-
Cowpea (Vigna unguiculata L.) is a significant annual legume crop used for the dual purpose of pulses and fresh vegetables[1]. Originally from central Africa, it is extensively cultivated in the tropics and sub-tropical regions[2]. In Nepal, it occupies a considerable cultivation area in Terai and inner-Terai regions[3]. Farmers cultivate indigenous and improved varieties of cowpea for grain production[4]. However, despite the increasing trend in cowpea production, its productivity remains lower compared to other legumes[5]. The lower productivity can be attributed to various physical, biotic, and socio-economic constraints[3,6]. Furthermore, there is a lack of efficient integrated nutrient management (INM) practices specifically tailored for legume production in Nepal[3]. Currently, farmers heavily rely on the application of chemical fertilizer, leading to increasing trends in their usage[5,7]. However, the excessive use of chemical fertilizer beyond recommended rates not only results in soil degradation, air and water pollution, reduced soil fertility, and crop production[7,8] but also lowers the inherent nitrogen-fixing process of legumes[9]. To address these long-term effects and promote sustainable agriculture with improved production, the integration of bio-fertilizers into the farming system becomes crucial[10].
Cowpea, as a leguminous crop, possesses the remarkable ability to fix atmospheric nitrogen, which is crucial for its early vegetative growth and development[11,12]. Prior to the initiation of nitrogen fixation in these legumes, especially those growing on poor organic matter soils, nitrogen fertilizer is typically provided during planting. When applying nitrogen, the rate must not exceed the advised dosage. The nitrogen fixation process is physically slowed down or halted by the legumes when too much nitrogen (N) is given[9].
The sustainability of farming has been advocated by organic agriculture[7]. It relies on the utilization of microbial-based fertilizers, commonly known as biofertilizers. Among them, Phosphate Solubilizing Bacteria (PSB), Arbuscular Mycorrhiza Fungi (AMF), potassium-solubilizing bacteria, and nitrogen-fixing bacteria have been extensively employed to enhance soil fertility[13−15]. In the case of grain legumes, biofertilizers such as rhizobium species and mycorrhiza are widely used[16]. Biofertilizers play a crucial role in plant metabolism and nutrient availability, facilitating nutrient uptake from the soil[17]. The symbiotic association of rhizobium species with legumes promotes biological nitrogen fixation, phosphate solubilization, and production of Indole-3 Acetic Acid (IAA), siderophores, and chitinase. These activities contribute to increased vegetative biomass and improved yield parameters in legumes[18,19]. Similarly, the incorporation of AMF into legume crops establishes a mutually beneficial symbiotic relationship. AMF enhances nutrient mineralization, increases the effective root area, and improves nutrient availability to crops, and in return, AMF obtains carbon from the crops[17,20].
Despite their nutrient richness, inorganic fertilizers give rise to a myriad of issues[7,21]. Biofertilizers, though less mobile than their inorganic counterparts, offer a plethora of benefits to the cultivated soil[22−24]. The necessity for environmentally safe agricultural practices, coupled with the scarcity and high cost of synthetic fertilizers, has prompted extensive research into organic-based alternatives, leading to their adoption by many farmers[7]. Nonetheless, much of the prevailing research has been unidirectional, focusing solely on either biofertilizers or inorganic fertilizers. The objective of this research was to investigate the effects of biofertilizers, inorganic fertilizers, and combined application of chemical fertilizers and biofertilizers on the growth attributes and yield parameters of different varieties of cowpea variety in Nawalparasi West, Nepal. Additionally, the study aimed to compare the performance of the Malepatan-1 variety with the Stickless variety in terms of growth parameters, yield attributes, and overall yield. The result holds immense value for both the practical and theoretical realms, as they provide invaluable recommendations to farmers on adopting sustainable and cost-effective practices that enhance yield while minimizing nutrient losses.
-
The study was conducted on a farmer's field in February 2022 at Ramgram-17, Nawalparasi West, Nepal. The study site is situated at a geographic position of 27°32' N latitude and 83°40' E longitude, with an elevation of 119 m above sea level. Nawalparasi West has a sub-tropical and humid climate.
Soil characteristics
-
Soil samples were taken from each replication by using a shovel from 0 to 15 cm depth. Then the sample was analyzed for soil properties as presented in Table 1. The soil was tested by the technical personnel from Soil and Fertilizer Testing Laboratory, Khajura, Banke. The total nitrogen was determined by Kjeldhal distillation method[25]; available phosphorous by Olsen's method[26], and available potassium by the ammonium acetate method. Organic matter was determined by the Walkley-Black method[27], pH (1:1 soil : water suspension) by Beckman Glass electrode pH meter and soil texture by the hydrometer method.
Table 1. Physico-chemical characteristics of soil in the experimental field.
S. N. Soil properties Values 1. Textural class Clay loam (Alluvial) 2. Chemical properties 2.1 pH 7.7 (Alkaline) 2.2 Nitrogen (%) 0.09 (Low) 2.3 Phosphorus, P2O5 (kg·ha−1) (Low) 2.4 Potash, K2O (kg·ha−1) (Low) 2.5 Organic matter (%) 1.95 (Low) Experimental details
-
The experiment was designed using a two-factor Randomized Complete Block Design (RCBD) with 12 treatments replicated three times. The factors considered were variety and fertilizer. Variety consisted of two types; Malepatan-1 (V1) and Stickless (V2). The fertilizer factor included no fertilizer treatment (F0), mycorrhiza (F1), rhizobium (F2 = 100 g per 5 kg of seed), the recommended dose of fertilizer (F3 = 20:40:20 kg NPK ha−1), mycorrhiza + rhizobium (F4), and mycorrhiza + rhizobium + RDF (F5). Cowpea varieties and fertilizers were obtained from Dawadi biofertilizer center, Buddha chowk, Chitwan (Nepal).
Each experimental plot had an area of 2.4 m × 1.5 m, accommodating four rows. The rows were spaced 60 cm apart, with each row containing five hills spaced 30 cm apart. Two seeds were sown per hill. The inter-plot distance was set at 0.3 m, while the distance between replications was maintained at 0.75 m. The field was plowed 15 d before seed sowing by using a rotavator to achieve a good soil tilth. This was followed by shallow ploughing, and planking to level the land. After leveling, clods were broken, and weeds and stubbles of the previous crop were removed.
The chemical fertilizers were supplied to the experimental fields as nitrogen, phosphorus, and potassium through urea, single super phosphate (SSP), and muriate of potash (MOP), respectively, at the rate of 20 kg N, 40 kg P and 20 kg K ha−1 i.e. 20:40:20 kg NPK ha−1. The recommended amounts of urea, SSP, and MOP were calculated per area basis and weighed separately for each treatment. The total nitrogen dose was split into two equal doses. The full dose of phosphorus and potassium and half of the nitrogen dose was applied as a basal dose one day prior to sowing. The second dose of nitrogen was applied as a top dress in the third week of sowing in March 2022. Gap filling was conducted two weeks after sowing to maintain the plant population. Irrigation was provided at weekly intervals until maturity. Weeding was performed manually, with the first round at 15 d after sowing (DAS), followed by the second and third rounds at 30 DAS and 45 DAS, respectively. Pesticides, specifically, Cypermethrin at 1 ml·L−1 and Emamectin benzoate at 5 ml per 15 L, were used during the research period to control pod borers.
Seed treatments
-
Commercial rhizobia inoculants are available to farmers in various forms, such as solid, liquid, and freeze-dried formulations[28]. When selecting rhizobia inoculants, it is important to consider specific host legumes and ecological settings to ensure superior nodulation and nitrogen fixation efficiency compared to natural rhizobia populations[29]. Seeds were inoculated with Bradirhizobium sp. at 100 g per 5 kg of seed. To prepare the inoculant, 100 g of jaggary was initially dissolved in 250 ml of water to create jaggery slurry. The slurry was then boiled and cooled to room temperature. The recommended dose of Bradirhizobium sp. was added to the paste, and the required quantity of seeds was thoroughly mixed with the inoculant to achieve a uniform coating. The coated seeds were shade dried for 20 min prior to sowing, as outlined by Pandey et al.[30].
For mycorrhiza treatment, mycorrhiza AVESTA (all endo mycorrhiza including Glomus sp. and Acaulospora sp. coated on carriers through Agrinos Gel Technology along with nutrients, AVESTA: 3,000−3,500 spores g−1 at 4 kg 10 katha−1 (1 katha = 0.00138 ha) was incorporated in soil at 14 DAS. Each plot was inoculated with 3.5 g plot−1 inoculate for mycorrhiza treatment; with inoculate placed 4 cm below the seeds. This was immediately followed by light irrigation.
Data collection and observation
Plant height (cm) and number of branches
-
Six plants were randomly selected excluding border crops and from which parameters were observed and measured. The plant height was measured with the help of a scale and measuring tape from the base of the plant at the ground surface to the tip of the highest visible part at 30, 45 and 60 DAS. All the pod bearing as well as non-bearing branches were observed at 30, 45 and 60 DAS for number of branches.
Number of pods plant−1, pod length and seed weight plant−1
-
Number of pods plant−1, pod length and seed weight plant−1 were obtained from randomly selected six plants excluding border plants and finally, after averaging, the total number of pods plant−1, pod length and total weight of seed plant−1 in gram was obtained.
Yield plot−1, yield in kg hectare−1 and thousand grain weight
-
Sun-dried seeds of each plot were weighted to obtain yield per plot in grams. The seed yield per plot for each plot was converted into hectare to obtain the total yield in kg·ha−1. The weight of sun-dried thousands seed grain samples were weighted in grams.
Data analysis
-
The data recorded on different parameters were tabulated in Microsoft Excel 2010. Analysis of Variance (ANOVA) was performed using the "agricolae" package in R-Studio (R version 4.2.0). To compare means, Duncan's Multiple Range Test (DMRT) was applied to the analyzed data at a significance level of 5%.
-
Significant variation was observed in height of the plant of cowpea at different stages of growth as influenced by different varieties (Table 2). It was noted that the tallest plant height (42.06 cm) was recorded from Stickless at 30 DAS whereas the tallest plant height was recorded from Malepatan-1 at 77.56 and 125.73 cm at 45 and 60 DAS respectively. This variation in plant height between varieties might be due to individual varietal genetic superiority.
Table 2. Effect of different varieties and fertilizers on the plant height of cowpea.
Treatment Plant height (cm) 30 DAS 45 DAS 60 DAS Variety Malepatan-1 (V1) 40.06b 77.56a 125.73a Stickless (V2) 42.72a 64.37b 86.19b LSD (0.05) 1.26 1.26 7.30 SEM (±) 0.21 0.44 1.24 F-test *** *** *** CV (%) 4.42 2.57 9.96 Fertilizer Control 37.06d 60.16e 86.46c Mycorrhiza 40.58c 68.60d 101.79bc Rhizobium 40.99bc 67.89cd 98.40b RDF 41.45bc 72.97bc 109.86b Mycorrhiza + Rhizobium 43.15ab 74.27b 111.38b Mycorrhiza + Rhizobium + RDF 45.10a 81.90a 127.85a LSD (0.05) 2.19 4.56 12.64 SEM (±) 0.12 0.25 0.71 F-test *** *** *** CV (%) 4.42 5.37 9.96 Grand mean 41.39 70.96 105.96 Data in columns with the same letters in DMRT are not significantly different (p = 0.05), SEM = Standard Errors of Means, CV = Coefficient of Variation, LSD = Least Significant Difference, *** = significant at p < 0.001. The plant height of cowpea was influenced significantly at different growth stages influenced by different fertilizers. The results revealed that the tallest plant height was obtained from F5 at all the stages of growth. At 30 DAS, the tallest plant height (45.10 cm) was achieved from F5 which was at par with F4 (43.15 cm). Similarly, the shortest plant height (37.06 cm) was achieved from F0. The plant height obtained from F1 (40.58 cm), F2 (40.99 cm) and F3 (41.45 cm) were statistically similar. At 45 DAS, the tallest plant height (81.90 cm) was achieved which was significantly superior over all the other treatments viz; F0 (60.16 cm), F1 (68.60 cm), F2 (67.89 cm), F3 (72.97 cm) and F4 (74.27 cm) whereas the shortest plant height (60.16 cm) was achieved from F0. Similarly, at 60 DAS the tallest plant height (127.85 cm) was achieved which was significantly superior over all the other treatments. The height of the plant was not influenced significantly due to the effect of the interaction between variety and fertilizer at any stage of growth.
Number of branches
-
Significant variation was observed in the number of branches of cowpea at different stages of growth as influenced by different varieties (Table 3). It was noted that the higher number of branch (2.71) was recorded from Malepatan-1 at 30 DAS whereas the higher number of branches were recorded from Stickless, 4.49 and 6.36 at 45 DAS and 60 DAS respectively.
Table 3. Effect of different varieties and fertilizers on the branches per plant of cowpea.
Treatment Number of branches plant−1 30 DAS 45 DAS 60 DAS Variety Malepatan-1 2.71a 4.09b 5.39b Stickless 2.03b 4.49a 6.36a LSD (0.05) 0.26 0.34 0.45 SEm (±) 0.044 0.059 0.078 F-test *** * *** CV (%) 15.90 11.74 11.12 Fertilizer Control 2.03c 3.72b 5.25c Mycorrhiza 2.13c 4.13b 5.34bc Rhizobium 2.28bc 3.80b 5.61bc RDF 1.94c 4.13b 5.94bc Mycorrhiza + Rhizobium 2.69b 4.33b 6.14b Mycorrhiza + Rhizobium + RDF 3.16a 5.63a 6.97a LSD (0.05) 0.45 0.604 0.78 SEM (±) 0.025 0.034 0.045 F-test *** *** ** CV (%) 15.90 11.74 11.12 Grand mean 2.37 4.29 5.87 Data in columns with the same letters in DMRT are not significantly different (p = 0.05), SEM = Standard Errors of Means, CV = Coefficient of Variation, LSD = Least Significant Difference, * = significant at p < 0.05, ** = significant at p < 0.01, *** = significant at p<0.001. The number of branches per plant of cowpea was influenced significantly at different growth stages as influenced by different fertilizers (Table 3). The results revealed that higher numbers of branches were recorded from F5 at all the stages of growth. At 30 DAS, a higher number of a branch (3.16) was achieved from F5 which was significantly superior over all the other treatments viz; F0 (2.03), F1 (2.13), F2 (2.28), F3 (1.94) and F4 (2.69) whereas the lower number of a branch (1.94) was achieved from F3. At 45 DAS, a higher number of the branch (5.63) was achieved from F5 which was significantly superior over all the other treatments. Similarly, at 60 DAS, a higher number of a branch (6.97) was achieved from F5, which was significantly superior over all the other treatments whereas the lower number of a branch (5.25) was achieved from F0 but was at par with F1 (5.34), F2 (5.61), and F3 (5.94). The number of branches per plant was not influenced significantly due to the effect of interaction between variety and fertilizer at any stage of growth.
Yield parameters
Number of pods per plant
-
Significant variation was observed in the number of pods per plant of cowpea as influenced by different varieties (Table 4). It was noted that the higher number of pods plant−1 (31.34) was recorded from Stickless whereas the lower number of pods plant−1 (27.50) was recorded from Malepatan-1.
Table 4. Effect of different varieties and fertilizers on the pods per plant, seed weight and pod length of cowpea.
Treatment Pods plant-1 Seed weight plant−1 (g) Pod length (cm) Variety Malepatan-1 27.50b 72.29a 25.39 Stickless 31.34a 66.08b 26.31 LSD (0.05) 2.62 4.85 SEM (±) 0.44 0.82 F-test ** * NS CV (%) 12.92 10.15 Fertilizer Control 24.61c 47.92d 23.47 Mycorrhiza 28.47bc 62.21c 27.01 Rhizobium 27.86bc 57.01c 25.25 RDF 26.52c 57.65c 25.81 Mycorrhiza + Rhizobium 31.86b 88.98b 25.71 Mycorrhiza + Rhizobium + RDF 37.19a 101.35a 27.84 LSD (0.05) 4.55 8.41 SEM (±) 0.25 0.47 F-test *** *** NS CV (%) 12.92 10.15 Grand mean 29.42 69.19 Data in columns with the same letters in DMRT are not significantly different (p = 0.05), SEM = Standard Errors of Means, CV = Coefficient of Variation, LSD = Least Significant Difference, * = significant at p < 0.05, ** = significant at p < 0.01, *** = significant at p < 0.001, NS = Non-significant. Number of pods per plant of cowpea were influenced significantly due to fertilizer (Table 4). The results revealed that the higher number of pods plant−1 (37.19) was achieved from F5 which was significantly superior over all the other treatments. Lowest number of pods plant−1 (24.61) were obtained from F0, which was statistically at par with treatments F1, F2, and F3. Similarly, number of pods plant−1 as obtained due to the effect of F4 were statistically similar with treatments F1, and F2. These findings are in accordance with the findings of Jayshree & Umesha[31], who demonstrated that there was significant increase in growth parameter viz., plant height, number of branches, number of nodules, plant dry weight, crop growth rate and yield attributing parameters viz., pods plant−1, seeds pod−1, 1000-seed weight, seed yield and biological yield were recorded with dual inoculation of Phosphate Solubilizing Bacteria and VAM along with optimum dose of inorganic fertilizers. The numbers of pods per plant were not influenced significantly due to the effect of interaction between variety and fertilizer.
Seed weight plant−1
-
Significant variation was observed on seed weight per plant of cowpea as influenced by different variety (Table 4). It was noted that the higher seed weight plant−1 (72.29 g) were recorded from Malepatan-1 where the lower seed weight plant−1 (66.08 g) were recorded from Stickless. The findings of the present study are in line with the findings of Kwaga[32], who demonstrated that the variations among varieties in terms of parameters like plant height, number of leaves, pod length, pods plant-1 and yield is due to the individual varietal superiority, due to genetic variation, and environmental variations.
Seed weight per plant of cowpea was influenced significantly due to fertilizer (Table 4). The results revealed that the higher seed weight per plant (101.35 g) was achieved from F5 which was significantly superior over all the other treatments. Lowest seed weight per plant (47.92 g) was obtained from F0. The seed weight per plant was not influenced significantly due to the effect of interaction between variety and fertilizer.
Pod length
-
The pod length was not influenced significantly due to the effect of fertilizer (Table 4). However, mathematically higher pod length was observed from Stickless (26.31 cm). The pod length was not influenced significantly due to the effect of fertilizer. However, mathematically higher pod length was observed from F5 (27.84 cm) while shortest pod length was obtained from F0 (23.47 cm). The pod length was not influenced significantly due to the effect of interaction between variety and fertilizer.
Yield plot−1
-
Significant variation was observed on yield plot−1 of cowpea as influenced by different variety (Table 5). It was noted that the highest seed yield plot−1 (1,273.24 g) was recorded from Malepatan-1 where the lowest yield plot−1 was recorded from Stickless. The findings of the present study are in line with the findings of Bhattarai et al.[4] and this variation might be due to the genetic make-up of the varieties, genotype x environment interaction and nutrient use efficiency of varieties[4].
Table 5. Effect of different varieties and fertilizers on the yield and test weight of cowpea.
Treatment Yield plot−1
(g)Yield hectare
(kg ha−1)1,000 seed weight
(g)Variety Malepatan-1 1,273.24a 3,536.83a 151.62a Stickless 1,173.21b 3,258.94b 125.46b LSD (0.05) 92.82 257.86 13.06 SEM (±) 15.82 43.96 2.23 F-test * * *** CV (%) 10.97 10.97 13.64 Fertilizer Control 933.27b 2,592.43b 116.38b Mycorrhiza 1,024.27b 2,845.21b 123.08b Rhizobium 1,061.67b 2,949.10b 123.03b RDF 1,067.35b 2,964.90b 124.71b Mycorrhiza + Rhizobium 1,555.69a 4,321.41a 167.19a Mycorrhiza + Rhizobium + RDF 1,697.12a 4,714.26a 176.83a LSD (0.05) 160.78 446.62 22.62 SEM (±) 9.13 25.38 1.28 F-test *** *** *** CV (%) 10.97 10.97 13.64 Grand mean 1,223.23 3,397.89 138.54 Data in columns with the same letters in DMRT are not significantly different (p = 0.05), SEM = Standard Errors of Means, CV = Coefficient of Variation, LSD = Least Significant Difference, * = significant at p < 0.05, *** = significant at p < 0.001. Seed yield per plot was influenced significantly due to fertilizer. The results revealed that the highest yield plot−1 (1,697.12 g) was achieved from F5 which was at par with fertilizer treatment F4 (1,555.69 g) but significantly superior over all other treatments viz; F0 (933.27 g), F1 (1,024.27 g), F2 (1,061.67 g), and F3 (1,067.35 g). Lowest yield plot−1 (933.27 g) was obtained from F0 which is statistically at par with treatments F1, F2 and F3. These findings are in accordance with the findings of Sharma & Jaga[33], who demonstrated that there was a significantly higher nutrient uptaking, higher nodule number and better quality of oil of soybean found due to the combined application of VAM + Rhizobium + PSB + 75% RDF. The higher yield plot−1 might be due to the effective absorption of mineral nutrients by mycorrhiza and biological nitrogen fixation by Rhizobium. Non-significant result was obtained for yield plot−1 due to interaction between variety and fertilizer.
Yield hectare−1
-
Significant variation was observed on yield ha−1 of cowpea as influenced by different variety (Table 5). It was noted that the highest yield ha−1 (3,536.83 kg) was recorded from Malepatan-1 where the lowest yield ha−1 (3,258.94 kg) was recorded from Stickless. The findings of the present study are in line with the findings of Bhattarai et al.[4], who demonstrated that there is a correlation between yield plot−1 and yield ha−1.
Yield ha−1 was significantly influenced due to fertilizer. The results revealed that the highest yield ha−1 (4,714.26 kg·ha−1) was achieved from F5 which was at par with F4 (4,321.41 kg·ha−1) but significantly superior over other treatments viz; F0 (2,592.43 kg·ha−1), F1 (2,845.21 kg·ha−1), F2 (2,949.10 kg·ha−1) and F3 (2,964.90 kg·ha−1). Lowest yield (2,592.43 kg·ha−1) was obtained from F0 which was statically at par with treatments F1, F2, and F3. These findings are in accordance with the findings of Molla and Solaiman[34], Sharma & Jaga[33] and Yadav et al.[35]. The increase in yield in kg ha−1 for F5 treatment might be due to the dual inoculation of mycorrhiza + rhizobium along with NPK fertilizer and the better utilization of inorganic fertilizers. Another possible reason for higher yield in kg per ha might be due to the translocation of essential metabolites and photosynthates to the economic parts of plants[33]. Non-significant results were obtained for yield ha>−1 due to interaction between variety and fertilizer.
Thousands seed weight (test weight)
-
Significant variation was observed on thousands seed weight of cowpea as influenced by different variety (Table 5). It was noted that the higher test weight (151.62 g) was recorded from Malepatan-1 where the lowest test weight (125.46 g) was recorded from Stickless. The findings of the present study are in line with the findings of Bhattarai et al.[4], who demonstrated that cowpea varieties show a great variation in terms of growth parameters and yield parameters. This variation in test weight might be due to the genetic variation among cowpea variety.
Thousands seed weight was significantly influenced due to fertilizer. The results revealed that the highest test weight (176.83 g) was achieved from F5 which was at par with F4 (167.19 g) but significantly superior over other treatments viz; F0 (116.38 g), F1 (123.08 g), F2 (123.03 g) and F3 (124.71 g). These findings are in accordance with the findings of Dobo[36], who demonstrated that since rhizobia bacteria supply nitrogen to the host plant through biological nitrogen fixation and AM fungi deliver phosphate from the soil beyond root access, the tripartite interactions in legumes have a synergistic effect on the host plant growth response. Non-significant result was obtained for thousands seed weight due to interaction between variety and fertilizer.
Economics of cowpea production
-
The economic analysis of crop treatments, including different varieties and fertilizer applications, revealed distinct variations in key economical parameters. Among the varieties, Malepatan-1 demonstrated the highest gross return of NRs. 424,419.6 ha−1, net return of NRs. 304,419.6 ha−1 and benefit cost ratio (BCR) of 2.53. Stickless, another variety, exhibited a lower gross return of NRs. 391,072.8 ha−1 and a net return of NRs. 266,572.8 ha−1. In terms of fertilizers, the Mycorrhiza + Rhizobium combination demonstrated the highest BCR of 3.76, reflecting efficient cost management (Fig. 1). The RDF treatment resulted in lowest BCR of 2.18.
Table 6 illustrates a sensitivity analysis conducted on cowpea cultivation, specifically employing the mycorrhiza + rhizobium treatment. The analysis explores the impact of variations in total cost and gross benefit on key economic parameters. Under the new condition of a 10% increase in total cost, the corresponding figures show a new total cost of NRs. 119,625, with a gross return of NRs. 518,569.2. This adjustment results in a net return of NRs. 398,944.2 and a BCR of 3.33. Despite the increased cost, the venture remains economically feasible. In the scenario where gross benefit is decreased by 10%, the new total cost is NRs. 108,750, with a gross return of NRs. 466,712.3. The net return and BCR stand at NRs. 357,962.3 and 3.29, respectively. Even with reduced gross benefit, the enterprise remains economically viable. Under simultaneous conditions of a 10% increase in total cost and a 10% decrease in gross benefit, the new total cost is NRs. 119,625, with a gross return of NRs. 466,712.3. This adjustment yields a net return of NRs. 347,087.3 and a BCR of 2.90. Despite these compounded adjustments, the venture remains economically feasible.
Table 6. Sensitivity analysis of cowpea with the application of mycorrhiza + rhizobium treatment.
S.N. New condition New total cost (NRs.) New gross return (NRs.) Net return (NRs.) BCR Remarks 1 Total cost increased by 10% 119625 518569.2 398944.2 3.33 Still feasible 2 Gross benefit decreased by 10% 108750 466712.3 357962.3 3.29 Still feasible 3 Total cost increased by 10% and
gross benefit decreased by 10%119625 466712.3 347087.3 2.90 Still feasible Pearson correlation analysis among various parameters
-
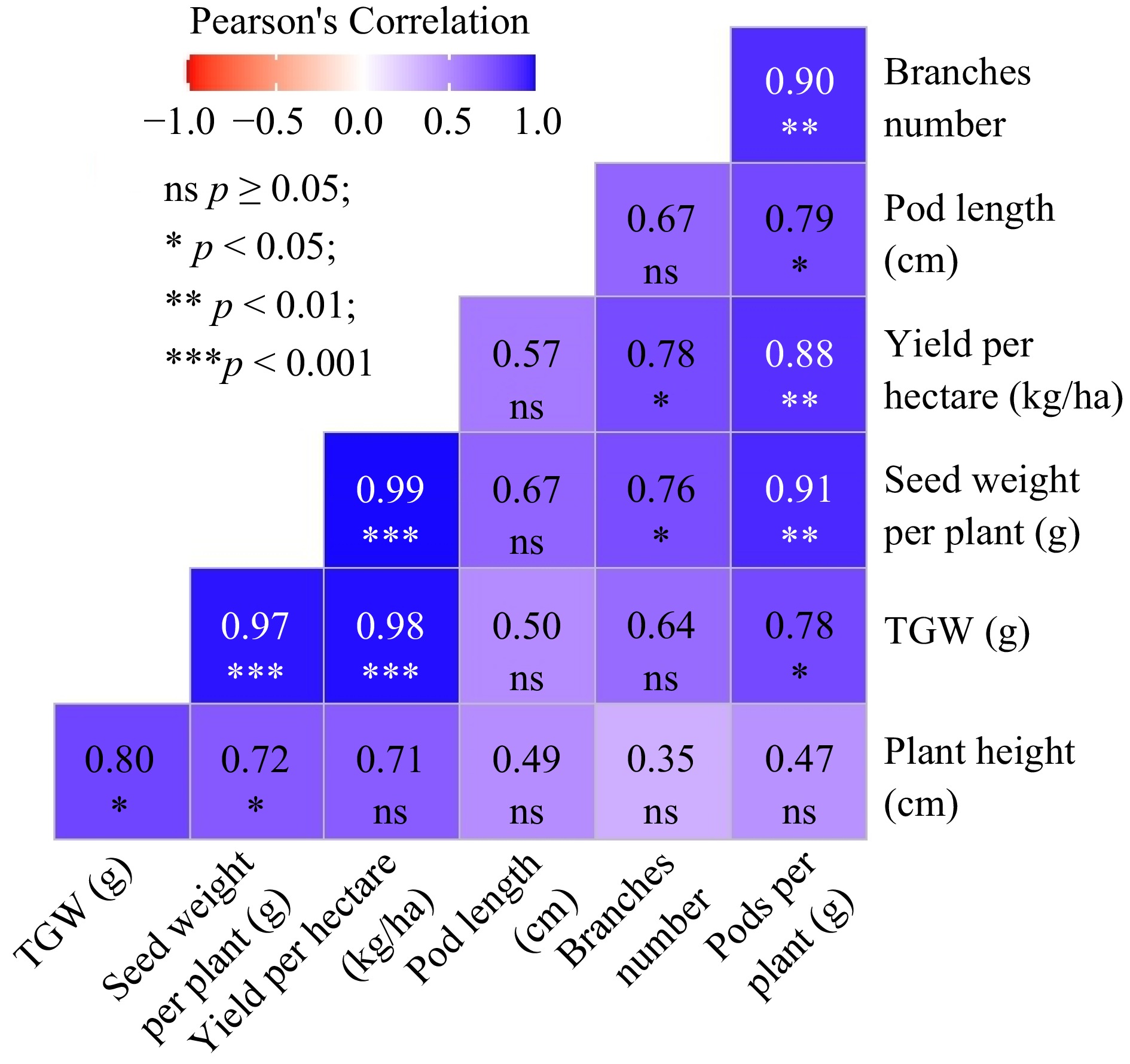
The Pearson correlation reveals valuable insights into the interrelationships among different variables influencing the growth and yield of cowpea varieties (Fig. 2). Plant height demonstrates a moderate positive correlation with the number of branches (0.35) and strong positive correlations with pods per plant (0.47), seed weight per plant (0.72), pod length (0.49), thousand grain weight (TGW) (0.80), and yield per hectare (0.71). Similarly, the number of branches exhibits a moderate positive correlation with plant height (0.35) and strong positive correlations with pods per plant (0.90), seed weight per plant (0.76), pod length (0.67), TGW (0.64), and yield per hectare (0.78). Notably, pods per plant demonstrate a strong positive correlation with both plant height (0.47) and the number of branches (0.90). Seed weight per plant shows strong positive correlations with plant height (0.72), the number of branches (0.76), and pods per plant (0.91). Furthermore, pod length exhibits strong positive correlations with plant height (0.49), the number of branches (0.67), and pods per plant (0.79). TGW shows strong positive correlations with plant height (0.80), the number of branches (0.64), pods per plant (0.78), and seed weight per plant (0.97). Finally, yield per hectare displays strong positive correlations with plant height (0.71), the number of branches (0.78), pods per plant (0.88), seed weight per plant (0.99), pod length (0.57), and TGW (0.98). These findings suggest that the use of bio-fertilizers and chemical fertilizers significantly influences the growth and yield of cowpea varieties, with positive correlations observed across multiple parameters.
-
Both of the studied varieties show significant variation in terms of growth and yield parameters. In comparison to the stickless variety, Malepatan-1 exhibited superior traits such as increased plant height, seed weight, thousand grain weight, and overall yield when compared to the stickless variety. Despite the Stickless variety exhibiting a higher number of branches plant−1 and a greater number of pods plant−1, its yield is significantly lower compared to the Malepatan-1 variety. This difference in yield can be attributed to the significantly greater seed weight observed in the Malepatan variety. This observation highlights the crucial role of seed weight in determining the overall yield of cowpea varieties. It is important to note that the environmental conditions and nutrient availability were consistent for both varieties during the study; therefore, the discrepancy in seed weight and subsequent yield can be attributed to the inherent genetic makeup of the varieties. These findings are in accordance with the findings of Bhattarai et al.[4] and Jannat[37], who observed significant variation among varieties in terms of growth and yield parameters except for pod length. These variations might be due to the genetic causes existing within different varieties[4].
The application of mycorrhiza + rhizobium + RDF demonstrated favorable outcomes, including increased plant height, a higher number of branches plant-1, a greater number of pods plant−1, and higher seed weight plant−1. However, when considering thousand seed weight and yield, the combined application of biofertilizers (mycorrhiza + rhizobium) and the combined application of RDF, mycorrhiza, and rhizobium yielded the highest results. Cowpea, like other legumes, benefits from nitrogen fixation by rhizobium bacteria. When combined with mycorrhiza, which enhances nutrient absorption, it ensures an ample supply of essential nutrients for plant growth and reproductive development, leading to increased pod production and seed weight[38−40]. It is noteworthy that the individual application of either biofertilizers or chemical fertilizers did not lead to significant improvements in growth and yield parameters compared to the control plot. The combined application of mycorrhiza, rhizobium, and RDF likely created a synergistic effect on plant growth. Mycorrhiza can improve nutrient uptake[41], rhizobium facilitates nitrogen fixation[42,43], and RDF provides essential macro and micronutrients. Together, these factors promote better plant development, resulting in higher pod number and seed weight. The higher seed weight per plant might be due to the combined application of bio-fertilizer and inorganic fertilizers might be due to the translocation of essential metabolites and photosynthates to the economic parts of plant. Furthermore, there was no significant difference in yield between the plots that received the combined application of biofertilizers and the plots that received the combined application of biofertilizers and inorganic fertilizers. Therefore, it is advisable to apply only the combination of biofertilizers, as the additional cost of chemical fertilizers does not justify the same yield as achieved without their application.
Molla & Solaiman[34] also reported the findings for mung bean, who observed that dual inoculation of mycorrhiza and rhizobium with N, P fertilizers increases growth parameters, nodulation, and enhances nutrient uptake. Also, our findings are in accordance with the findings of Yadav et al.[35], who demonstrated that there was a significant increment in vegetative growth, yield attributes, yield, and test weight of Vigna Mungo due to the inoculation of PSB, VAM along with phosphorus. Researchers such as Channaveerswami[44] in groundnut and Ghimire et al.[7] in bitter gourd have found that the combination of organic and inorganic fertilizers yielded better outcomes compared to the use of inorganic fertilizers only. This synergistic effect can be attributed to the fact that organic fertilizers contain essential nutrients, growth-promoting substances, and beneficial microflora, which, when combined with inorganic fertilizers, create favorable soil conditions that enhance nutrient use efficiency[45].
Mycorrhizal fungi form symbiotic relationships with plant roots, extending the root system and increasing the plant's ability to access nutrients and water[46−48]. This enhanced root development likely contributed to the observed improvements in yield parameters. Biofertilizers have been recognized for their ability to enhance soil organic matter content, leading to reduced bulk density and decreased compaction[49,50]. This, in turn, creates a favorable growing environment for plants, promoting improved growth and development. The improved plant height observed in certain studies can be attributed to enhanced nodulation and nutrient uptake facilitated by the inoculation of biofertilizers[51]. The increase in the number of branches may be attributed to the mineralization and easy solubilization of nutrients by biofertilizers, leading to excessive vegetative growth and an increased number of branches plant−1[52]. Additionally, Sharma & Jaga[33] found that the combined application of vesicular-arbuscular mycorrhiza, rhizobium, phosphate-solubilizing bacteria, and 75% NPK fertilizer resulted in a higher number of leaves plant−1, increased leaf area, improved photosynthesis efficiency, and ultimately higher yield and yield attributes. The combined application of mycorrhiza and rhizobium with NPK fertilizer, along with the effective utilization of inorganic fertilizers, can account for the improved yield attributes such as the number of pods plant−1, seed weight plant−1, overall yield, and thousand seed weight. The higher in thousands seed weight for treatment F5 might be due to the dual inoculation of mycorrhiza and rhizobium and their synergistic effect on cowpea plant. This combination may facilitate the translocation of essential metabolites and photosynthates to the economically important parts of the plant[33].
The mycorrhiza and rhizobium treatment, excluding chemical fertilizer (RDF), has demonstrated a higher BCR, primarily owing to two crucial factors. Firstly, the reduced treatment costs contribute to this outcome, and secondly, the heightened crop yields play a pivotal role. The combination of biofertilizers ensures a balanced and synergistic nutrient supply to crops, effectively enhancing their growth and maximizing yield potential. As a result, this approach frequently results in increased crop productivity when compared to the use of individual fertilizer types, as noted in previous studies[45]. The results indicate the feasibility of cowpea cultivation under diverse conditions, encompassing both favorable and unfavorable scenarios. The calculated BCR exceeding 1 underscores the economic viability of cowpea cultivation. The positive net return further substantiates its profitability within the specified geographic area. Furthermore, a rigorous sensitivity analysis of mycorrhiza and rhizobium treatment, involving a 10% increase in variable costs and a corresponding 10% decrease in benefits, was conducted. Remarkably, the BCR ratio remained above 1 even under these altered conditions, affirming the robust feasibility of the enterprise, particularly in less favorable circumstances. The sensitivity analysis demonstrates the resilience of cowpea cultivation with the mycorrhiza + rhizobium treatment, as evidenced by sustained positive net returns and BCR values across various altered conditions.
-
Cowpea, being a leguminous nitrogen-fixing plant, requires a relatively low amount of fertilizers, particularly chemical fertilizers. Based on the obtained results, it can be concluded that the application of a combination of rhizobium and mycorrhiza would be more desirable. This approach gives a yield equivalent to those achieved through the combined application of chemical fertilizers with pronounced growth and yield attributes and biofertilizers while also significantly reducing fertilizer costs, making it a more economical choice. Mycorrhiza + rhizobium achieved the highest BCR, surpassing the BCR obtained with the combined application of biofertilizers and chemical treatments, making it more environmentally friendly and economically sound. Furthermore, the Malepatan-1 variety outperforms the stickless variety across various growth parameters, yield attributes, and seed yield. Consequently, it is strongly recommended for cultivation for maximizing cowpea production. Thus, the cultivation of the Malepatan-1 cowpea variety with the application of a combination of biofertilizers is the most suitable approach in terms of both production outcomes and economic viability. Notwithstanding the favorable outcomes achieved, it is imperative to emphasize that the effectiveness of diverse fertilizers and cultivars must undergo comprehensive evaluation on a broader scale, encompassing various ecological regions.
-
The authors confirm contribution to the paper as follows: study conception and design, data collection, analysis and interpretation of results: Gautam N, Ghimire S, Kafle S, Dawadi B; draft manuscript preparation: Ghimire S, Gautam N. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
-
The authors are thankful to the Agriculture and Forestry University, Chitwan, Nepal, Prime Minister Agriculture Modernization Project, Nepal and Assistant Professor Tulsi Parajuli for continuous support during the research work.
-
The authors declare that they have no conflict of interest.
- Copyright: © 2024 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Gautam N, Ghimire S, Kafle S, Dawadi B. 2024. Efficacy of bio-fertilizers and chemical fertilizers on growth and yield of cowpea varieties. Technology in Agronomy 4: e007 doi: 10.48130/tia-0024-0004
Efficacy of bio-fertilizers and chemical fertilizers on growth and yield of cowpea varieties
- Received: 07 December 2023
- Revised: 12 February 2024
- Accepted: 21 February 2024
- Published online: 10 April 2024
Abstract: Cowpea is valued for its nutrition and nitrogen-fixing ability, and investigating bio-fertilizers offers a sustainable way to enhance cowpea growth and yield. A field experiment was conducted from February to June 2022 in the Nawalparasi West, Nepal, to investigate the impact of different fertilizers, including bio-fertilizers and chemical fertilizers, on the growth parameters and yield of cowpea varieties. Employing a double factorial Randomized Complete Block Design (RCBD), the experiment considered two factors: cowpea varieties (Malepatan-1 and Stickless) and fertilizers (control, mycorrhizal, rhizobia, recommended dose of chemical fertilizer (RDF), mycorrhizal + rhizobia, and mycorrhiza + rhizobium + RDF). Malepatan-1 exhibited superior growth and yield compared to Stickless, with higher plant height (125.73 cm), seed weight plant−1 (72.29 g), thousand grain weight (151.62 g), and yield ha−1 (3,536.83 kg ha−1). While the application of rhizobia + mycorrhiza and chemical fertilizers increased various growth parameters, mycorrhiza + rhizobium showed comparable results in terms of yield (4,321.41 kg ha−1) and thousand grain weight (167.19 g) compared to the combination of the former (4,714.26 kg ha−1 and 176.83 g, respectively). Moreover, mycorrhiza + rhizobium demonstrated a higher benefit-cost ratio (3.76), making it economically and environmentally preferable to biofertilizers with chemicals. The study recommends mycorrhiza + rhizobium for its comparable yield, superior economic returns, and environmental sustainability over biofertilizers combined with chemicals. For maximizing economic and sustainable production, the study suggests using the Malepatan-1 cowpea variety with biofertilizers, excluding chemical (inorganic) fertilizers.
-
Key words:
- Arbuscular mycorrhizal fungi /
- Bio-fertilizers /
- Cowpea /
- Integrated nutrient management /
- Rhizobium