-
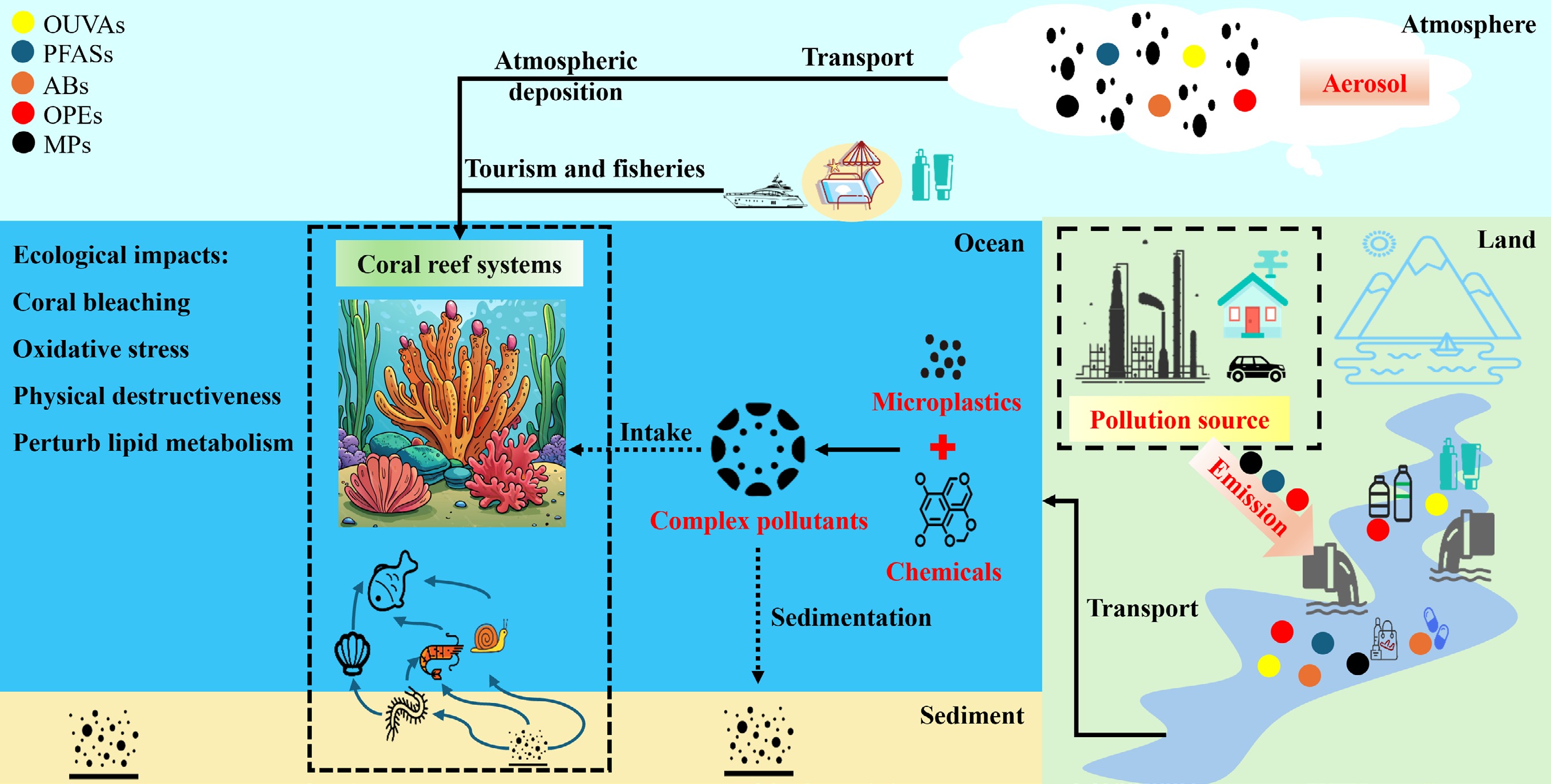
With the sustainable development of human society, emerging contaminants (ECs) such as microplastics (MPs), antibiotics (ABs), organic ultraviolet absorbers (OUVAs), organophosphate esters (OPEs), and perfluoroalkyl compounds (PFASs) have gradually become the focus of environmental science research in recent years because of their wide use in industrial production, daily consumption, agricultural and medical fields, and the multiple ecological and health risks that may be caused[1−5]. These chemicals enter the freshwater environment through discharges from urban sewage treatment plants, animal husbandry, and medical systems; agricultural runoff; industrial wastewater discharge; the degradation of plastic products; the use of cosmetics and sunscreen agents' and atmospheric deposition[3,6−9]. They are finally transported to offshore waters through coastal runoff, river-sea water system convergence, and tidal exchange[10−14]. Coral reefs, as one of the most biodiverse marine ecosystems with important ecological functions in the world, not only support the complex food web for marine organisms such as fish, but also play an irreplaceable role in coastal protection, fishery resource supply, and carbon cycle regulation[15]. However, in recent years, a number of studies have detected the presence of the above five ECs in seawater, sediments, coral tissues, symbiotic algae, and surrounding marine biological samples in coral reef areas[16−21]. The persistence, bioaccumulation, and multiple potential toxicities of these compounds may pose a threat to the structure, function, and health of coral reef ecosystems[22,23]. Therefore, the environmental occurrence, temporal and spatial distribution, toxic effects, and ecological risk assessment of these ECs in coral reef ecosystems are receiving considerable attention in the field of marine pollution and ecotoxicology.
Existing studies have gradually revealed the environmental behavior and potential impact of these five ECs on the coral reef ecosystem. In recent years, a number of monitoring surveys have shown that ECs such as MPs and PFAS have shown a significant enrichment trend in tropical coral reef waters, and their concentrations are spatially characterized by coastal hot spots, which may be controlled by current transport, tidal processes, and shore-based pollution discharge[20,24−26]. At the same time, pollutants with more source input characteristics, such as ABs, OPEs, and OUVAs, show obvious time series pulses, especially during the peak tourism season, the late stage of agricultural pesticide application, or during local sewage discharge events[3,18,27−31]. Moreover, increasing experimental and field exposure evidence shows that these ECs can accumulate in organisms through enrichment in symbiotic algae, capture by coral mucus, uptake by filter-feeding organisms, and food chain transmission, and further induce physiological damage such as photosynthetic inhibition, energy metabolism disorder, immunosuppression, and symbiotic system rupture in corals[17,22,23,32−37]. Some studies also point out that when a variety of pollutants exist at the same time, they may have synergistic or mutually reinforcing toxic effects, which aggravate the risk of coral bleaching and weaken the recovery ability of the coral reef ecosystem[38−42]. These findings indicate that ECs not only have conventional exposure risks in coral ecosystems but also may have a profound impact on coral reef habitats by altering energy balance, microbial composition, and stability of the host symbiotic system.
Although the above studies have continued to expand our understanding of the behavior and effects of ECs in coral ecosystems, there are still many scientific problems to be solved. First, existing monitoring data remain primarily concentrated in a few sea areas or single pollutant types, and systematic assessments across different regions, pollution input backgrounds, and coral ecological types are still insufficient[18,43]. Second, there remains a lack of in-depth understanding of the process of migration and transformation of these pollutants between marine media, the mechanisms of absorption and elimination in coral tissues, and their interactions with seawater physical and chemical factors, microbial communities, and symbiotic algae[20,44]. In addition, most current toxicological studies remain focused on single-compound exposure scenarios, while pollutants in real ecosystems tend to be mixed, multi-channel, multi-dose, and long-term exposure, which complicates the prediction of toxic effects and introduces significant uncertainty into risk assessment[22,23,33,45,46]. Therefore, it is urgent to integrate environmental chemistry, ecotoxicology, ecology, and risk science from the perspective of the overall system to sort out the environmental occurrence, distribution patterns, bioaccumulation, toxicity mechanisms, and ecological risk framework of five emerging pollutants in coral reef ecosystems. This review aims to systematically summarize the existing research progress, analyze the behavioral characteristics and impact pathways of various pollutants in coral reef ecosystems, and identify key knowledge gaps to provide a scientific basis for future pollution control, ecological restoration, and coral reef protection strategies.
-
This review systematically searched the ScienceDirect and Web of Science databases and additionally included relevant studies from Google Scholar, using keywords such as 'coral', 'microplastics', 'antibiotics', 'organic UV filters', 'organophosphate esters', 'per- and polyfluoroalkyl substances', 'marine risk', and 'toxicity'. After categorical screening, we summarized the physicochemical properties, spatiotemporal distribution patterns, and pollution trends of the relevant contaminants in coral reef habitats, explored their toxic mechanisms toward corals, and assessed the ecological risks in reef regions.
-
As analytical resolution improves and monitoring efforts expand, microplastic (MP) concentrations in the environment continue to climb. MPs reach the ocean via atmospheric fallout, rain wash-off, surface runoff, and effluent outfalls, as well as through direct inputs from coastal tourism, shipping, marine aquaculture, and fishing activities[47]. An estimated 15–51 trillion plastic particles were floating on global ocean surfaces in 2014[48], composed mainly of polypropylene (PP), low- and high-density polyethylene (LDPE/HDPE), polyvinyl chloride (PVC), polystyrene (PS), and polyethylene terephthalate (PET)[49,50]. In 2021 alone, China discharged 70,707 t of plastic waste into the sea[51]. Their small size facilitates long-range transport and ingestion by virtually all marine taxa, embedding MPs throughout marine food webs and rendering them 'everywhere' in the ocean. Moreover, their high surface-area-to-volume ratio enables adsorption of co-occurring toxicants, such as heavy metals and persistent organic pollutants, potentiating overall toxicity. Once ingested, these contaminant-laden particles negatively affect organismal health, with cascading impacts on every ecological community that depends on marine ecosystems.
Lamb and co-workers surveyed 124,000 reef-building corals on 159 Asia-Pacific reefs and estimated that the habitats currently harbour 1.11 × 1010 macroplastic items (> 50 mm); they forecast a 40% increase to 1.57 × 1010 pieces by 2025[52]. Exposure to macro-plastics dramatically compromises coral immunity, raising disease prevalence from 4% to 89%. Furthermore, when colonies are grouped by architecture—massive, branching, or tabular—micro- and macro-plastics preferentially entangle on structurally complex (tabular and branching) morphotypes, rendering these taxa disproportionately susceptible to plastic-associated pathogenic stress.
A rapidly growing body of work documents the widespread occurrence of MPs in coral reef ecosystems. Table 1 compiles published data on MP abundance and characteristics in Chinese reef waters, sediments, and coral tissues. To date, most studies have focused on the South China Sea, notably the Xisha Islands, Sanya Bay, Hong Kong, and Hainan Island; MPs have been detected in seawater, surficial sediments, coral tissue, and associated fauna. Unless otherwise stated, the following summary is biased toward the South China Sea due to data availability. Two patterns are conspicuous: (1) > 80% of reef-borne MPs are < 2 mm in longest dimension; and (2) particles recovered from coral tissue are predominantly < 1 mm[53], implying preferential ingestion or retention of smaller fragments. Distinct composition and distribution patterns are evident across environmental compartments: PET, cellophane (CP), and polypropylene (PP) dominate the polymer composition, while color characteristics vary systematically among matrices, with black fibers prevailing in seawater and sediments and transparent fibers being preferentially retained in coral tissues[54]. This bias probably reflects the visual-feeding strategy of polyps that target translucent zooplankton, rendering clear fibres 'cryptic' prey.
In addition to size-dependent patterns, recent research indicates that weathered or aged microplastics exhibit altered surface oxidation and structural features that may enhance their interaction with co-occurring toxicants such as additives or degradation byproducts, thereby potentially increasing their ecological impact relative to pristine MPs (e.g., photo-aged MPs show surface oxidation and structural defects that promote leachate release and toxicity)[55,56].
Furthermore, laboratory and field studies demonstrate that microplastic ingestion by corals is influenced by particle size, with species-specific calyx dimensions governing the range of sizes actively ingested, supporting the observation that smaller particles are more often retained in coral tissues than larger fragments[57,58].
Table 1. Concentrations of MPs in seawater, sediments, and coral tissues across selected Chinese reef regions
Sea area MP type Physical property Seawater Sediment Fish Coral tissue Ref. The Xisha Islands, South China Sea PP (53.85%), PS (11.54%),
HDPE (9.62%), EVA (7.69%)Fragment (58.08%) and film (36.54%); transparent (76.92%) and yellow (15.38%) − 0–2,444 items kg−1; average: 682 ± 780
items kg−1− − [59] The Xisha Islands, South China Sea CP (61.13%),
PET (33.49%)Transparent 9.5 ± 3.7 particles L−1 280.9 ± 231.9
particles kg−1− P. damicornis (0.9 ± 0.5), G. fascicularis (1.2 ± 0.6), P. lutea (2.5 ± 1.6) particles cm−2 [53] The Xisha Islands, South China Sea PET, PVDC, PE,
PA, PVC, CPEFiber (79.9%), fragment (13.2%), and particle (5.2%) 1.0–12.2 particles L−1;
average: 6.1 particles L−1− 0–4.7 items g−1 1.0–44.0 items individual−1; 0.02–1.3 items g−1 [60] The Nansha Islands,
South China SeaPP, PE Transparent, blue and green 0.0112–0.149 nm−3; average: 0.0556 ± 0.0355 nm−3 − − − [61] Sanya Bay,
South China SeaPET, CP, PE, PS,
PA, PP, PEFibrous; transparent and black 18.37 ± 2.60 particles L−1 − − A. millepora (0.27 ± 0.26 items/polyp), G. fascicularis (2.32 ± 0.86 items/polyp) [62] Hainan Island in the South China Sea CP, PET, PE, PS, PA Fibrous; black 14.90 ± 7.76 particles L−1 343.04 ± 266.13
particles kg−10.67–3.12 items g−1 P. damicornis (3.68 ± 3.94 particles cm−2), G. fascicularis (5.89 ± 5.15 particles cm−2) [54] Coral Reef Islands in the South China Sea PP, PE, PET, PC, PA Less than 1 mm accounted for 75% Xisha Islands: 60–610 particles kg−1; Nansha Islands: 40–100 particles kg−1; Weizhou Island: 60–90 particles kg−1; Sanya Luhuitou: 50–530 particles kg−1 − − − [63] Hong Kong Bay PE (51.9%), PET (29.3%),
PP, PS, PVCFiber (48.5%),
film (40.8%)− 171.7–223 items kg−1; average: 196.0 ± 39.5 items kg−1 − 189 ± 50 items kg−1 [64] PP stands for polypropylene; PE for polyethylene; PET for polyethylene terephthalate; PS for polystyrene; PVC for polyvinyl chloride; PC for polycarbonate; CP for cellophane; HDPE for high-density polyethylene; EVA for ethylene vinyl acetate polymer; PA for polyamide, nylon. Additionally, Rahman et al. reported significantly higher MP loads adjacent to industrialized/urbanized coasts[51]. Collectively, current evidence indicates that coastal municipal and industrial effluents, tourism, fisheries, and shipping are the principal sources of MPs to South China Sea coral reefs.
Antibiotics (ABs)
-
Over the past decade, field surveys have documented the occurrence of antibiotics in Chinese coral reef systems. Table 2 summarizes reported concentrations in seawater, surficial sediments, and coral tissue. Dissolved antibiotic levels in reef waters range from below detection to 3,302 ng L−1[65,66], while sediment-associated residues span 0.023–525 ng g−1 dry weight (dw)[67]. Coral tissues contain ∑antibiotics at 31.4 ± 24.9 ng g−1 dw. The dominant compounds are enrofloxacin (ENR), ofloxacin (OFX), norfloxacin (NOX), fleroxacin (NFC), demeclocycline (DETM), and erythromycin-H2O, encompassing sulfonamides, fluoroquinolones, macrolides, and chloramphenicols. Coastal municipal sewage and mariculture effluents are the principal sources driving this antibiotic load.
Over the past decade, coral-reef waters of China and Southeast Asia have emerged as global hotspots for antibiotic contamination. Lu et al. analyzed seawater samples collected along 18,000 km of the Chinese coastline; total antibiotic concentrations ranged from 389–3,302 ng L−1[66], with norfloxacin (NFC), roxithromycin (RTM), and ciprofloxacin (CFC) dominating at 1,990, 1,230, and 109 ng L−1, respectively. Du et al. screened 25 antibiotics in the nearshore region of Dalian (Bohai Sea); all target compounds were detected at 22.6–2,402 ng L−1[68], with enrofloxacin, trimethoprim, and sulfamethoxazole present in 100%, 100%, and 90.9% of samples. Zhang et al. first reported antibiotics in surface water of the South China Sea reef tract at 10−2–10 ng L−1[27]; concentrations declined exponentially with distance from shore, peaking adjacent to urban outfalls. Subsequent analyses revealed the same contaminant suite in coral tissue, surface mucus, and reef-associated fish[69,70]. Fourteen antibiotics were quantified in corals (10−2–10−1 ng g−1 dw); coastal colonies were enriched in fluoroquinolones (ΣFQs = 18 ng g−1 dw), whereas offshore colonies contained predominantly sulfonamides (ΣSAs = 23 ng g−1 dw). Seventeen antibiotics were detected in fish muscle (10−2–10−1 ng g−1 ww). Liu et al. examined antibiotics around two typical SCS islands; all targets exhibited pronounced seasonality, with significantly higher loads during the wet season—an effect attributed to increased tourist-derived discharge[28]. Collectively, these studies demonstrate that antibiotics are now ubiquitous contaminants throughout the coral-reef ecosystems of the South China Sea.
Table 2. Concentrations of antibiotics in selected sea areas and coral reef regions of China
Area Matrix ΣABs DF% Max Ref. Bohai Sea, Dalian Seawater 22.6–2,402.4 ng L−1 ENR (100%), TMP (100%), SMX (90.9%) ENR: 2,364.8 ng L−1 [68] Along the coast of Liaodong Peninsula, China Seawater Nd–25.0 ng L−1 SMX (62.5%), ENR (43.8%), OFX (37.5%) OFX: 25.0 ng L−1 [65] Sanya Coral Reef Reserve in the South China Sea Seawater 17.7–44.2 ng L−1 Quinolones (88%), NFC (34.83%), Sulfonamides (6.83%) NFC: 5.07 ng L−1 [17] Coral host 0.54 ± 0.34 ng cm−2 MAR (76%), TIL (32%), SPZ (26%) TIL: 2.315 ng cm−2 Symbiotic algae 0.05 ± 0.05 ng cm−2 STZ (84%), SMX (44%), SCP (40%) SDZ: 0.312 ng cm−2 Beibu Gulf, South China Sea Seawater Winter: Nd–364 ng L−1 FF (96%), TMP (93%), SMX (89%), SDZ (89%) FF: 72.1 ng L−1 [71] Seawater Summer: Nd–70.1 ng L−1 DETM (97%), FF (93%), TMP (90%) FF: 48.6 ng L−1 Northern South China Sea Seawater 43.2–441 ng L−1 – ETM-H2O: 31.6–234 ng L−1 [28] Coral Reef Area in the South China Sea Seawater Nd–6.3 ng L−1 DETM (100%), RTM (100%), CTM (100%) SMX: 6.3 ng L−1 [27] Coral Reef Area in the South China Sea Coral reef fish 0.019–5.3 ng g−1 ww DETM (100%), NOX (100%), SDZ (78%) NOX: 4.7 ng g−1 ww [70] Coral Reef Area in the South China Sea Coastal seawater 4.36 ± 1.71 ng L−1 DETM (100%), RTM (100%), CTM (100%) RTM: 1.01 ± 0.78 ng L−1 [69] Offshore seawater 0.71 ± 0.60 ng L−1 DETM (100%), RTM (100%), CTM (100%) SMZ: 0.26 ± 0.37 ng L−1 Coastal coral 28.2 ± 43.7 ng g−1 dw NOX (100%), DETM (88%), RTM (88%) CIX: 8.40 ± 36.0 ng g−1 dw Offshore coral 31.4 ± 24.9 ng g−1 dw TMP (100%), SDZ (100%), SMZ (100%) SDZ: 9.74 ± 5.72 ng g−1 dw Beibu Gulf Water 1.79–885 ng L−1 NOX (100%), CIX (100%), CTM (100%) FF: 578 ng L−1 [67] Sediment 0.023–525 ng g−1 TMP (100%), NOX (92%), CIX (83%) NOX: 52.5 ng g−1 Feed 0.03–95.4 ng g−1 – TMP: 95.4 ng g−1 Shrimp 1.8–10.55 ng g−1 NOX (87.5%), CTM (62.5%), DETM (50%) SMX: 8.21 ng g−1 Crab 1.66–5.67 ng g−1 NOX (100%), DETM (50%), TMP (37.5%) NOX: 3.14 ng g−1 Oysters 1.81–6.77 ng g−1 DETM (100%), CIX (75%), SPD (75%) DETM: 3.76 ng g−1 Beibu Gulf Water 52.94–77.76 ng L−1 ETM-H2O (100%), NOX (100%), ENX (100%) ETM-H2O: 27.85 ng L−1 [72] Sediment 9.69–15.43 ng g−1 dw NOX (100%), OFX (100%), CIX (100%) NOX: 7.63 ng g−1 dw Marine life 0.68–4.75 ng g−1 ww ENX (92.86%), NOX (82.14%), OFX (64.29%) ENX: 3.15 ng g−1 ww Qinzhou Bay, Guangxi Aquaculture water 0.81–1,386 ng L−1 NOX (86.7%), ENX (86.7%), FF (53.3%) DETM: 1,290 ng L−1 [73] Sediment 1.56–80.6 ng g−1 dw NOX (100%), ENR (100%), CIX (80%) NOX: 3.06 ng g−1 dw Biota 0.17–5.26 ng g−1 ww NOX (100%), TMP (84.6%), SMX (61.5%) NOX: 2.23 ng g−1 dw Feed Nd–5.06 ng g−1 ENR (66.7%), SDZ (66.7%), CIX (66.7%) ENR: 1.86 ng g−1 Bohai Sea, Yellow Sea,
East China Sea and
South China SeaSeawater 389–3,302.3 ng L−1 NFC (100%), RTM (100%), CFC (94.12%) NFC: 1,990 ng L−1 [66] DF: Detection frequency, Nd: Not detected, ENR: Enrofloxacin, TMP: Trimethoprim, FF: Florfenicol, SMX: Sulfamethoxazole, SDZ: Sulfadiazine, DETM: Dirithromycin, ETM-H2O: Erythromycin hydrate, RTM: Roxithromycin, CTM: Clarithromycin, NOX: Norfloxacin, SMZ: Sulfamethazine, CIX: Ciprofloxacin, AZM: Azithromycin, ENX: Enoxacin, OFX: Ofloxacin, NFC: Norfloxacin, CFC: Cefaclor, TIL: Tilmicosin, MAR: Marbofloxacin, SPZ: Sulfaphenazole, STZ: Sulfathiazole, SCP: Sulfachloropyridazine. Organic UV absorber (OUVAs)
-
Since 2016, the contamination baseline of organic UV absorbers (OUVAs) in tropical and subtropical coral reef regions has been progressively uncovered, and their spatial coupling with tourism pressure is systematically illustrated in Table 3. Across seawater, sediments, and coral tissues, concentrations consistently follow an 'onshore-high, offshore-low, peak-in-peak-season' gradient; the maximum concentration of the same compound can differ by 1–2 orders of magnitude among different coral reef areas, implying biodilution or biomagnification processes. The first Hawaii data introduced the 'µg L−1 alert level'[74]; at beaches in the U.S. Virgin Islands and Waikīkī, seawater BP-3 reached 1.4 mg L−1—already exceeding the 24-h LC50 (139 µg L−1) later determined for coral larvae—thereby establishing the first field-to-toxicity overlap. Follow-up campaigns in the Pearl River Estuary and Oʻahu Island normalized the 'ppb' range[75,76], rainy-season BP-3 in the estuary peaked at 31.8 ng L−1, with > 20% of coral-tissue samples exceeding the teratogenic threshold, whereas Waikīkī BP-3 dropped to 136 ng L−1 but coral tissue contained up to 241 ng g−1 dw, evidencing 1–2-order bioconcentration and a shift toward octisalate (OS) and homosalate (HMS) as dominant tissue residues. Temperate, low-tourism estuaries further showed filter-feeding bivalves accumulating BP-3 to 118 ng g−1[77], confirming that sunscreen can be discharged into the ocean from wastewater and accumulate in marine organisms.
Entering the 2020s, the research focus moved to the South China Sea. Kish Island (Persian Gulf) linked visitor numbers to bleaching incidence[78], extrapolating the concentration–effect relationship to the community scale. Pei et al. quantified Σ10OUVAs simultaneously in nearshore seawater (102 ng L−1) and coral tissue (2.31 µg g−1 dw) using an onshore–offshore transect, and for the first time reported log bioaccumulation factors (BAFs) of 1.22–5.07, demonstrating that offshore corals can exhibit higher enrichment because of physiological differences[31]. Two latest Hainan Island surveys pushed spatial resolution to the island scale[43,79], BP-3 (35–76 ng L−1) and 4-MBC (38–61 ng L−1) in Sanya tourist hotspots were 2–3-fold higher than background, while 65% of coral samples from Luhuitou reef exceeded the larval bleaching threshold for BP-3 and 99% of macroalgae exceeded the growth-inhibition threshold for ODPABA. By incorporating primary producers into risk endpoints, these studies reveal that OUVFs can amplify ecological impacts through benthic–pelagic coupling.
Synthesis of Table 3 yields four overarching conclusions: (1) background levels of reef-associated OUVAs have settled into a 'ng L−1–ng g−1' window globally, but can spike transiently to 'µg L−1–µg g−1' during tourist peaks or monsoonal pulses; (2) BP-3, BP-8, HMS, and OS consistently dominate the contaminant profile, with environmental maxima overlapping published toxicity thresholds, signifying tangible risk; (3) coral tissues accumulate target compounds 10–100-fold above ambient seawater, and both morphological traits (polyp density > smooth surface) and seasonal hydrology (wet > dry) are key drivers of bioaccumulation factors (BAFs); (4) macroalgae and bivalves also concentrate OUVAs, indicating that the hazard propagates vertically from the benthos throughout the reef food web. These quantitative baselines provide an essential foundation for formulating regional emission-control strategies.
Table 3. Concentrations of OUVAs in some coral reef environments around the world
Area Matrix OUVAs DF% ΣOUVAs Max Ref. Virgin Islands Seawater BP-3 − − BP-3: 75–1,400 µg L−1 [74] Hawaii Seawater BP-3 − − BP-3: 0.8–19.2 µg L−1 [74] Pearl River Estuary, South China Sea Seawater 7 types including
BP-3, BP-8, OC, etc.BP-3 (100%) − BP-3: 13.2–31.7 ng L−1 [75] Sediment BP-3 (100%) − BP-3: 4.2–17.0 ng g−1 dw Coral BP-3 > 65%,
BP-8 > 65%− BP-3: 1–38.4 (31.8 ± 8.6) ng g−1 ww;
BP-8: 1.3–19.9 (24.7 ± 10.6) ng g−1 wwOahu Island, Hawaii Seawater 13 types including
BP-3, HMS, OS, etc.BP-3 (100%), OC (95%), HMS (65%), OS (63%) 6.8–745.3 ng L−1 HMS: 53.0–625.7 ng L−1;
OS: 33.1–96.0 ng L−1[76] Sediment HMS (91%), OS (88%),
OC (65%), BP-3 (56%)0.38–67.37 ng g−1 dw HMS: 0.08–38.54 (25.85 ± 13.72) ng g−1 dw;
OC: 0.16–19.64 (10.52 ± 7.26) ng g−1 dwCoral tissue HMS (100%), OS (100%), OC (96%), BP-3 (82%) 666.2–1,178.4 ng g−1 dw OC: 210.4–527.1 (404.1 ± 75.3) ng g−1 dw;
HMS: 188.7–441.1 (368.5 ± 51.5) ng g−1 dwCoral Reef Area, South China Sea Coral 10 types including
BP-3, EHMC, OCR, etc.4-MBC, OD-PABA, EHMC and UV-327 > 80% 2.31 ± 1.96 μg g−1 dw − [31] Hainan Island and the coastal waters of the South China Sea Seawater 14 types including
OC, PABA, BP-3, etc.All 100% 77.7–313.9 ng L−1 BP-3: 53.2 ng L−1 [80] Sediment All 100% 52.3–110.2 ng g−1 dw BP-3: 17.0 ng g−1 dw Hainan Island, South China Sea Seawater 14 types including
BP-3, 4-MBC, BP-8, etc.All 100% 174.5–343.8 ng L−1 BP-3: 35.3–75.6 ng L−1 [43] Sediment All 100% 84.0–175.8 ng g−1 dw BP-3: 13.2–25.9 ng g−1 dw Coral All 100% 32.1–91.8 ng g−1 dw BP-3: 4.5–21.3 ng g−1 dw Sanya Luhuitou Coral tissue 8 types including BP-3, OC, OD-PABA, etc. All ≥ 83.3% 7.0–35.8 (25.1 ± 10.9) ng g−1 dw OC: 2.7–31.8 ng g−1 dw [79] Macroalgae All ≥ 90.9% 15.6 ± 6.4 ng g−1 dw − DF: Detection frequency, BP-3: Benzophenone-3, BP-8: Benzophenone-8, OC: Octocrylene, HMS: Homosalate, OS: Octyl Salicylate, EHMC: Ethylhexyl Methoxycinnamate, OCR: Octocrylene, PABA: 4-Aminobenzoic acid, 4-MBC: 4-Methylbenzylidene Camphor, OD-PABA: Octyl Dimethyl-p-Aminobenzoic acid. Organophosphate esters (OPEs)
-
The ocean is widely recognized as the ultimate sink for anthropogenic contaminants. As land-derived pollutants, organophosphate esters (OPEs) reach the marine environment through atmospheric transport and deposition, surface runoff, riverine discharge, and ocean-current advection. Previous studies have documented the accumulation of MPs and OUVAs in coral ecosystems, and reef habitats are now known to contain appreciable quantities of OPEs. Their high bioaccumulation potential translates into elevated ecological risk for coral reef regions. Recent surveys conducted in the South China Sea have detected OPEs in both reef fish and coral tissue; strikingly, detection frequencies in coral tissue are generally lower than those in ambient seawater[19,81,82]. Fish concentrations range from 38.7 to 2,090 ng g−1 lipid weight (lw), with a mean of 420 ± 491 ng g−1 lw, with tributyl phosphate (TBP) and tris(1-chloro-2-propyl) phosphate (TCPP) being the dominant congeners[83]. Coral tissues contain 197 ± 412 ng g−1 dry weight (dw), whereas dissolved OPEs in seawater average 1.56 µg L−1—comparable to, or slightly below, global oceanic baseline levels. Yan et al. further observed that seawater OPE concentrations decrease with increasing distance from the mainland; however, no statistically significant difference was found in OPE burdens between coastal and offshore coral colonies, underscoring the complexity of bioaccumulation processes in reef organisms[18].
In recent years, OPEs, as a class of emerging organic pollutants, have been frequently detected in the global marine environment, attracting widespread academic attention. Table 4 systematically summarizes the contamination characteristics of OPEs in various marine regions and coral-reef ecosystems worldwide, covers multiple matrices such as seawater, sediment, marine organisms, and coral tissues, and reveals significant differences in their spatial distribution, composition profiles, and potential ecological risks.
Seawater concentrations of OPEs exhibit pronounced regional gradients that mirror the intensity of anthropogenic activity. In nearshore areas such as the South China Sea, Tokyo Bay, the Pearl River Delta (PRD), and the Yellow River mouth, ΣOPEs range from 1.25 to 1,790 ng L−1[84]; maxima in the PRD and Yellow River estuary reach 1,790 and 1,720 ng L−1, respectively, with TEP, TCPP and TPPO dominating, signatures consistent with strong manufacturing and construction emissions. By contrast, Arctic surface waters contain 2.71 to 8.38 ng L−1[83]; nevertheless, the detection of chlorinated congeners such as TDCIPP confirms long-range atmospheric and oceanic transport, underscoring the global dispersal potential of OPEs.
Table 4. OPEs pollution in some sea areas and coral reefs around the world
Area Matrix OPEs category ΣOPEs OPEs (max) Ref. South China Sea Seawater 14 types including TEP, TCPP, TPPO, etc. 1.25–147 ng L−1 TEP: 53.1 ng L−1 [84] Tokyo Bay Seawater 14 types including TEP, TCPP, TPPO, etc. 107–284 ng L−1 TCPP: 126 ng L−1 Pearl River Delta Seawater 14 types including TEP, TCPP, TPPO, etc. 14.9–1790 ng L−1 TPPO: 759 ng L−1 Yellow River Estuary Seawater 14 types including TEP, TCPP, TPPO, etc. 253–1,720 ng L−1 TEP: 555 ng L−1 Arctic Seawater 9 types including TCIPP, TDCIPP, TPHP, etc. 2.71–8.38 ng L−1 TDCIPP: 8.38 ng L−1 [83] Western Mediterranean Fish 13 types including TDClPP, TCEP, TPPO, etc. 0.38–73.4 ng g−1 ww TDClPP: 27.2 ng g−1 ww [85] Liaodong Bay,
Bohai SeaMarine life 7 types including TCEP, TEHP, TCIPP, etc. 2.60–776 ng g−1 ww TCEP: 281 ± 234 ng g−1 ww [86] The Great Lakes of North America Fish 22 types including TBOEP, TCIPP, TEP, etc. 9.6–74 ng g−1 ww TBOEP: 50.7 ± 1.4 ng g−1 ww [87] Coral area of South China Sea Seawater 11 types including TCIPP, TBOEP, TNBP, etc. 0.735–4.949 ng L−1 TCIPP: 1.551 ng L−1 [18] Coral tissue 128.05–924.29 ng g−1 dw TBOEP: 601.48 ng g−1 dw Coral mucus 29.62–1,798.8 ng g−1 dw TNBP: 802.98 ng g−1 dw Xisha Islands in the South China Sea Seawater and
marine life11 types including TCIPP, TBOEP, TCEP, etc. Seawater: 1.52 ± 0.33 ng L−1 TCIPP [19] Coral fish 2,227 ± 2,062 ng g−1 lw Benthic mollusc 1,024 ± 606 ng g lw Coral 1,800 ± 1,344 ng g−1 lw TBOEP Nansha Islands,
South China SeaCoral reef fish 9 types including TBEP, TCEP, TCPP, etc. 38.7–2,090 ng g−1 lw EDP: 795.24 ng g−1 lw [82] Zhushan Bay,
Taihu Lake, ChinaLake water 11 types including TCIPP, EHDPP, TEHP, etc. 1,150 ± 232 ng L−1 TCIPP: 835 ± 168 ng L−1 [88] Sediment 40.4 ± 9.24 ng g−1 dw TCIPP: 18.6 ± 3.65 ng g−1 dw Plankton 10.758 ± 0.534 ng g−1 ww EHDPP: 5.223 ± 0.186 ng g−1 ww Invertebrate 21.754 ± 1.06 ng g−1 ww TEHP: 6.910 ± 0.92 ng g−1 ww Fish 15.739 ± 0.611 ng g−1 ww TCIPP: 12.441 ± 0.54 ng g−1 ww TCIPP: Tris(2-chloroisopropyl)phosphate, TBOEP: Tris(2-butoxyethyl)phosphate, TNBP: tri-n-butyl phosphate, TCEP: Tris(2-chloroethyl)phosphate, TBEP: tris(2-butoxyethyl)phosphate, TCEP: Tris(2-chloroethyl)phosphate, TCPP: Tris(1-chloro-2-propyl)phosphate, EDP: 2-ethylhexyl diphenyl phosphate, EHDPP: 2-ethylhexyl diphenyl phosphate, TEHP: tris(2-ethylhexyl)phosphate, TDCIPP: tris(chloroisopropyl)phosphate, TPHP: tris(phenyl)phosphate, TPPO: triphenylphosphine oxide, TEP: triethyl phosphate. Biota show species- and ecosystem-specific bioaccumulation of OPEs. Small pelagic fish from the western Mediterranean exhibited muscle concentrations up to 73.4 ng g−1 ww, higher than their predators, indicating that trophic transfer is not the dominant exposure route. In contrast, TCEP in marine organisms from Liaodong Bay, Bohai Sea, reached 281 ng g−1 ww, with lipid content identified as the key factor governing bioconcentration. Within coral-reef systems, elevated OPEs are ubiquitous in coral tissue, mucus, and reef fish across multiple South China Sea sites: TBOEP in Xisha Islands coral tissue peaked at 1,800 ng g−1 lw, while EDP in Nansha Islands reef fish attained 795 ng g−1 lw, underscoring severe regional contamination. Coral mucus plays a pivotal role in OPEs enrichment, and pollutant levels correlate positively with human-activity intensity, highlighting the pronounced ecological risk posed by OPEs in nearshore coral ecosystems.
In summary, OPEs exhibit a clear 'nearshore high, offshore low' spatial pattern across the global ocean and display pronounced bioaccumulation and ecological risk in sensitive ecosystems such as coral reefs. Future efforts should prioritize research on the environmental fate, toxic mechanisms, and synergistic interactions of OPEs within marine food webs to comprehensively evaluate their long-term impacts on ocean ecosystems and human health.
Per- and polyfluoroalkyl substances (PFASs)
-
In recent years, the global detection of PFAS has intensified concern over their contamination of marine environments. Owing to their persistence, bioaccumulative capacity, and latent toxicity, PFAS are now recognized as a major threat to ocean-ecosystem health. A synthesis of current data, compiled in Table 5, summarizes PFAS concentrations from multiple ocean basins and representative coral reef systems, revealing pronounced regional heterogeneity, matrix-specific signatures, and marked seasonal dynamics.
Table 5. Pollution status of PFAS in several sea areas and typical coral ecosystems around the world
Area Matrix Classification PFAS of the top three DF% ΣPFAS Max Ref. South China Sea Egret Heart PFDoDA, PFTrDA and PFUnDA
(DF = 100%)0.25–9.01 ng g−1 ww PFOS: 5.04 ng g−1 ww [89] Liver PFDoDA, PFTrDA and PFUnDA
(DF = 100%)1.20–17.1 ng g−1 ww PFOS: 11.4 ng g−1 ww Kidney PFDoDA, PFTrDA and PFUnDA
(DF = 100%)1.30–33.7 ng g−1 ww PFTrDA: 11.4 ng g−1 ww Hainan Island Seawater Rainy season PFOA (97.5%), PFOS (91.5%) and
PFHpA (78.5%)0.212–11.963 ng L−1 PFOS: 8.023 ng L−1 [90] Dry season PFOA (100%), PFBA (100%) and
PFOS (99.5%)0.263–20.330 ng L−1 PFOS: 12.867 ng L−1 Biscayne Bay Seawater Nearshore surface water PFBA, PFPeA and PFHxA (DF > 94 %) 29.52 ± 15.25 ng L−1 PFOS: 19.84 ng L−1 [91] Nearshore bottom water PFBA, PFPeA and PFHxA (DF > 94 %) 21.45 ± 7.85 ng L−1 Offshore surface water PFHpA, PFOA and PFNA (DF > 94 %) 5.17 ± 2.66 ng L−1 PFOS: 3.30 ng L−1 Offshore bottom water PFHpA, PFOA and PFNA (DF > 94 %) 2.20 ± 1.60 ng L−1 Biscayne Bay Marine life Thunnus atlanticus DF > 80% (6:2 FTS, PFNA and PFOS) 0.15–3.40 ng g−1 ww PFTrDA: 0.14 ±
0.16 ng g−1 ww[93] Lobster DF > 80% (PFBA, FHxSA and PFOS) 0.37–5.15 ng g−1 ww N-MeFOSAA:
1.62 ± 0.45 ng g−1 wwNew York Bay and Bahamas Shark Muscle tissue PFTrDA (95%), PFOA (92%), PFBA (92%) 1.1–58.5 ng g−1 PFOA: 28.6 ng g−1 [94] Florida Surface water – PFOA (94%), PFBS (65%), PFHxA (61%) 1–3048 ng L−1 PFOS: 1,135 ng L−1 [92] Mangrove estuary in Zhangzhou, Fujian Surface water, aquatic organisms Surface water PFPeA, PFOA and PFBS (DF = 100%) 5.27–12.54 ng L−1 PFPeA: 3.65 ng L−1 [96] Fish PFBA, PFHxA and PFHpA (DF = 100%) 0.45–17.67 ng g−1 ww PFBA: 7.68 ng g−1 ww Shrimp – 3.20 ng g−1 ww – Crab – 5.29 ng g−1 ww – Indian Ocean Seabird egg – PFOS, PFUdA and PFNA (DF = 100%) 1.9–10.3 ng g−1 ww PFOS: 3.8 ng g−1 ww [97] The Red Sea Seawater – PFOA (96.4%), 6:2 FTS (76.0%) and PFHpA (75.0%) < LOQ–956 ng L−1 6:2 FTS: 450 ng L−1 [95] South China Sea Coral Dry season PFOA, PFNA and PFDoDA (DF = 100%) 0.84–26.05 ng g−1 dw PFHxS: 18.9 ng g−1 dw [20] Rainy season PFOA, PFHxS and PFOS (DF = 100%) 2.05–20.31 ng g−1 dw PFHxS: 13.5 ng g−1 dw South China Sea Coral reef fish – PFOS, PFUnDA and PFTrDA (DF > 94%) 0.38–20.43 ng g−1 ww PFOS: 20.23 ng g−1 ww [26] Beibu Gulf Seawater Summer PFBA, PFPeA, PFBS (DF = 100%) 1,609–4,727 pg L−1 PFBA: 1,771 pg L−1 [98] Winter PFHxA, PFHpA, PFHxS (DF = 100%) 610–4,920 pg L−1 PFPeA: 1,968 pg L−1 DF: Detection frequency, PFDoDA: Perfluorododecanoic acid, PFTrDA: Perfluorotridecanoic acid, PFUnDA: Perfluoroundecanoic acid, PFOS: Perfluorooctane sulfonate, PFOA: Perfluorooctanoic acid, PFHpA: Perfluoroheptanoic acid, PFBA:Perfluorobutanoic acid, PFPeA:Perfluoropentanoic acid, PFHxA: Perfluorohexanoic acid, PFNA: Perfluorononanoic acid, 6:2 FTS: Sodium 1H,1H,2H,2H-perfluoro-1-octanesulfonate, N-MeFOSAA: N-methylperfluoro-1-octanesulfonamidoacetic acid, FHxSA: Perfluoro-1-hexanesulfonamide, PFBS (Perfluorobutane sulfonate, PFUdA: Perfluoro-n-undecanoic acid, PFHxS: Perfluorohexane sulfonate. PFAS contamination is most pronounced in tropical and subtropical waters. Extensive surveys across the South China Sea (SCS) and adjacent areas report ubiquitous detection in seawater, seabirds, fish, and corals, with perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) as the dominant congeners[20,26,89,90]. Seawater PFOS at Hainan Island, for example, peaks at 12.9 ng L−1 during the rainy season[90]—almost triple the dry-season value—highlighting the importance of precipitation-driven runoff. In biota, SCS little egrets exhibit kidney PFAS burdens significantly higher than those in liver or heart, indicating the kidney as a primary site of accumulation and metabolism[89]. Coral–algal holobionts also contain measurable residues; PFHxS and PFOA predominate in SCS coral samples and fluctuate markedly between monsoonal phases[20]. Such temporal variability suggests that PFAS can perturb the coral–algal symbiosis and, consequently, destabilize the entire reef ecosystem.
Along US coasts, point sources—municipal wastewater outfalls, airports, and military installations—drive PFAS loading[91,92]. In Biscayne Bay, Florida, surface-water PFOS reaches 19.8 ng L−1[91], and tuna as well as spiny lobster contain measurable PFAS burdens[93]; PFOS and 6:2 FTS correlate significantly with lysosomal membrane destabilization, implying measurable cytotoxicity. Muscle of sharks from New York Bay and the Bahamas shows > 90% detection frequency for PFTrDA, PFOA, and PFBA, while long-chain homologues (C ≥ 10) increase with trophic position (δ15N) and total mercury[94], evidencing biomagnification potential.
Even remote seas—the Red Sea, Beibu Gulf, and the Indian Ocean—are not exempt. Red Sea surface water carries up to 450 ng L−1 of 6:2 FTS[95], implicating urban and industrial effluents; the Beibu Gulf exhibits current-driven seasonality, with PFBA and PFPeA dominating year-round but distinct winter vs summer assemblages. On isolated Indian Ocean atolls, seabird eggs contain PFOS, PFUdA, and PFNA[26], indicating that marine debris and long-range atmospheric transport can inject PFAS into the most secluded ecosystems.
In summary, PFAS are now ubiquitous across multiple marine basins and coral ecosystems; their levels are governed by anthropogenic intensity, hydro-meteorological drivers, and species-specific bioaccumulation capacity. Future efforts must strengthen long-term monitoring of representative waters, quantify ecological risks to coral symbioses and marine food webs, and promote inter-regional coordination on source control to mitigate the continuing pressure of PFAS on oceanic environments.
-
MPs are now dispersed throughout coral-reef environments. Recent work shows that reef-building corals readily ingest and absorb them: Hall et al. exposed Dipsastrea pallida to 10 µm–2 mm blue polypropylene beads (0.4 g L−1) and recorded ingestion by > 20% of colonies[99]. Particles were found trapped on polyp surfaces, embedded in tissues, and even incorporated into the skeleton, demonstrating that corals act as MP 'hot-spots' [25,100]. Reichert et al. further revealed genus-specific uptake modes: large-polyp species (Acropora, Pocillopora) capture MPs via direct phagocytosis, whereas small-polyp forms such as Porites lutea employ mucus adhesion[46]. Regardless of route, MP-exposed tissues display epithelial lesions, necrosis, and bleaching[101]. Fibrous MPs physically lacerate the tissue, creating entry points for viruses and ciliated protozoa that can fracture the skeleton.
Mechanistically, MPs trigger ROS accumulation, disrupt membranes, induce DNA damage, restrict nitrogen supply, and impair energy metabolism, weakening immunity and promoting symbiont expulsion[42]. PVC fragments carry Rhodobacterales bacteria—known opportunistic pathogens—into coral tissue, fueling disease outbreaks that reduce species richness and erode reef function[102]. MPs also interfere with nutrient cycling: they decrease light availability, curtail photosynthesis, and, once ingested, elicit satiation cues that lower zooplankton capture, cutting the energy budget for growth and reproduction[58, 103,104]. Coral degradation diminishes habitat complexity, drives down associated biodiversity, and can ultimately precipitate ecosystem collapse.
Antibiotics (ABs)
-
Over the past decade, the impact of antibiotics—as ECs—on coral health has gained increasing attention. Sweet et al. first demonstrated that ciprofloxacin reduced the bacterial abundance of Acropora muricata by > 99% within 8 h, disrupted community structure, and allowed potential pathogens such as Clostridium and Vibrio to displace native symbionts, paving the way for disease outbreaks[105]. Field surveys further revealed that 19 antibiotics were detectable in South China Sea surface waters (ΣABs = 17.69–44.22 ng L−1). Host tissues of Galaxea fascicularis and Pocillopora damicornis accumulated 11 of these compounds, and their Symbiodiniaceae symbionts contained five, with elevated ΣABs correlating positively with enhanced total antioxidant capacity in P. damicornis but abnormally increased symbiont density in G. fascicularis[17], indicating species-specific accumulation patterns and physiological responses among coral-symbiont pairings.
At the morphological level, antibiotic exposure induced polyp swelling and other phenotypic anomalies in both symbiotic and aposymbiotic corals. During recovery, symbiotic colonies rapidly re-stabilized their microbiome via symbiont-mediated regulation, whereas aposymbiotic colonies showed high inter-individual variability[106], demonstrating that symbiotic status is a key determinant of microbial resilience.
Mechanistic studies further show that roxithromycin triggers dose-dependent apoptosis, oxidative defense, and symbiont-health-related gene reprogramming in Acropora cervicornis[17]. Tetracycline inhibits nitrogen or carbon metabolism in symbionts, blocking nutrient supply to the host and creating nitrogen competition and carbon shortage that ultimately disrupts the symbiosis and intensifies bleaching risk[107]. Acute exposure assays also reveal that doxycycline hyclate (DOX, 1–10 mg L−1) suppresses protein synthesis in soft corals, activates oxidative-stress and lipid-peroxidation pathways, and weakens immune and detoxification functions, potentially collapsing the defense system and inducing bleaching[108].
Collectively, antibiotics pose multi-level threats to coral health—from molecules to ecosystems—by disrupting microbial communities, damaging host–symbiont metabolic coupling, and eliciting oxidative stress.
Others
-
Danovaro et al. first hypothesized that organic UV absorbers (OUVAs) could exert toxicological effects on corals[109], sparking a wave of exposure assays and field surveys that continues today. Mortality, growth inhibition, loss of photosynthetic efficiency, and bleaching are the universal endpoints. For BP-3, an 8-h pulse induces bleaching at every tested concentration (1–0.00001 mM); symbiont density and chlorophyll-a decline in parallel, transmission electron microscopy reveals epidermal necrosis and autophagosome formation, and bleaching severity increases dose-dependently[74]. Mitchelmore et al. confirmed that chlorophyll activity is significantly suppressed within the same time frame[110]. Recent work shows that benzophenone-type filters can kill coral larvae within 10 d, prompting a 'moderate-to-high' health-risk ranking[111]. Octocrylene (OC), owing to its high lipophilicity, accumulates in coral tissue, binds to lipid droplets, and disrupts mitochondrial function, culminating in energy-metabolism disorder[112].
For OPEs, most data derive from fish models: exposure of zebrafish embryos, larvae, and adults reveals endocrine-disrupting, reproductive, neuro-, and hepatotoxic potential[113−115]. The detection of OPEs in coral tissue confirms that similar risks extend to reef-building corals[18,19].
Direct toxicological information for PFASs remains scarce. Bednarz et al. first provided evidence that the co-exposure of PFOS and heat stress significantly exacerbates oxidative damage in corals[23]. Because Photosystem II (PSII) proteins of symbionts are highly ROS-sensitive, PFAS accumulation may fragment PSII, trigger symbiont loss, and manifest as bleaching[20]. A 24-h, 1 mg L−1 PFOS pulse recently suppressed larval uptake of Symbiodiniaceae and perturbed lipid homeostasis in Acropora millepora[22]. Other evidence indicates that PFAS can cause protein damage throughout the entire organism of corals, disrupt the mutually beneficial host-symbiont relationship, and trigger bleaching[116,117].
Synergistic and carrier-mediated effects of EC mixtures
-
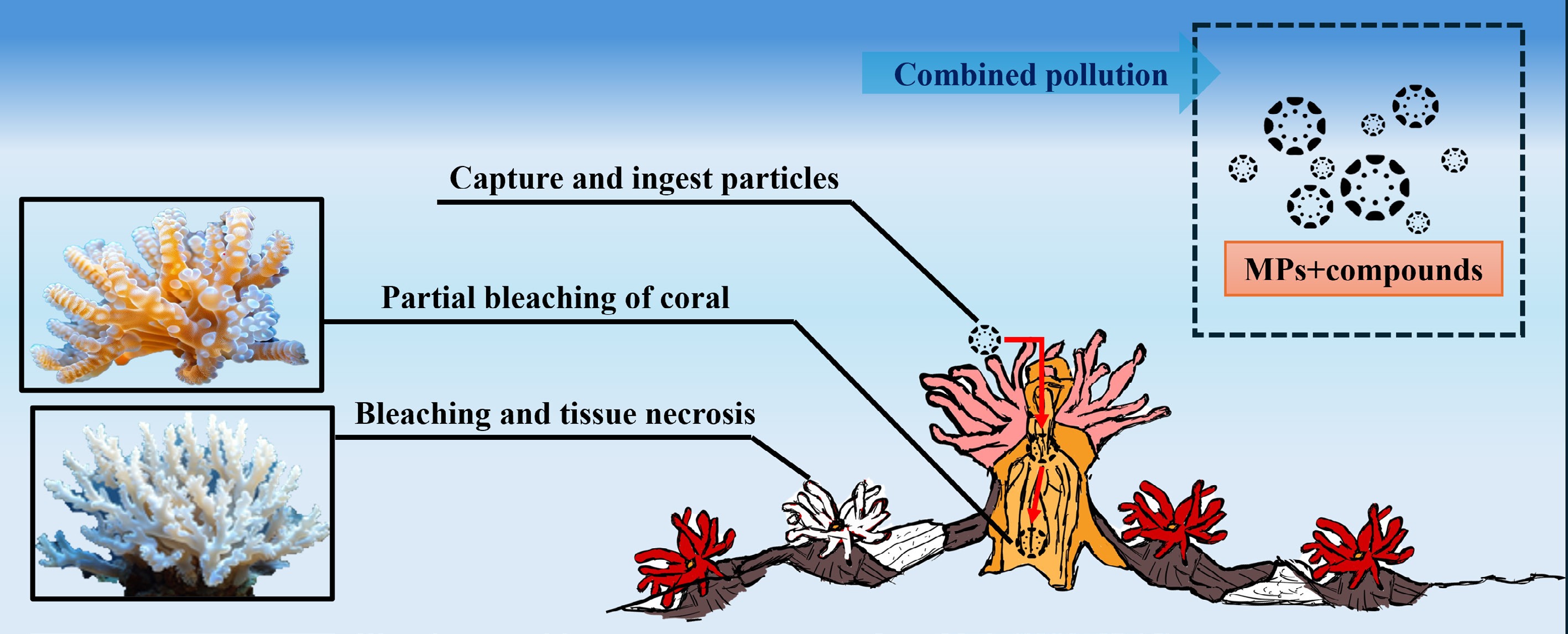
Owing to their high surface-area-to-volume ratio and hydrophobicity, MPs act as a 'Trojan horse' for multiple ECs in coral-reef systems. Experimental evidence demonstrates that MPs not only are ingested or adhered to by corals, but also concentrate and deliver co-occurring pollutants such as PFAS, heavy metals (Cu2+, Cr3+), and antibiotics, thereby significantly amplifying combined toxicity[118]. Adsorption of PFAS to MPs can raise local exposure concentrations by one order of magnitude and generate synergistic effects with plastic additives[119]. Figure 1 depicts the process of coral bleaching caused by compound pollution. In a 7-day co-exposure assay, polyethylene MPs plus Cu2+ (≥ 50 µg L−1) caused greater oxidative stress, lower symbiont density, and higher tissue-Cu accumulation in Porites cylindrica than either stressor alone[38], indicating that MPs function as 'ion carriers' that intensify metal toxicity. Similarly, combined PE-MP and Cr(III) suppressed glycolysis and the pentose-phosphate pathway, decreased the NAD+/NADH ratio, and activated caspase cascades, inducing energetic failure and apoptosis in coral symbionts[40]. Notably, low-level Cr(III) toxicity was transiently alleviated by MP dilution adsorption, but further Cr(III) increases under high-MP backgrounds produced synergistic collapse, revealing that 'carrier vs dose' interactions dictate toxicological outcomes.
For antibiotics, MPs enhance their bioavailability in coral tissues; together they trigger ROS bursts, inhibit host–symbiont nitrogen or carbon metabolism[120,121] and block nutrient exchange[122], leading to DNA damage, immune dysregulation, and reduced microbial stability[121, 123], ultimately manifesting as symbiont expulsion and elevated bleaching risk.
Overall, MPs, through a three-stage mechanism of 'concentration-transportation-synergy', transform originally dispersed low-dose ECs into locally high-dose, multi-stress mixtures, posing a systemic threat to the entire coral-symbiotic algae and microbial community, from the subcellular to the community level.
Impacts of ECs on coral-Symbiodiniaceae holobiont
-
As an emerging contaminant, antibiotics are now systematically destabilizing the coral-algal holobiont through multiple, hierarchical pathways[123]. At the community level, therapeutic concentrations restructure the surface-mucus microbiome: the bacterial assemblage of Acropora poculata shifts markedly after exposure[106], while isolates from Fungia fungites mucus acquire heritable resistance[29]. Functionally, the drugs selectively suppress or eliminate keystone taxa such as Rhodobacteraceae and Neptuniibacte[121], interrupting nitrogen and sulfur cycling and cutting off essential amino-acid supply lines. The resulting metabolic bottleneck lowers whole-holobiont respiration and frees niches for opportunistic pathogens (e.g., Vibrio spp.), forcing the host immune system to operate continuously at high load. Energy is thereby diverted from growth and repair to defense, precipitating a rapid decline in overall fitness. At the cellular scale, tilmicosin, sulfapyridine, ofloxacin, and lincomycin hydrochloride each impair the antioxidant machinery of Symbiodiniaceae. When combined with thermal stress, this suppression amplifies ROS formation and apoptotic signaling, significantly lowering the coral's threshold for subsequent environmental shocks[123].
Ecosystem-level impacts of ECs on coral reefs
-
ECs are now undermining coral-reef ecosystem functions through a three-tier mechanism of 'direct toxicity–carrier effect–synergistic stress'. (1) MPs are actively ingested or adsorbed onto coral surfaces, causing tissue necrosis, excessive mucus secretion, and depressed photosynthetic efficiency; simultaneously, they accumulate heavy metals, polycyclic aromatic hydrocarbons, and plastic additives, markedly increasing combined toxicity[101, 124]. (2) PFAS, owing to their extreme chemical stability and high protein affinity, accumulate in coral tissues, inhibit Photosystem II activity of Symbiodiniaceae, disturb host lipid metabolism, and become more adsorbent and bioavailable as aging MPs introduce oxygen-containing surface groups[125,126]. (3) Antibiotics selectively eliminate keystone nitrogen or sulfur-cycling bacteria (e.g., Rhodobacteraceae), cutting off essential amino acid and vitamin supplies while stimulating opportunistic pathogens (Vibrio spp.); this forces continuous immune activation and energy reallocation, culminating in symbiont apoptosis, microbiome dysbiosis, and reduced stress tolerance[17, 105, 123]. In addition, OUVAs such as oxybenzone cause photoinhibition and DNA damage at ng L−1 levels and decrease larval settlement; it is estimated that 10% of world reefs annually face an extra bleaching risk from tourist sunscreens[74, 127]. Importantly, these pollutants overlap with climatic stressors (warming, acidification) to amplify oxidative stress, block energy metabolism, and suppress tissue repair, markedly narrowing coral’s niche width and recovery potential[128].
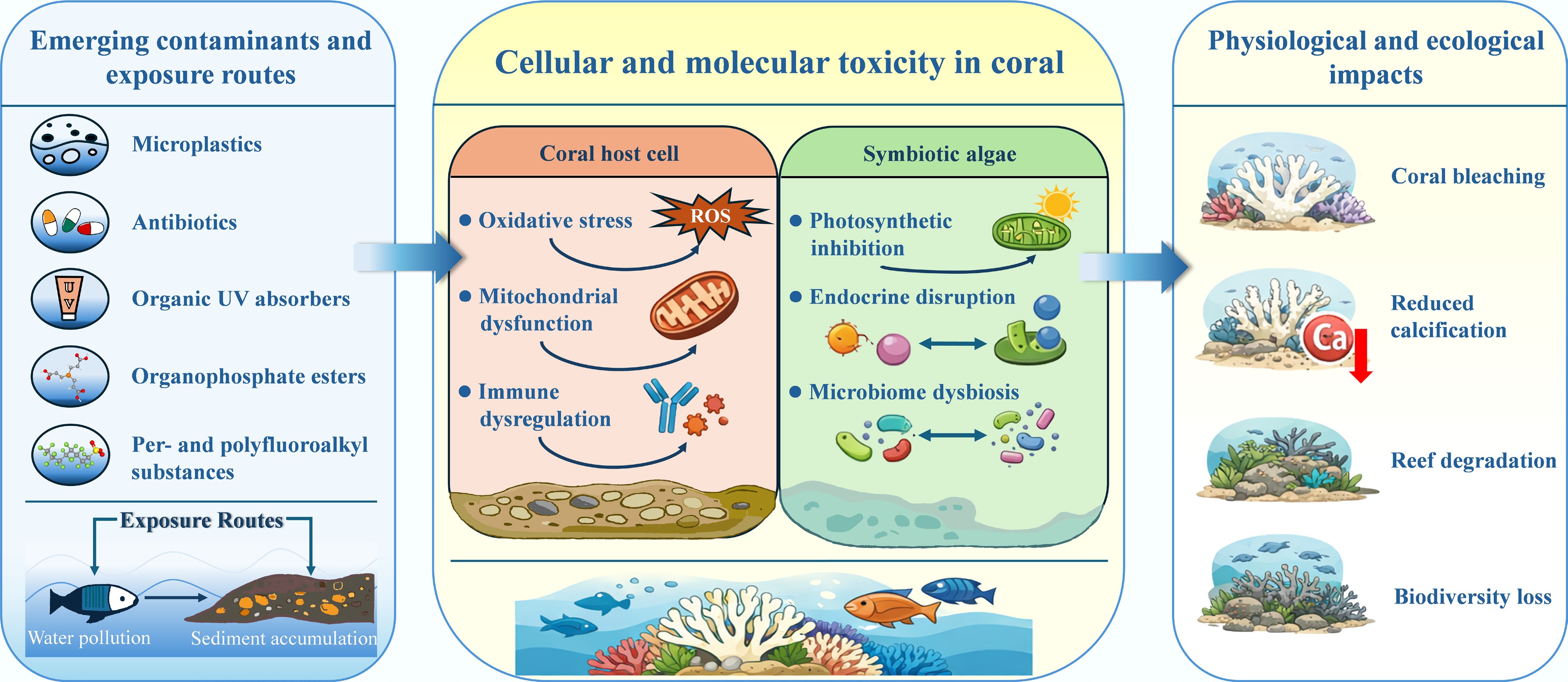
In general, ECs have gone beyond the traditional 'single toxicity' paradigm and imposed long-term, cumulative, and cross-generational multi-level pressure on coral reefs. The toxic mechanisms of ECs in coral reef ecosystems are summarized in Fig. 2. Therefore, urgent and systematic research on source apportionment, threshold derivation, and comprehensive risk assessment is needed to support accurate coral reef protection policies.
-
Overall, ECs are now ubiquitously distributed across coral reef ecosystems, occurring in seawater, sediments, coral tissues, and associated biota, with pronounced spatial, temporal, and compositional heterogeneity. Across the five major contaminant classes considered in this review, concentrations generally follow the pattern of nearshore > offshore reefs, highlighting the dominant influence of land-based anthropogenic inputs such as wastewater discharge, urban runoff, tourism, aquaculture, and maritime transport.
Spatially, reefs located in densely populated and industrialized coastal regions—particularly in the South China Sea, Southeast Asia, the Mediterranean Sea, and parts of the Caribbean—consistently exhibit elevated contaminant burdens, whereas remote reefs generally show lower background levels, albeit still subject to long-range atmospheric and oceanic transport. Temporally, higher concentrations are commonly observed during wet or monsoonal seasons, reflecting enhanced terrestrial runoff and episodic pollution pulses associated with storm events and tourism peaks.
In terms of composition, distinct source-related signatures are evident. MPs are dominated by polyethylene, polypropylene, and polyester fibers; ABs and OPEs are strongly linked to municipal effluents and aquaculture; OUVAs primarily reflect recreational activities; and PFAS profiles indicate mixed industrial and consumer-product origins. Collectively, these patterns demonstrate that coral reefs increasingly function as integrative sinks and indicators of multi-class contaminant pollution, underscoring the need for long-term, multi-compartment monitoring frameworks.
Key toxic mechanisms and ecological implications
-
At the mechanistic level, ECs exert multi-scale toxic effects on coral reef organisms, spanning molecular, cellular, physiological, and ecological dimensions. Despite their diverse physicochemical properties, several conserved pathways consistently emerge, including oxidative stress, mitochondrial dysfunction, inhibition of photosynthesis in symbiotic dinoflagellates, immune dysregulation, endocrine disruption, and perturbation of the coral-associated microbiome.
Excessive production of reactive oxygen species (ROS) represents a central initiating event, leading to metabolic impairment, apoptosis, and reduced energy availability for calcification, growth, and immune defense. Concurrently, contaminants such as OUVAs and ABs directly suppress photosynthetic efficiency, destabilizing coral–algal symbiosis and increasing bleaching susceptibility. MPs further exacerbate these effects through physical abrasion, energetic dilution, and vector-mediated transport of co-occurring toxicants and pathogens.
At the ecosystem scale, chronic exposure to EC mixtures can restructure species composition, weaken trophic connectivity, and reduce reef functional complexity. Importantly, contaminant stress frequently interacts with climate-driven stressors, including warming and acidification, often amplifying biological damage and eroding coral tolerance thresholds, thereby threatening long-term reef resilience.
Research gaps and future directions
-
Despite recent advances, major knowledge gaps persist in understanding the sources, fate, and ecological consequences of ECs in coral reef ecosystems. Future research should prioritize the development of standardized, long-term, and multi-compartment monitoring frameworks to improve spatiotemporal coverage and cross-study comparability.
Mechanistic investigations need to move beyond single-contaminant approaches toward environmentally realistic multi-stressor designs that integrate chemical mixtures with climate-driven pressures such as warming and acidification. Coupling multi-omics techniques with physiological and imaging tools will facilitate pathway-level understanding and early-warning biomarker identification.
Greater emphasis should also be placed on early life stages, trophic transfer, and ecosystem-scale responses, which remain poorly constrained yet critical for predicting reef vulnerability. Finally, translating scientific insights into management practice—through the establishment of ecologically relevant thresholds, predictive modeling, and integrated land–sea pollution control strategies—will be essential for mitigating contaminant impacts and enhancing reef resilience under global change.
-
The authors confirm their contributions to the paper as follows: Guanghui Xiang: conceptualization, investigation, methodology, formal analysis, writing − original draft; Xiaoying Liang: conceptualization, resources; Bibai Du: funding acquisition, supervision; Lixi Zeng: funding acquisition, supervision; Hui Chen: data curation, writing − review and editing. All authors reviewed the results and approved the final version of the manuscript.
-
All data used in this article are derived from public domain resources.
-
This work was supported by the National Natural Science Foundation of China (Grant Nos 42477398, 42277362, U25A20824, and 22276071), the Guangdong Basic and Applied Basic Research Foundation (Grant Nos 2024A1515030109 and 2023A1515012253), the Youth Fund Special Project of Guangdong Provincial Academy of Environmental Science (Grant No. YFSP2024-01), and the Fundamental Research Funds for the Central Universities (Grant No. 21624218).
-
The authors declare no competing financial interest.
-
Full list of author information is available at the end of the article.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Xiang G, Liang X, Chen H, Du B, Zeng L. 2026. Emerging contaminants in coral reef ecosystems: environmental occurrence, distribution patterns, toxic mechanisms, and ecological risks. New Contaminants 2: e011 doi: 10.48130/newcontam-0026-0008
Emerging contaminants in coral reef ecosystems: environmental occurrence, distribution patterns, toxic mechanisms, and ecological risks
- Received: 13 December 2025
- Revised: 02 February 2026
- Accepted: 28 February 2026
- Published online: 23 March 2026
Abstract: Coral reefs are among the most biodiverse and ecologically valuable ecosystems on Earth. However, the increasing discharge of pollutants into marine environments from human activities, such as domestic sewage, industrial effluents, and agricultural runoff, has intensified stress on coral communities. These anthropogenic pollutants not only accelerate coral bleaching but also disrupt the ecological stability of reef-associated marine systems. In recent years, growing attention has been paid to the presence and impacts of emerging contaminants (ECs) in coral reef ecosystems. Major EC classes of concern include microplastics (MPs), antibiotics (ABs), organic UV absorbers (OUVAs), organophosphate esters (OPEs), and per- and polyfluoroalkyl substances (PFASs). This review systematically synthesizes current knowledge on the environmental occurrence, spatial distribution, and ecological risks of these five major EC classes in coral reef environments. Key knowledge gaps are highlighted, particularly with respect to mixture toxicity, chronic exposure, and interactions with climate-driven stressors. Finally, an integrated multi-stressor risk framework is proposed to support future monitoring, pollution management, and coral reef conservation under global environmental change.
-
Key words:
- Coral holobiont /
- Emerging contaminants /
- Spatial distribution /
- Bioaccumulation /
- Oxidative stress