-
Chrysanthemum (Chrysanthemum × morifolium [Ramat.] Kitamura) is native to China and is cultivated for sale as fresh cut flowers, planting in ornamental gardens, landscaping, and medicinal use. Floral fragrance is an important trait which mediates the intraspecific and interspecific interactions of plants[1]. Floral volatiles can attract pollinators, which promotes sexual reproduction, as well as natural enemies of phytophagous insects, which prevents attack by insect pests[2]. In addition, volatile compounds protect plants from abiotic stresses, such as strong light, high temperature, and oxidative stress[3,4]. The importance of floral fragrance is receiving increased research attention.
Floral fragrance is determined by the type and content of volatile organic compounds (VOCs)[5]. A variety of VOCs are synthesized in plants. Depending on their source, VOCs are classified into three categories: terpenoids, phenylpropanoids/benzenoids, and fatty acid derivatives[1].
Terpenoids, which form the largest class of VOCs are composed of several isoprene (C5) structural units. Depending on the number of structural units, terpenoids are classified as monoterpene (C10), sesquiterpene (C15), and diterpene (C20) compounds[6]. For example, monoterpenoids are the main aromatic substances in rose (Rosa × hybrida) flowers[7] , while linalool and ionone are the main compounds in Osmanthus fragrans flowers[8].
Phenylpropanoids/benzenoids for the second largest class of plant VOCs[9], however, the complete biosynthetic pathway of phenylpropanoid compounds remains unclear. According to current knowledge, the direct precursor of phenylpropanoid/benzenoid compounds is phenylalanine, which is synthesized mainly through the shikimate pathway[10]. The flower fragrance of Petunia (Petunia hybrida) is mainly attributable to phenylpropanoid/benzenoid compounds, among which benzaldehyde, phenylacetaldehyde and methyl benzoate are the most abundant[11].
Fatty acid derivatives are the third group of plant volatile substances. Acetyl coenzyme A (acetyl CoA) is the precursor of fatty acid derivatives. Acetyl CoA enters the lipoxygenase (LOX) pathway, and produces volatile substances through a series of reactions. According to a recent study, (E)
-2-hexenal is one of the main compounds responsible for the floral fragrance of carnation[12]. Previous research on the floral fragrance of chrysanthemum has mainly focused on the identification of aromatic compounds. In chrysanthemum and its wild relatives, monoterpenoids and oxygenated monoterpenoids, including camphor, α-pinene, laurene, and eucalyptus alcohol, are the predominant volatile components[13]. Monoterpenoids and sesquiterpenoids, including hydrocarbons, esters, aldehydes, ketones, phenols, and organic acids, are the predominant compounds of chrysanthemum volatile oil[14]. Investigation of the relationship between the accumulation and release of terpenoids in 44 related species and cultivars of chrysanthemum revealed that the release of terpenoids is strongly correlated with their internal concentration, whereas the concentration of terpenoids is associated with the release of the compound and the size of the capitulum. Tubular florets have a greater impact on the release of volatile substances than ligulate florets. In addition, the involucre and receptacle serve as the main sites for the accumulation of terpenoids[15]. The volatiles of chrysanthemum cultivar 'Boju' are mainly eucalyptus alcohol, filifon, pyrethrone, and trans- and cis-pyrethroid acetates[16]. An aromatic wild species, Dendranthema indicum (Chrysanthemum indicum var. aromaticum) was introduced to breed aphid-resistant offspring through hybridization with Chrysanthemum nankingense. Nineteen compounds of aphid resistant lines were selected and cis-4-thujanol was confirmed to be an effective aphid repellent[17]. Thus, the composition and content of volatiles differ substantially among chrysanthemum species. Although the volatile substances of chrysanthemum have been researched, the classification of chrysanthemum fragrance has not yet been reported.
As stated above, the aroma of flowers determines the commercial value of chrysanthemum cultivars, especially those used for tea and edible purposes. Because of long-term natural selection and evolution, the fragrance type of chrysanthemum is highly diverse. Nevertheless, previous research on chrysanthemum floral fragrance mainly focused on the determination of the volatile compounds and their contents, and research on the classification of chrysanthemum fragrance types is lacking. In this study, the aroma type of among a large sample of chrysanthemums was investigated using a sensory evaluation method, and volatile substances of representative chrysanthemums of each aroma type were analyzed by gas chromatography–mass spectrometry (GC–MS). Based on the aroma type, chrysanthemum accessions were classified into six categories, providing a theoretical basis for the accelerated breeding of new chrysanthemum germplasm with specific aroma types.
-
Chrysanthemum materials used for fragrance classification were collected from major parks in Beijing (China) and the chrysanthemum resource garden at the Shangzhuang Experimental Station of the China Agricultural University, Beijing, China.
Chrysanthemums used for GC-MS determination were Chrysanthemum × morifolium 'Qihuang', C. indicum L., C. × morifolium 'Bairuixiang', C. × morifolium 'Quehuan', C. × morifolium 'Xiaokuixiang', and C. × morifolium 'Sigong'.
Artificial pollination
-
Rooted cuttings of 'Xiaokuixiang' were planted at the Shangzhuang Experimental Station (Beijing, China), and reproductive isolation was carried out. Artificial self-pollination was conducted at the onset of flowering. The seeds were collected when mature.
Open hybridization with 'Xiaokuixiang' as the female parent
-
A chrysanthemum cultivar with the same flowering period as 'Xiaokuixiang' was planted on either side of the female parent ('Xiaokuixiang'). Sterilized tweezers were used to remove the stamens at the onset of flowering of 'Xiaokuixiang', and the upper portion of the corolla of the outer florets in the capitulum was removed to expose the pistils. The seeds were collected when mature.
Sensory evaluation
-
The aroma type of chrysanthemum was determined by means of a questionnaire. The members of the research group randomly distributed questionnaires to recipients. The aroma types were determined after statistical analysis. On the basis of the questionnaire, 24 students and teachers who were familiar with chrysanthemums and had the ability to distinguish aroma types were invited as sensory evaluators to screen representative cultivars of each fragrance type.
Solid phase microextraction
-
Flowering stems of chrysanthemum were cut with secateurs, immediately placed in a bucket containing clean water, and transported to the laboratory for sampling. The capitulum (0.2–1.0 g) of each chrysanthemum was placed in a sampling bottle, with three replicates per cultivar, and then 15 μL of the internal standard (43.25 ng/g ethyl decanoate) was added to each bottle.
Solid phase extraction head comprised 50/30 μm divinylbenzene/carbon/polydimethyl siloxane. The sample was placed in a 15 ml glass bottle in a 45 °C water bath, the extraction head was inserted, and the headspace was extracted for 30 min. The extraction head was analyzed in the 250 °C injection port for 3 min.
Gas chromatography–mass spectrometry
-
The GC-MS analysis was conducted using a GCMS-QP2010 mass spectrometer (Shimadzu, Kyoto, Japan). The chromatographic conditions were as follows: injection port temperature, 250 °C; injection mode, split flow; total flow rate, 27.4 mL/min; split ratio, 20; ion source temperature, 200 °C; and interface temperature, 250 °C.
The total analysis time was 30 min. The initial temperature was 40 °C, held for 1 min, then increased to 280 °C at 10 °C/min, held for 5 min, and the solvent delay time was 2.5 min. Mass spectrum conditions were: detector, 1 kV; mass scanning range 30–500 m/z; and full scanning mode.
Statistical analysis
-
All determinations were performed with three biological replicates. Microsoft Excel and Graphpad Prism 8 were used to process and analyze the data. The results are expressed as the mean ± standard deviation (SD). Statistical significance was assessed using one-way analysis of variance (p < 0.05).
-
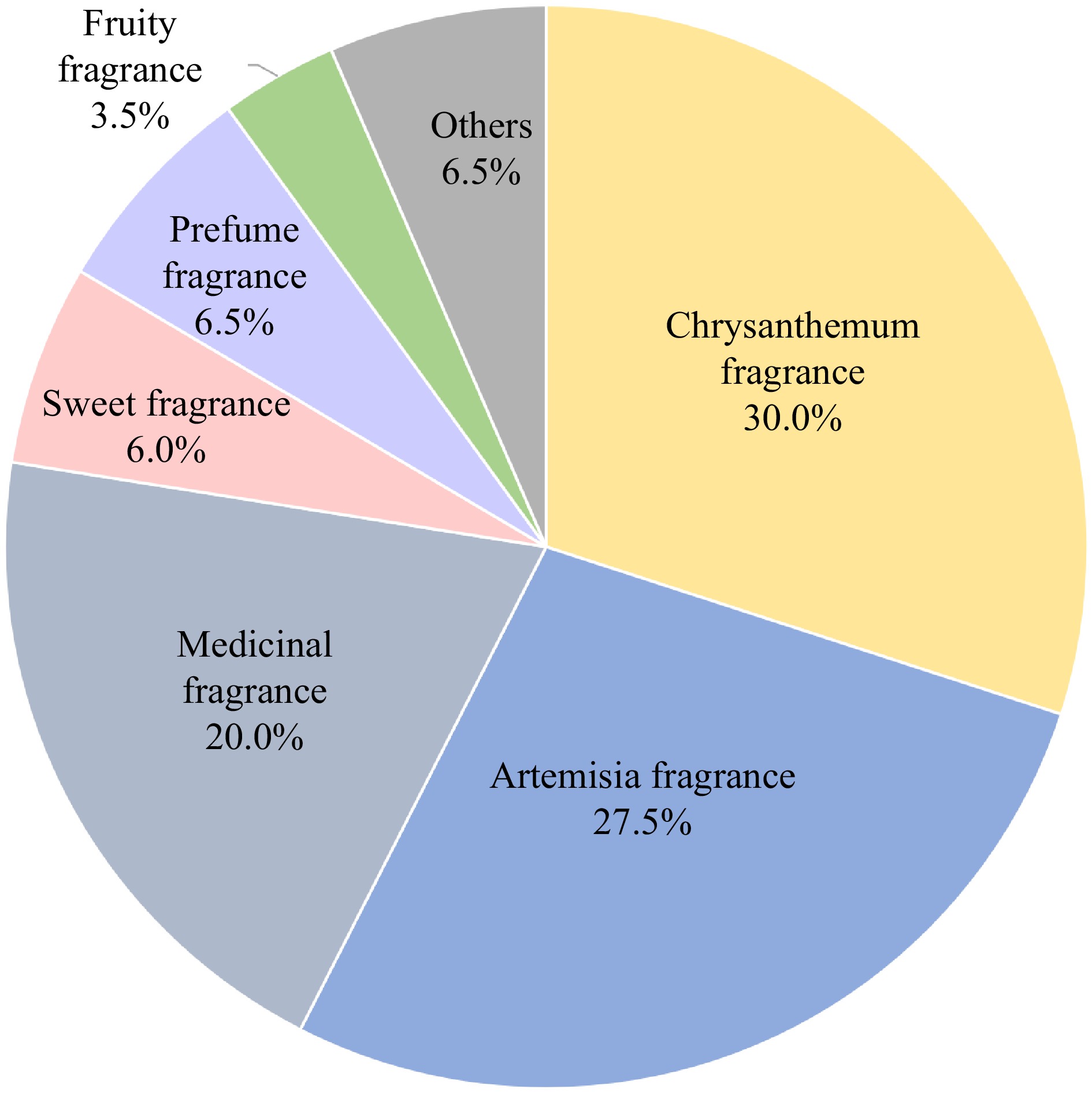
To classify the fragrance types of chrysanthemums, we performed sensory evaluation of the aroma characteristics of 520 chrysanthemum accessions. The fragrance of chrysanthemums could be grouped into six types: chrysanthemum fragrance, artemisia, medicinal, sweet, perfume fragrance, and fruity (Fig. 1). Among these types, chrysanthemum fragrance accounted for 30% of the cultivars, artemisia for 27.5%, medicinal for 20.0%, sweet for 6.0%, perfume fragrance for 6.5%, fruity for 3.5%, and others for 6.5%.
According to the sensory evaluation results, the chrysanthemum accessions with the highest score in each fragrance type was selected as the representative of that category. The results showed that 91.67% of the evaluators considered that 'Sigong' was the most typical chrysanthemum cultivar with chrysanthemum fragrance, 91.67% considered that wild chrysanthemum (Chrysanthemum indicum L.) was the most typical chrysanthemum with artemisia fragrance, 87.50% considered that 'Qihuang' was the most typical cultivar chrysanthemum with medicinal fragrance, 75.00% considered that 'Quehuan' was the most typical chrysanthemum cultivar with sweet fragrance, 79.17% considered that 'Bairuixiang' was the most typical chrysanthemum cultivar with perfume fragrance, and 83.33% considered 'Xiaokuixiang' was the most typical chrysanthemum cultivar with fruity fragrance (Table 1). Therefore, to further analyze the aroma components of chrysanthemums of different fragrance types, the identity components and contents of the volatile substances were determined by selecting 'Sigong', C. indicum, 'Qihuang', 'Quehuan', 'Bairuixiang', and 'Xiaokuixiang' as the representative accessions of the chrysanthemum fragrance, artemisia, medicinal, sweet, perfume fragrance, and fruity fragrance, respectively (Fig. 2).
Table 1. Statistics foruation of chrysanthemum fragrance.
Chrysanthemum cultivars Chrysanthemum
fragrance (%)Artemisa
fragrance (%)Medicinal
fragrance (%)Sweet
fragrance (%)Perfume
fragrance (%)Fruity
fragrance (%)Sigong 91.67 8.33 0 0 0 0 Chrysanthemum indicum 8.33 91.67 0 0 0 0 Qihuang 0 12.50 87.50 0 0 0 Quehuan 16.67 0 0 75.00 0 8.33 Bairuixiang 0 0 0 12.50 79.17 8.33 Xiaokuixiang 0 0 0 12.50 4.17 83.33 
Figure 2.
Chrysanthemum materials used in the experiment. (a) Chrysanthemum indicum L.; (b) C. × morifolium 'Xiaokuixiang'; (c) C. × morifolium 'Quehuan'; (d) C. × morifolium 'Bairuixiang'; (e) C. × morifolium 'Qihuang'; (f) C. × morifolium 'Sigong'.
Identity and content of VOCs in chrysanthemum accessions of different aroma types
-
To explore the biochemical basis of different chrysanthemum fragrance types, the identity and content of volatile substances of representative chrysanthemum accessions were determined by GC–MS. Terpenoids were predominant in C. indicum (artemisia fragrance) and fatty acid derivatives were the most in 'Xiaokuixiang' (fruity fragrance), and the number of phenylpropanoid/benzenoid compounds was low in accessions of all fragrance types (Supplemental Table S1). As shown in Fig. 3, accessions with chrysanthemum fragrance, artemisia, medicinal, sweet, and perfume fragrance were dominated by terpenoids, accounting for more than 50% of all VOCs, followed by fatty acid derivatives, however, no significant difference was observed in the types and proportions of terpenoids and fatty acid derivatives in the of 'Xiaokuixiang' (fruity fragrance).

Figure 3.
Proportions of volatile organic compounds in chrysanthemum with different fragrance types.
To compare the differences among chrysanthemum accessions of different aroma types, the VOCs of different fragrance types were analyzed quantitatively. As shown in Fig. 3, except for 'Xiaokuixiang', other representative chrysanthemum accessions showed the highest content of terpenoids. Fatty acid derivatives were the most volatile substances.
In 'Sigong', terpenoids were the most abundant, followed by fatty acid derivatives, and lastly phenylpropanoids/benzenoids. Eucalyptol was the main terpenoid, (E)-2-hexenal was the main fatty acid derivative, and o-cymene was the main phenylpropanoid/benzenoid compound (Fig. 4a). The compounds with the highest contents in 'Sigong' were eucalyptol, 2-pinene-6-one, and α-pinene, with the contents of 2,158.89, 849.00 , and 743.28 ng/µL/g, respectively (Fig. 4a).
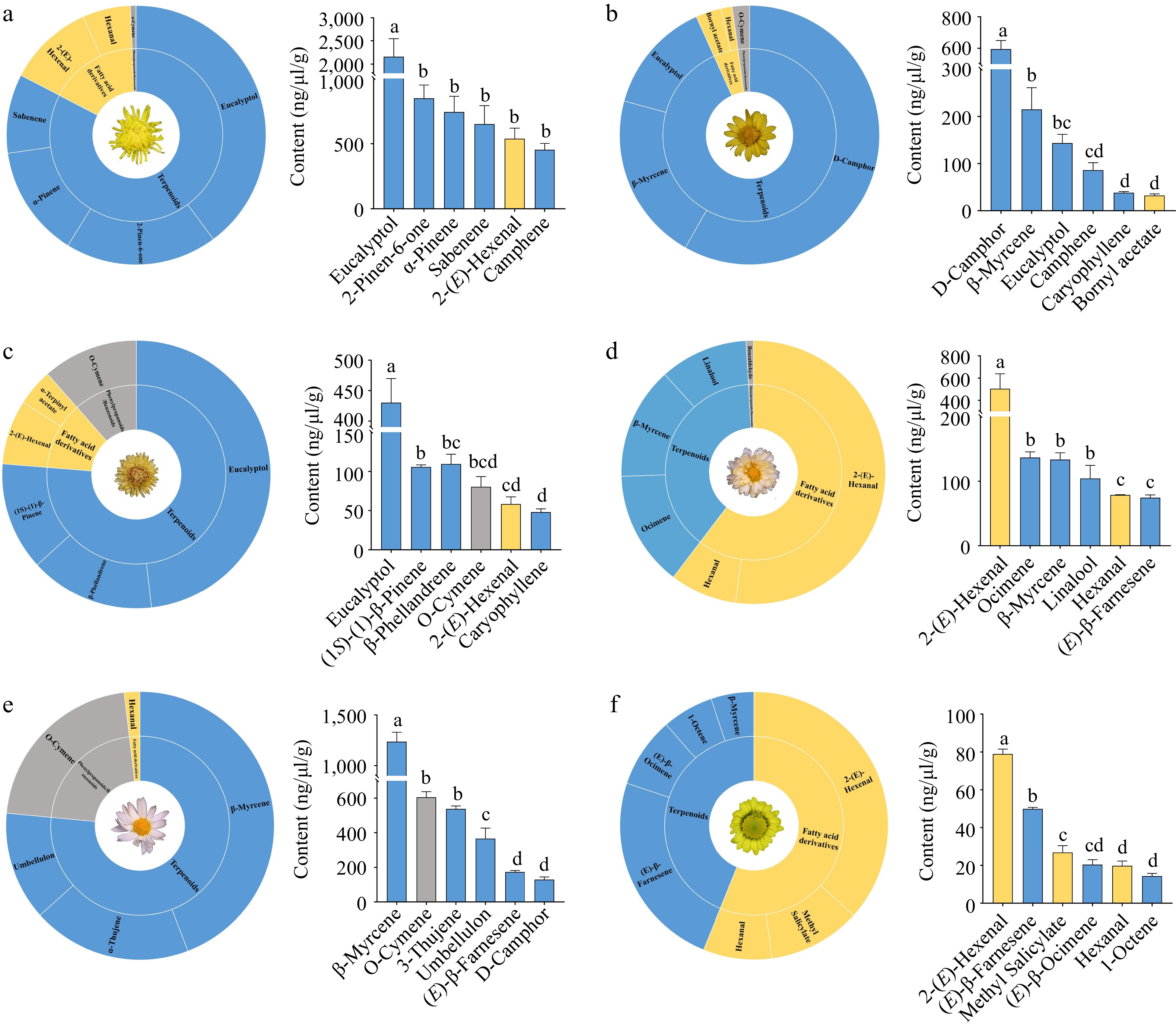
Figure 4.
Analysis of main volatile substances in Chrysanthemum with different fragrance types. (a) Analysis of main volatile substances in 'Sigong' with chrysanthemum fragrance; (b) Analysis of main volatile substances in Chrysanthemum indicum with artemisia fragrance; (c) Analysis of main volatile substances in 'Qihuang' with medicinal fragrance; (d) Analysis of main volatile substances in 'Bairuixiang' with perfume fragrance; (e) Analysis of main volatile substances in 'Quehuan' with sweet fragrance; (f) Analysis of main volatile substances in 'Xiaokuixiang' with fruity fragrance. FW: fresh weight.
In C. indicum (artemisia fragrance), terpenoids were the most abundant, followed by fatty acid derivatives and phenylpropanoid/benzenoid compounds (Fig. 4b). D-camphor, β-myrcene, and eucalyptol were the main terpenoids (595.18, 214.92, and 143.53 ng/µL/g, respectively), and o-cymene was the main phenylpropanoid/benzenoid (Fig. 4b).
In 'Qihuang' (medicinal fragrance), the content of terpenoids was the highest, followed by fatty acid derivatives and phenylpropanoid/benzenoid compounds (Fig. 4c). Eucalyptol was the main terpenoid compound, and (E)-2-hexenal and o-cymene were the main fatty acid derivatives and phenylpropanoid/benzenoid compounds, respectively (Fig. 4c). Eucalyptol, β-phellandrene, and (1S)-(−)-β-pinene showed the highest concentrations in 'Qihuang' (430.24, 109.42, and 105.67 ng/µL/g, respectively) (Fig. 4c).
In 'Bairuixiang' (perfume fragrance), the content of fatty acid derivatives was the highest, followed by terpenoids, and that of phenylpropanoid/benzenoid compounds was lowest (Fig. 4d). (E)-2-hexenal was the main component of fatty acid derivative, whereas ocimene, β-myrcene, and linalool were the main components among terpenoids (Fig. 4d). (E)-2-hexenal, ocimene and β-myrcene showed the high concentrations in 'Bairuixiang' (504.72, 136.79, and 133.58 ng/µL/g, respectively) (Fig. 4d).
In 'Quehuan' (sweet fragrance), the content of terpenoids were the most abundant, followed by fatty acid derivatives and phenylpropanoid/benzenoid compounds (Fig. 4e). β-myrcene, α-thujene and umbellulonl were the main components among terpenoids, (E)-2-hexenal were the main fatty acid derivatives, and o-cymene was the main phenylpropanoid/benzenoid compounds (Fig. 4e). The compounds with the high contents were β-myrcene, o-cymene, and α-thujene (1,236.08,604.88 and 537.86 ng/µL/g, respectively) (Fig. 4e).
In 'Xiaokuixiang' (fruity fragrance) fatty acid derivatives were the most abundant, followed by terpenoids (Fig. 4f). (E)-2-hexenal and methyl salicylate were the main fatty acid derivatives, and (E)-β-farnesene and (E)-β-ocimene were the main terpenoids (Fig. 4f). The compounds with the high contents in 'Xiaokuixiang' were (E)-2-hexenal, (E)-β-farnesene, and methyl salicylate (78.94, 49.94, and 26.88 ng/µL/g, respectively) (Fig. 4f).
Volatile organic compounds specific to accessions of different fragrance types
-
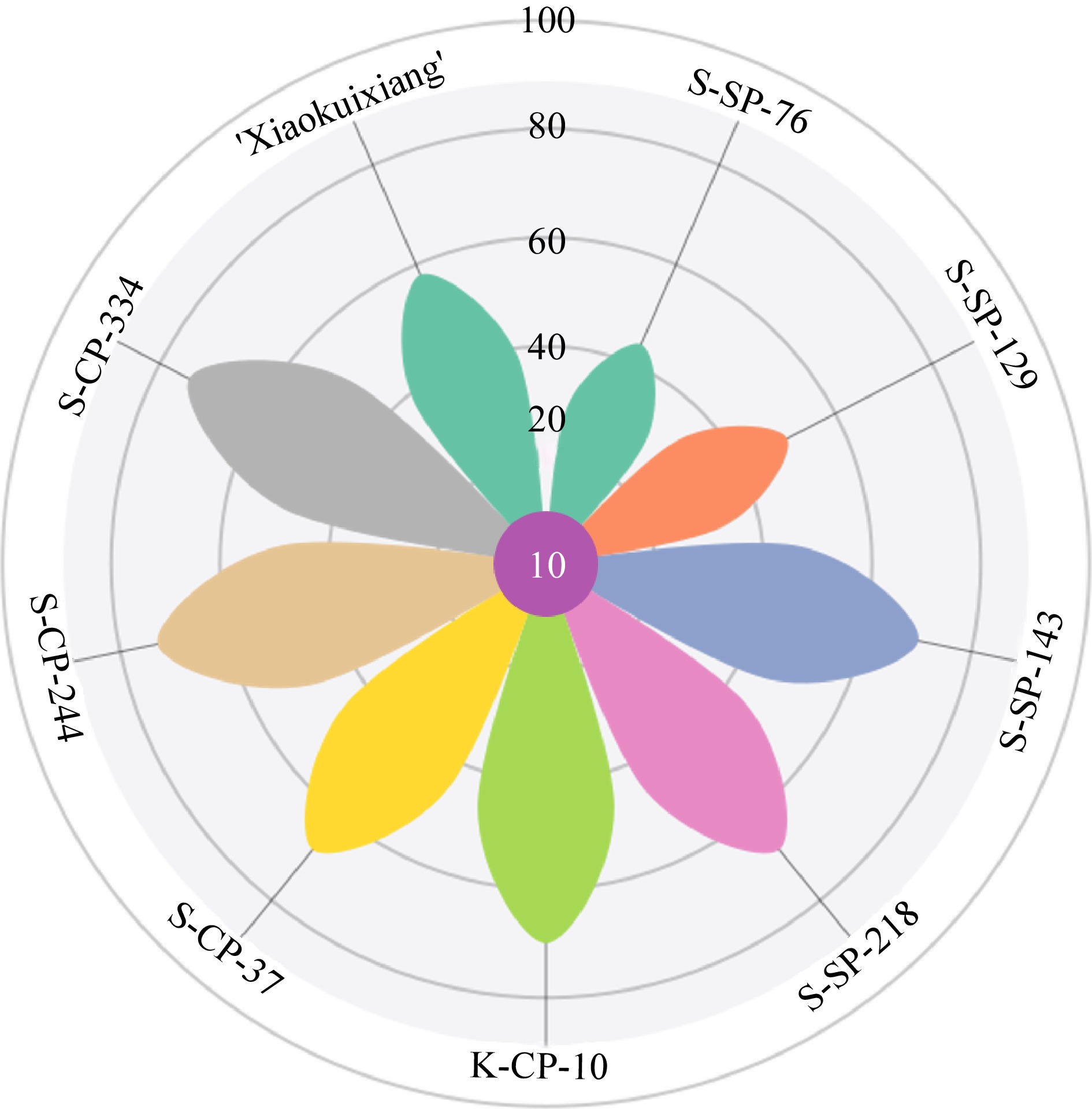
The preceding analysis showed that (E)-2-hexenal was the main volatile substance associated with the chrysanthemum, medicinal, perfume, and fruity fragrance types, and that eucalyptol was the main volatile substance associated with chrysanthemum, artemisia, and medicinal fragrance types. 2-Pinene-6-one, α-pinene and sabenene were the main volatile substances peculiar to the fragrance of 'Sigong', bornyl acetate was the main volatile substance peculiar to the artemisia fragrance of C. indicum. (1S)-(−)-β-pinene and β-phellandrene were the main volatile substances peculiar to the medicinal fragrance of 'Qihuang', ocimene and linalool were the main volatile substances peculiar to the perfume fragrance of 'Bairuixiang', 3-thujene and umbellulon were the main volatile compounds unique to the sweet fragrance of 'Quehuan', and methyl salicylate, (E)-β-ocimene, and 1-octene were the main volatile compounds unique to the fruity fragrance of 'Xiaokuixiang' (Fig. 5).
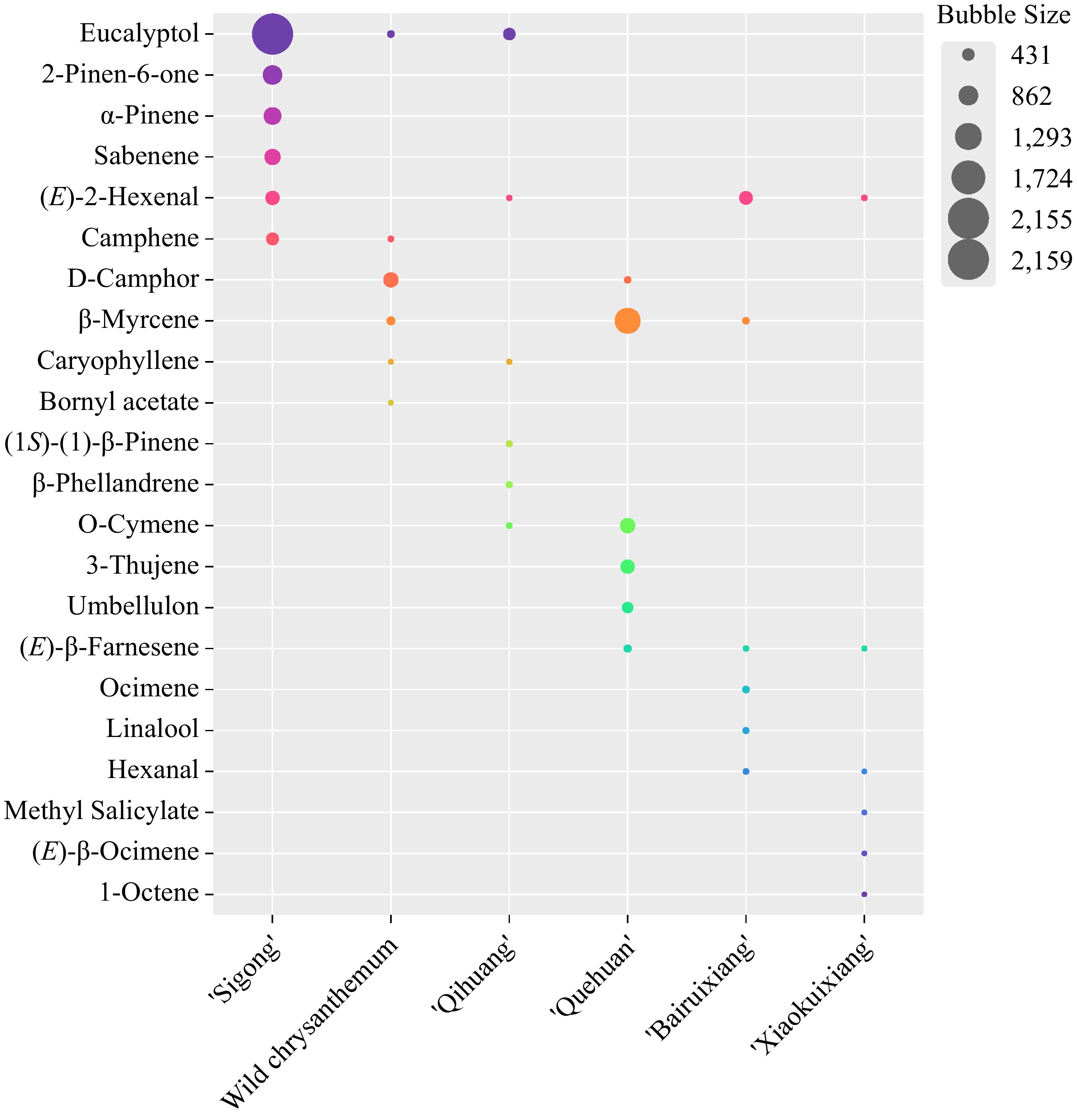
Figure 5.
Bubble chart of volatile organic compounds specific in different fragrance types of chrysanthemum accessions.
Analysis of aroma compounds of 'Xiaokuixiang' at different development stages
-
As shown in Fig. 6a, 'Xiaokuixiang' flowers were collected at three stages: bud stage, early flower stage and full flower stage. The aroma substances released by flowers at these three stages were divided into four categories: terpenoids (47%), fatty acid derivatives (32%), phenylpropanoid/benzenoid compounds (5%) and others (16%) (Fig. 6b). Quantitative analysis of different volatile substances showed that the content compounds were (E)-β-farnesene, (E)-2-hexenal, methyl salicylate and hexanal were high (Fig. 6c).

Figure 6.
Analysis of volatile substances in different stage of flower in 'Xiaokuixiang' . (a) Different stage of flower in 'Xiaokuixiang'; (b) Proportions of volatile organic compounds in three stages; (c) Analysis of main volatile substances in different stage of flower in 'Xiaokuixiang'.
Analysis of fragrance types in 'Xiaokuixiang' offspring
-
To analyze the genetic heritability of fruity fragrance type, we obtained 248 self-pollinated progenies and 383 hybrid progenies from 'Xiaokuixiang' as the female parent. Then, we determined the volatile substances of eight fruit-scented progenies by GC-MS (Supplemental Table S2), and compared the results with the volatile substances identified in 'Xiaokuxiang' . Ten volatile compounds were identified in nine fruit-scented chrysanthemums (Fig. 7), namely (E)-β-farnesene, 1-octene, caryophyllene, α-bergamotene, 1-hexanol, butanoic acid 2-methyl ethyl ester, butanoic acid 2-methyl propyl ester, butanoic acid 3-methyl hexyl ester, hexanoic acid ethyl ester and hexanal (Fig. 8).
-
Flower fragrance is an important trait of flowering plants. It not only attracts pollinators for sexual reproduction but also promotes the interaction between plants and the environment, thus protecting plants from attack by pathogens, parasites, and herbivores[18,19]. Chrysanthemum is an important commercial floriculture crop. After long-term interspecific hybridization and artificial selection, a variety of chrysanthemum types have been developed, which are enriched in secondary metabolites that affect the floral fragrance of chrysanthemum.
Aroma, as a trait perceptible by humans is particularly suitable for determining the fragrance type of chrysanthemum accessions through sensory evaluation. At present, the sensory evaluation method is used more systematically for the perception of food flavors. Although the sensory evaluation procedure for ornamental plants is not perfect, we used a relatively simple and convenient sensory evaluation method, employing a questionnaire survey to directly evaluate the perception of chrysanthemum fragrance. Through sensory evaluation of the fragrance of a large collection of accessions, we classified the accessions into six fragrance types: chrysanthemum fragrance represented by 'Sigong', artemisia fragrance represented by wild chrysanthemum, medicinal fragrance represented by 'Qihuang', sweet fragrance represented by 'Quehuan', perfume fragrance represented by 'Bairuixiang', and fruity fragrance represented by 'Xiaokuixiang'. Ongoing research will help improve the sensory evaluation of the aroma of ornamental plants.
Aroma is dependent on volatile substances perceived by olfactory organs[20]. Therefore, the fragrance of plants is determined by the type and content of volatile substances. Detection of volatile substances responsible for the floral fragrance of chrysanthemum by GC-MS indicated that the main volatile substances in most chrysanthemums accessions were terpenoids (Fig. 3). The finding that terpenoids are important components of floral fragrance in chrysanthemum is consistent with previous studies[15].
Further analysis showed that eucalyptol, 2-pinene-6-one, and α-pinene were the main volatile substances responsible for the fragrance of 'Sigong' (Fig. 4a). D-camphor, myrcene, and eucalyptol for artemisia fragrance (Fig. 4b), eucalyptol, β-phellandrene and (1S)-(-)-β-pinene for the medicinal fragrance of 'Qihuang' (Fig. 4c) and β-myrcene, o-cymene, and α-thujene for the sweet fragrance of 'Quehuan' (Fig. 4e). Previous studies have reported that the main volatile compounds of German chamomile (Matricaria recutita) are sesquiterpenes and monoterpenes, including (−)-γ-elemene, β-elemene, piperone, o-cymene, 3-perylene, and γ-terpene. The main volatile compounds reported for Roman chamomile (Chamaemelum nobile) are esters, including 3-methyl-2-butenoic acid, 3-methyl-2-alkenyl ester, 3-methyl-2-enoic acid, 2-methyl butyl ester, and 3-methyl-2-butenoic acid allyl ester[21]. These results indicate that the differences in volatile substances also exist among the different species of chamomile. 'Xiaokuixiang' is a novel chrysanthemum cultivar with a unique fragrance (fruity type) developed in the laboratory, whereas 'Bairuixiang' with perfume fragrance is a hybrid offspring of 'Xiaokuixiang'. The main volatile substances of 'Bairuixiang' are D-camphor, ocimene, and β-myrcene (Fig. 4d), and those of 'Xiaokuixiang' are (E)-2-hexenal, (E)-β-farnesene, and methyl salicylate (Fig. 4f). Analysis of the components of the various fragrance types by GC-MS revealed that the identity and content of the main volatile substances differed considerably among chrysanthemum cultivars.
However, the content of volatile substances alone cannot confirm the characteristic volatiles of each chrysanthemum accession. Given the diversity and complex composition of volatile substances, their absolute content is not the only standard to measure their contribution to fragrance, an additional important factor is the aroma threshold of volatile substances[22]. Aroma threshold is a quantitative expression of aroma[23]. At a certain concentration, the lower the aroma threshold, the stronger the aroma of the substance, and vice versa. Furthermore, the aroma threshold of a of volatile substance changes under different conditions and in different solvents Therefore, based on the quantitative data obtained for the volatile substances in the present study, we could only determine the chrysanthemum fragrance type through sensory evaluation, and the main volatile substances that contribute to each fragrance type. Determination of the characteristic aromatic substances responsible for each fragrance type requires further investigation. Overall, we divide chrysanthemum accessions into six categories based on the sensory evaluation of floral fragrance, and found a novel fragrance type (fruity) chrysanthemum. Furthermore, this work provides a theoretical basis for the accelerated breeding of new chrysanthemum germplasm with specific aroma types.
-
Chrysanthemum is an important ornamental and horticultural crop. However, there is no clear classification of its fragrance types. The results of this study divided chrysanthemum fragrance into six categories by sensory evaluation. We determined the types and content of VOCs in each chrysanthemum accession representative of different fragrance types by GC-MS. Furthermore, through genetic analysis, we determined the heritable aromatic substances of fruity fragrance chrysanthemum. Our present findings systematically classified the fragrance types of chrysanthemums and improved classification of chrysanthemum aroma types, providing a theoretical basis for the accelerated breeding of new chrysanthemum germplasm with specific aroma types.
This work was supported by National Natural Science Foundation (Grant no. 32002072).
-
The authors declare that they have no conflict of interest.
-
# These authors contributed equally: Zhiling Wang, Xin Zhao
- Supplemental Table S1 Statistics of various volatile substances in Chrysanthemum with different aroma types.
- Supplemental Table S2 Statistical table of sensory evaluation of chrysanthemum flavor.
- Copyright: © 2023 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Wang Z, Zhao X, Tang X, Yuan Y, Xiang M, et al. 2023. Analysis of fragrance compounds in flowers of Chrysanthemum genus. Ornamental Plant Research 3:12 doi: 10.48130/OPR-2023-0012
Analysis of fragrance compounds in flowers of Chrysanthemum genus
- Received: 14 February 2023
- Accepted: 19 April 2023
- Published online: 27 June 2023
Abstract: Chrysanthemum is one of the four major cut flowers in the world, with high ornamental and economic value. Fragrance is an important ornamental character of chrysanthemum flowers, especially those consumed as tea and other foods, and the flower fragrance is the major determinant of the commercial value of chrysanthemum cultivars. Currently, however, the research on chrysanthemum flower fragrance is mainly focused on the composition and content of fragrant compounds, and a clear classification of fragrance types is lacking. Here, we divided chrysanthemum fragrance into six categories based on sensory evaluation and determined the identity and content of fragrant compounds of chrysanthemum accessions representative of each fragrance type by GC-MS. In addition, we analyzed the conserved aromatic substances responsible for the fruity fragrance type chrysanthemum with multi-functional development potential, providing a theoretical basis for creating new chrysanthemum germplasm with specific fragrance types. The results of this study can accelerate the breeding process of chrysanthemum accessions with new fragrance types.
-
Key words:
- Chrysanthemum /
- Fragrance types /
- Sensory evaluation /
- Volatile compounds