33 ± 7.64 90.95 ± 2.88 85.98 ± 3.29 2.46 ± 0.03 3.18 ± 0.02 LS 6 80.00 ± 6.46 75.83 ± 5.15 71.67 ± 3.75 70.83 ± 2.89 92.46 ± 2.29 88.21 ± 2.46 2.33 ± 0.03 3.14 ± 0.06 LKC 2006 86.67 ± 1.44 78.33 ± 2.89 85.00 ± 5.00 70.83 ± 4.96 96.37 ± 2.34 91.57 ± 2.18 2.56 ± 0.04 3.24 ± 0.08 LKC 2007 85.83 ± 5.15 71.67 ± 2.89 80.00 ±6.45 69.17 ± 5.27 90.91 ± 1.76 85.35 ± 2.12 2.44 ± 0.03 3.04 ± 0.04 LKC 2010 74.17 ± 3.75 68.33 ± 2.89 87.50 ± 6.45 62.50 ± 4.79 89.38 ± 0.86 84.63 ± 2.03 2.35 ± 0.02 2.74 ± 0.34 LKC 2020 71.67 ± 3.75 65.83 ± 5.15 85.00 ± 2.89 64.17 ± 5.85 92.16 ± 2.11 88.28 ± 1.99 2.51 ± 0.07 3.23 ± 0.07 E2 IISR COMP 1 82.50 ± 4.33 70.00 ± 2.50 87.50 ± 2.50 65.00 ± 4.33 92.51 ± 1.99 88.54 ± 2.06 2.70 ± 0.18 3.10 ± 0.07 LS 6 90.83 ± 3.75 83.33 ± 2.89 95.83 ± 1.44 75.83 ± 1.44 93.28 ± 0.99 87.85 ± 1.42 2.52 ± 0.08 3.18 ± 0.02 LKC 2006 86.67 ± 5.00 81.67 ± 3.89 90.00 ± 2.89 73.33 ± 8.06 95.17 ± 0.91 91.84 ± 1.34 4.31 ± 0.09 4.84 ± 0.05 LKC 2007 84.17 ± 8.06 75.83 ± 7.5 82.50 ± 6.45 69.17 ± 7.50 91.74 ± 1.04 88.82 ± 1.01 4.35 ± 0.03 3.50 ± 0.34 LKC 2010 84.17 ±10.93 74.17 ± 16.07 80.83 ± 8.96 66.67 ± 17.56 91.51 ± 1.75 87.25 ± 1.87 4.08 ± 0.08 3.44 ± 0.06 LKC 2020 90.00 ± 2.89 81.67 ± 6.29 84.17 ± 3.75 74.17 ± 3.75 94.56 ± 0.88 90.87 ± 1.55 4.15 ± 0.04 3.55 ± 0.16 E3 IISR COMP 1 56.67 ± 7.64 61.67 ± 7.64 55.83 ± 3.75 51.67 ± 6.29 88.43 ± 1.45 86.35 ± 1.93 1.08 ± 0.02 1.53 ± 0.04 LS 6 55.83 ± 5.15 53.33 ± 5.00 54.17 ± 5.15 45.83 ± 3.75 95.22 ± 1.11 92.87 ± 1.57 1.35 ± 0.04 1.86 ± 0.09 LKC 2006 59.17 ± 3.89 55.83 ± 3.89 60.00 ± 6.29 53.33 ± 1.44 93.64 ± 0.99 89.74 ± 1.36 1.77 ± 0.04 2.12 ± 0.08 LKC 2007 62.50 ± 2.50 60.00 ± 6.29 57.50 ± 5.00 59.17 ± 4.79 90.32 ± 1.28 86.44 ± 1.45 1.46 ± 0.09 1.58 ± 0.11 LKC 2010 53.33 ± 3.79 50.00 ± 5.00 50.00 ± 4.33 46.67 ± 5.77 90.33 ± 1.45 86.18 ± 1.74 1.14 ± 0.05 1.29 ± 0.07 LKC 2020 65.00 ± 4.33 57.50 ± 5.00 61.67 ± 3.75 55.00 ± 5.00 91.63 ± 1.17 87.76 ± 1.68 1.15 ± 0.03 1.31 ± 0.06 * F1-120:60:60; F2-120:80:80; bold data denotes the highest values in each column (F1 and F2 separately for each trait), to highlight the most significant data. Effect of planting style, and fertilizer dose on germination percentage
-
Germination percentage varied significantly across planting styles, germplasm, and fertilizer levels (Table 3). Overall, E1 exhibited the highest germination rates, followed by E2, while E3 recorded comparatively lower values (p < 0.05). The F1 treatment consistently outperformed F2, indicating that higher fertilizer levels slightly reduced germination across most germplasm. Among the genotypes, LKC 2006 and LS 6 showed superior germination performance, whereas LKC 2010 and IISR COMP 1 recorded relatively low rates. The decline in germination under F2 was particularly evident in IISR COMP 1, suggesting its greater sensitivity to fertilizer concentration. These findings demonstrate that planting style and genotype interactions significantly influenced germination efficiency, with E1 and moderate fertilization (F1) providing optimal conditions for early establishment and uniform seedling emergence.
Effect of planting method and fertilizer doses on yield plant−1 and test weight
-
Significant differences were observed in yield and test weight among sugar beet varieties, planting methods, and fertilizer levels (Table 3, Supplementary Table S6). Across treatments, the F2 fertilizer dose consistently produced higher yields than F1 (p < 0.05), confirming that enhanced nutrient availability positively influenced growth and productivity. Among the genotypes, LKC 2006 and LKC 2007 demonstrated superior yield performance, indicating strong adaptability, while IISR COMP 1 maintained stable yields across environments, suggesting broader environmental tolerance. Test weight also increased significantly under F2 across most varieties, reflecting the positive role of higher P and K levels in seed filling and nutrient accumulation. The E2 planting method resulted in the highest test weight, followed by E1 and E3 (p < 0.05). The variation in seed weight among genotypes was largely attributed to differences in seed morphology, particularly the presence of multi-germ seed balls containing multiple embryos. Overall, the results suggest that combining the E2 planting method with F2 fertilization optimizes both yield and seed quality in sugar beet cultivation.
-
In India, variable agro-climatic zones, fluctuating winter intensity, and heterogeneous soil types necessitate region-specific strategies for sugar beet seed production[4,18]. As a temperate biennial crop, sugar beet requires vernalization to transition from vegetative to reproductive growth[19]. This requirement poses a challenge in Indian plains, where suboptimal winter temperatures hinder flowering induction[4,20,21]. Optimizing planting methods is therefore crucial for sugar beet seed production, as these directly influence crop establishment, flowering synchronization, and seed quality[3]. Among the tested approaches, E1 portrays precise selection and strategic transplanting, allowing only sturdy seedlings with desirable root characteristics (6–14 cm in length) to be propagated for seed production. This controlled approach enhances uniformity, optimizes plant-to-plant spacing, and ensures better field establishment at the outpost location. The transplantation shock is minimized with leaf and petiole trimming, which results in a healthy seed set and good seed yield[2,21]. While E1 is labour intensive and expensive, this precise technique requires meticulous planning and efficient resource use, but it supports stable genetic advancement and sustainable propagation[2,3]. On the contrary, Miandoab et al.[22] noted that the transplanting process can also include early sugar beet seedling establishment using controlled conditions in a greenhouse, especially where direct field sowing is hampered by adverse environmental conditions. In such systems, greenhouse or nursery-grown transplants not only allow early establishment but also increase germination and seedling vigour by protecting the crop from compromising external conditions[23]. Additionally, maintaining favourable and controlled environmental conditions significantly reduces the incidence of bolting in sugar beet by preventing premature exposure to low temperatures during early spring, which is a primary trigger for vernalization-induced bolting and subsequent yield losses[24]. From a sustainability standpoint, the transplanting method also aids in better water-use efficiency, as shown by Khozaei et al.[25], who indicated a 24% decrease in water use and evapotranspiration, and an increase in root yield of 7.7%. The E2 method depicts a traditional, low-intervention methodology where, in the same field, sugar beet is grown from seed to seed[5]. In this method, there is uninterrupted root development, nutrient accumulation is better, and good seed vigour is expected for the commercial production of seeds. The E3 method offers optimal cold treatment for transplanted roots during winter, preparing them for vernalization and flower induction, especially when planting is delayed due to snowfall.
Bolting dynamics are critical in defining seed yield and sugar content[26]. In this study, E1 induced the earliest bolting, suggesting that steckling-induced vernalization successfully bypasses the limitations of mild winters. This is consistent with the concept that early sowing and extended low temperatures, known as vernalization (ranging from 0 to +10 °C for 1 to 6 weeks), trigger bolting in sugar beet, especially in lowland regions, when cotyledons and true leaves emerge[27]. Furthermore, pre-vernalized mature roots are more responsive to long-day photoperiods, thus accelerating floral initiation[28]. Conversely, E2 delayed bolting due to gradual natural vernalization, where plants attain physiological maturity before responding to long-day signals. This aligns with earlier findings that prolonged low-temperature exposure delays reproductive induction in biennial crops[24,26]. E3 exhibited intermediate bolting, where pit storage moderated the temperature gradient, ensuring controlled vernalization[29]. Such temperature-buffering prevents premature bolting while preserving the cold requirement for reproductive transition. Molecularly, this may be linked to differential expression of GIGANTEA (GI) and FLOWERING LOCUS T (FT) homologs, key regulators integrating vernalization and photoperiod cues in sugar beet[29].
Fertilizer dose exerted a moderate yet consistent influence on bolting and flowering. The F2 treatment (enhanced P and K) slightly reduced bolting duration, particularly under E1 and E3, suggesting improved physiological readiness for reproduction. P is a key component of plant energy metabolism, supporting ATP synthesis and carbon fixation during photosynthesis, thereby sustaining the energy supply required for plant growth and development[30], while K enhances osmotic regulation and enzyme activation critical for assimilate translocation during reproductive initiation[31]. Thus, the combined action of P and K likely enhanced root vigor and carbohydrate mobilization, accelerating floral initiation. In contrast, E2's slower nutrient uptake through seed-to-seed growth may have delayed hormonal signaling, including gibberellin activation, which is necessary for bolting induction[27].
The observed differences in flowering timing among genotypes underscore their genetic variability in vernalization sensitivity and photoperiod responsiveness. Early-flowering genotypes such as LKC 2010 and LKC 2007 likely possess shorter juvenile phases or heightened sensitivity to photothermal cues, making them suitable for breeding programs targeting rapid seed multiplication. Conversely, delayed flowering in IISR COMP 1 indicates a prolonged vegetative phase, reflecting genotype-dependent variation in flowering and bolting responses, consistent with earlier reports demonstrating pronounced differences among sugar beet cultivars in bolting behaviour when grown under contrasting climatic conditions[32,33]. These findings indicate that combining physiological screening with region-specific vernalization management could streamline selection for early-flowering, high-yielding types[1].
Seed harvesting trends followed the sequence E1 < E3 < E2, reflecting direct links between bolting onset, flowering time, and seed maturation. Early flowering in E1 compressed the crop cycle, favoring rapid seed production, whereas the extended vegetative phase in E2 delayed maturity. The slight DSH reduction under F2 may be attributed to phosphorus-induced enhancement of reproductive metabolism and seed filling[34]. The synchronization of flowering in E1 also facilitated more efficient pollination, a key determinant of uniform seed set[35]. In contrast, the seed-to-seed E2 method occasionally produced multiple sprouts per seed ball, complicating selection and synchronization.
Germination percentage was notably higher under E1, confirming that controlled transplanting enhances seed physiological quality. The vigorous early growth and nutrient reserves in E1 likely improved endosperm development and embryo viability. Slightly reduced germination under F2 may result from excessive nutrient accumulation, causing an osmotic or hormonal imbalance during seed maturation, leading to lower vigor and non-synchronous pollination[36]. This supports the hypothesis that moderate fertilization ensures optimal physiological maturity, while nutrient excess can stress reproductive tissues.
Flowering percentage showed strong dependency on planting method and genotype interaction. The E2 method consistently yielded higher flowering percentages, reflecting uninterrupted growth and stable nutrient uptake. However, transplant-induced stress in E1 led to moderate flowering levels, while E3's prolonged cold exposure and reduced aeration suppressed floral differentiation. Fertilizer response was non-linear; while increased P and K levels in F2 supported metabolic activity and nutrient balance, further increases did not proportionally enhance flowering, indicating a threshold beyond which additional nutrient supply offers limited reproductive benefit[37]. Thus, maintaining a nutrient equilibrium rather than merely increasing input is crucial for reproductive efficiency.
Yield performance further substantiated the physiological advantage of E2. Continuous root anchorage enabled better resource allocation toward seed development, consistent with observations that overwintered beets benefit from stable, low temperatures for sustained growth. The positive yield response to F2 suggests that P and K synergistically enhance sucrose translocation and assimilate partitioning, supporting reproductive sink strength. In contrast, E1 exhibited moderate yields due to transient stress during steckling preparation, yet its shorter cycle makes it valuable for speed breeding and early-generation advancement. The E3 method, though least productive, remains important under niche agro-climatic conditions, where controlled cold exposure can ensure successful vernalization when field temperatures are inadequate.
Overall, these findings reinforce that integrated management of planting strategy, genotype selection, and balanced fertilization is essential for optimizing seed production in sugar beet under Indian conditions. By linking phenological responses with physiological mechanisms and genotype-specific adaptability, this study provides a framework for sustainable and region-specific sugar beet seed production systems capable of supporting both domestic and bioethanol industries.
-
Sugar beet seed quality is largely influenced by the growing conditions and production techniques used. This study found that the steckling method ensures uniform growth, better establishment, and higher seed yield while conserving rare genotypes, reducing risk, and accelerating seed production, breeding, and generation advancement in sugar beet. This method not only enhances seed quality and yield but also supports the conservation, multiplication, and breeding of sugar beet germplasm, thereby enabling generational advancement in a shorter period. As an indigenous seed production technology developed by ICAR–ISRI, Lucknow, it holds strong potential for large-scale commercial seed production in identified high-altitude regions of India. By reducing seed production time by 5–6 months and minimizing dependence on imported seeds, this approach contributes directly to India's agricultural self-sufficiency and bio-ethanol production goals by ensuring a consistent domestic supply of high-quality sugar beet seed—a key feedstock for ethanol and sugar industries. Furthermore, the pit method emerged as a valuable complementary technique, ensuring bolting and flowering with quality seed production, particularly beneficial for late or poorly developed stecklings or genotypes with lower seed yield potential. This method supports germplasm conservation and can be integrated into commercial seed production systems. Additionally, the split fertilizer dosage (120:80:80 NPK) optimized the balance between vegetative growth and reproductive output, showing promise for adoption in commercial-scale seed production. Economically, adopting an E1 + F2 combination can reduce dependence on imported seed by 25%–30%, shorten production cycles by up to 6 months, and improve cost-efficiency by 20%–25% through optimized fertilizer utilization and better resource management. Localized seed production also creates employment in hilly and transitional regions while strengthening domestic value chains for bioethanol and sugar industries. Specifically, the E1 + F2 combination (steckling method with split fertilizer dosage of 120:80:80 NPK) is recommended for high-altitude temperate and sub-temperate zones such as the Himachal Pradesh, Kashmir Valley, and upper Uttarakhand regions, where natural vernalization facilitates optimal bolting and flowering. These conditions allow full expression of reproductive potential and maximize seed yield and quality. Future research should focus on refining these techniques under varying agro-climatic conditions, integrating precision agricultural tools and controlled-environment facilities to further enhance seed productivity, genetic stability, and uniformity. These advances will strengthen India's capacity to establish a sustainable, self-reliant sugar beet seed system aligned with its bio-economy and renewable energy objectives.
-
The authors confirm their contributions to the paper as follows: conceptualization: Mall AK, Misra V; writing original draft: Mall AK, Misra V, Tiwari RK; review and editing: Mall AK, Misra V, Tiwari RK, Srivastava S, Shiv A, Viswanathan R, Singh D; statistical analysis: Tiwari RK. All authors reviewed the results and approved the final version of the manuscript.
-
All data generated or analyzed during this study are included in this published article and its supplementary information files.
-
The authors received no financial support for the research, authorship, and/or publication of this article.
-
The authors declare that they have no conflict of interest.
-
# Authors contributed equally: Ashutosh Kumar Mall, Varucha Misra
- Supplementary Table S1 Two-way ANOVA analysis for days to bolting under fertilizer treatments (Alpha 0.05).
- Supplementary Table S2 Two-way ANOVA analysis for bolting % under fertilizer treatments (Alpha 0.05).
- Supplementary Table S3 Two-way ANOVA analysis for days of stalk formation under fertilizer treatments (Alpha 0.05).
- Supplementary Table S4 Two-way ANOVA analysis for days to flowering under fertilizer treatments (Alpha 0.05).
- Supplementary Table S5 Two-way ANOVA analysis for days to harvesting under fertilizer treatments (Alpha 0.05).
- Supplementary Table S6 Two-way ANOVA analysis for yield per plant under fertilizer treatments (Alpha 0.05).
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Mall AK, Misra V, Tiwari RK, Srivastava S, Shiv A, et al. 2026. Fertilizer dosage regimen and steckling transplanting for accelerated sugar beet seed production. Technology in Agronomy 6: e005 doi: 10.48130/tia-0026-0001
Fertilizer dosage regimen and steckling transplanting for accelerated sugar beet seed production
- Received: 02 September 2025
- Revised: 26 October 2025
- Accepted: 04 November 2025
- Published online: 09 April 2026
Abstract: Sugar beet seed production in India remains restricted due to its high-altitude vernalization requirement. To meet the growing need for high-quality seeds for bioethanol production, this study aimed to investigate how different fertilizers and planting methods affected the yield and quality of seeds in six ICAR–ISRI-developed genotypes over two years. Three planting methods: Steckling (E1), Direct Seeding (E2), and the Pit Method (E3), were tested along with two levels of fertilizer doses, viz., conventional (120:60:60 NPK, F1), and optimized (120:80:80 NPK, F2). Unlike previous international studies, this investigation represents the integrated planting methods and fertilizer regimes specifically optimized for Indian subtropical agro-climatic conditions, where vernalization is a major constraint. The optimized regime (F2) increased early bolting (> 90%) and flowering (60%–80%), harvest seed maturity by 1–5 d, and increased test weight by 8%–12% compared to F1. E1 led to the earliest seed harvest (184–200 d), E3 was in the middle (234–252 d), and E2 took longer to mature (> 400 d), but had higher percentages of bolting and flowering. F2 improved seed yield across genotypes by 15%–18% when compared to F1. The largest increase in seed yield was observed in the F2 regime by LKC 2006, with increases of 26.56%, 12.29%, and 19.77% in E1, E2, and E3, respectively. This study establishes a region-specific framework for sugar beet seed production under suboptimal vernalization conditions, demonstrating that combining the Steckling or Pit methods with the optimized 120:80:80 NPK regime can effectively enhance reproductive success and seed productivity in subtropical India. Overall, seed yield, test weight, and germination were significantly increased by combining the Steckling or Pit methods with the optimal fertilizer regime (120:80:80 NPK), offering a novel, climate-adapted strategy toward national seed self-sufficiency, and sustainable sugar beet-based bioethanol production.
-
Key words:
- Steckling method /
- Fertiliser dosage /
- Sugar beet /
- Seed /
- Pit method















