-
Sweet cherry (Prunus avium L.) is a valuable fruit crop that has been cultivated for millennia and bred in recent centuries. This crop has been grown primarily in temperate climates around the world for 2,000 years[1], with hundreds of cultivars available commercially[2]. Before the late 1800s, improved cultivars were usually obtained by selection among locally grown landraces or among open-pollinated seedlings. Intentional sweet cherry breeding, involving all four stages of goal setting, creating new genetic variation, selection among offspring, and commercialization[3], began at least as early as 1880[4]. In 1991–2004, sweet cherry breeding programs worldwide released 230 new cultivars, mostly from Europe (116 cultivars), North America (71 cultivars), and Asia (33 cultivars)[5]. The general objectives of these programs are to develop a series of early-, mid-, and late-season cultivars having various skin colors, with each cultivar preferably being self-fertile, having resistance to rain-cracking and diseases such as bacterial canker, and bearing high-quality fruit in terms of size, firmness, and flavor[6−8]. Each breeding program also sets its specific breeding objectives based on regional growing conditions and target markets. These additional objectives include cold hardiness, low chilling requirement, very early or very late season maturity timing, suitability to mechanical harvesting, and powdery mildew resistance[5,8−11].
In 1949, a sweet cherry breeding program was established to develop regionally adapted new cultivars for the inland Pacific Northwest (PNW). The inland PNW of the U.S. and Canada has a suitable climate for large-scale sweet cherry production because the region has warm, dry summers and winters[12]. The region contributes 68% of the total USA fresh sweet cherry production for fresh consumption[13]. Dr. Harold W. Fogle of the US Department of Agriculture began sweet cherry breeding in 1949 at the Roza Research Farm near Prosser, WA, USA; he developed and released the popular cultivar Rainier ('Bing' × 'Van'), which is still widely grown in the PNW, and its high-quality fruit is often exported[14]. From 1963 to 1985, tree fruit breeder Dr. Thomas K. Toyama from Washington State University (WSU) managed this breeding program[6]. Toyama established three primary breeding objectives according to target attributes: (1) expanded ripening season, especially earlier than 'Bing'; (2) high-quality fruit, especially large, very firm, and good flavor; and (3) self-fertility[14].
Eleven cultivars have been named and released from Toyama's breeding program. Dr. Toyama was not sufficiently satisfied to commercially release any of his selections before retiring 'because in different years they performed differently'[15]. Evaluation of Toyama's germplasm since the mid-1980s led to the release of Chelan, Index, Cashmere, Glacier, Olympus, Simcoe, Benton, Selah, Tieton, Kiona, and Cowiche[6,14,16,17]. Selection PC 8011-3 was patented in 2005 but has not been named. While no further releases are expected[18], Toyama's cultivars and selections have been used extensively in recent decades as parents in the WSU breeding program, including several families created around the turn of the millennium using the selection PMR-1[6] and then, as the program was officially revived in 2004[7]. This elite 'Toyama germplasm' has several valuable features. For example, many are self-fertile, 'Tieton', 'Glacier', 'Cowiche', and 'Selah' have extra-large fruit[19], and PMR-1 has complete genetic resistance to the devastating disease of powdery mildew for leaves and fruit[20−22]. 'Chelan' is the most widely planted early-season cultivar in the PNW[23,24]. Extant Toyama germplasm comprises these 11 cultivars, which are available commercially worldwide, as well as 30 selections still existing in a collection at the WSU Roza Research Farm in Prosser, WA, USA.
The genetic origins and diversity of Toyama's commercially valuable sweet cherry germplasm are not clear. The objective of this study was to characterize the parentage and ancestry of Toyama's extant selected germplasm in the context of the breeder's entire efforts in new germplasm creation, via a detailed examination of the historical hand-written records.
-
Dr. Toyama kept a detailed handwritten 'Breeding Records' book (Fig. 1). Pertinent details of the year of each cross, names of parents, and numbers of pollinations, fruit, stratified seeds, and germinated seeds obtained from this book were typed into Microsoft Excel, from which calculations and summaries were made. All calculations of parentage here assumed that the recorded parents were correct, except in one case where recent genotypic analyses[25] revealed the correct name for one commonly used parent—Toyama's 'Precoce de Beaulieu' was actually 'Moreau'. Parental origins were categorized in eight geographical and temporal groups (as determined by historical reports such as[3]: 'Toyama own selections' (selections with a 'PC' prefix in the WSU program), 'North America − recent' (e.g., 'Van', 'Stella', and 'Rainier', as well as any coded-label selections without a 'PC' prefix, e.g., P8-79), 'North America − old' (e.g., 'Bing' and 'Lambert'), 'France – recent)' (e.g., 'Early Burlat' and 'Moreau'), 'France – old' (e.g., 'Empress Eugenie' and 'Lyons'), 'Germany − old' (e.g., 'Hedelfinger' and 'Buttner's Rote Knorpelkirsche'), 'Europe, other − recent' (e.g., 'Hollander' and JI 5773), and 'Europe, other − old' (e.g., 'Napoleon' and 'Bada Reisenkirsche'), with the temporal threshold between'old' and'recent' being the early 1900s. As each controlled pollination, fruit, and seed involved two parents, both parental origins were tracked for each such physical unit. Calculated proportions, therefore, represented the total number of available parental gametes per physical unit. For example, a 'Stella' × 'Early Burlat' family of 1,000 pollinations, 200 fruit, 150 seeds, and 50 seeds germinated would have these numbers repeated under both 'North America − recent' and ' France − recent'. Relative differences among parental origins for conversion rates (of pollinations to fruit to seeds to germinated seeds) were determined by one-way analysis of variance in the R programming language, with Games-Howell tests to identify significant differences among means (p < 0.05)[26].

Figure 1.
Toyama's 'Breeding Records' book (left) and an example of details within (right) in which crossing activities and outcomes were recorded. Records included in columns were the four-digit family number, parents used, location of the female parent tree, dates of flower emasculation, pollination, seed stratification, and planting, numbers of pollinations, fruit, seeds, and germinated seeds, and miscellaneous comments. Data were sometimes missing for certain columns or families. For sweet cherry, records in this book began in 1964 (records shown here for 1965) and ended in 1989; although the final year of crossing by Dr. Toyama before retirement was 1984, and the final year in which the numbers of successfully germinated seeds was recorded was 1981.
Ancestry was determined by tracking the pedigrees of parents back to their earliest known progenitors, according to pedigree information described by[3,25,27,28] that included much genotypic verification. For simplicity, the commonly used parent 'Sam' ([Windsor open-pollinated] × Open-pollinated) was classified here as an ancestor because three-quarters of its ancestry was unknown. The unknown father of Ebony ('F_Ebony') was tracked as a separate entity rather than adding it to the 'Unknown' category because of its relatively extensive representation, particularly in the first few years. Ancestry at each plant stage, from gametes in pollinations to extant selected material, was quantified as the estimated genomic proportion assuming exactly 50% the genetic background was transmitted each generation. Beyond parentages of cultivars already verified by genotypic analysis, including for most of Toyama's extant cultivars[25,27], all calculations of ancestry assumed that Toyama's recorded parentages and other reported pedigrees were correct. For the six families involving open pollination of Toyama selections, the numbers of paternal gametes involved in these families were estimated from the average proportion of pollinations that became fruit for all other parental origins, and the same estimated number of maternal gametes was then added to 'Toyama own selections' while the paternal gametes were recorded as 'Unknown − O.P.'.
-
Toyama's crosses from 1965 to 1984 involved pollinations of at least 88,108 flowers in 241 families and resulted in a total of at least 18,319 fruit, 14,994 seeds, and 5,182 germinated seeds (Table 1). Germinated seeds were obtained for only 70 of the 178 families for which such data were available (Supplementary Table S1). The overall conversion rate of pollinations to germinated seeds was 5.5%, considering only the families for which data were available for both germplasm types (Supplementary Table S1). The overall conversion rates varied greatly among years, with the highest in 1971 (23%, 583 seeds from 2,502 pollinated flowers) and no obtained germinated seeds (0%) for seven of the years (Fig. 2). In the year that the most pollinations were performed, 1966, with more than 13,000 pollinations, the conversion rate was 6.8%. Within each year for which germinated seeds were successfully obtained, wide variation was observed among families in conversion rates. For example, among the series of attempted large families in 1966, rates varied from 0% to 32%; every parent provided some germinated seeds, but families with 'Lambert' as the mother had a consistently high conversion rate, while all other mothers and most fathers were inconsistent (Table 2). In the year of the highest conversion rate, 1971, 'Stella', an offspring of 'Lambert', was a parent for all seven families with pollination and germination data available. Two other parents with high conversion rates were 'Gros Blanc' and 'Dönissens Gelbe Knorpelkirsche', used in 1970 but not when they were used in 1969 (Supplementary Table S1).
Table 1. Total numbers of recorded pollinations conducted, fruit, seeds, and germinated seeds obtained for 234 families attempted in 1965–1984 by Dr. Thomas Toyama, categorized by parental origins and counting the two parental gametes per physical unit (except in totals of the bottom line).
Parental origin Pollinations Fruit Seeds Germinated seeds Number Families* Number Families Number Families Number Families Toyama own selections 33,439 68 (147) 9,303 147 6,980 139 1,165 68 North America − recent 82,679 138 (151) 14,946 125 11,136 123 4,512 122 North America − old 29,715 56 (61) 5,405 42 7,083 54 2,362 46 France − recent 7,881 15 (16) 1,069 16 817 13 540 16 France − old 5,146 28 (28) 1,534 28 640 25 380 27 Germany − old 4,702 26 (26) 851 26 595 26 321 26 Europe, other − recent 8,160 23 (24) 1,039 24 870 21 180 24 Europe, other − old 4,494 22 (22) 1,346 22 946 21 214 21 Unknown − O.P. − − (6) 1,145 6 921 6 690 6 Total (gametes) 176,216 376 (480) 36,638 436 29,988 428 10,364 356 Total (physical units) 88,108 188 (240) 18,319 218 14,994 214 5,182 178 The physical units of pollinations were flowers. O.P. = open-pollinated. * Numbers of families in parentheses represent the actual number of families listed in the 'Breeding Records' book; figures outside parentheses represent only those families with recorded numbers of pollinations, as for other parts of this table. For example, the '147' listed families involving Toyama own selections consisted of 12 in which such a selection was the mother only (six of these being O.P.), 15 as the father only, and 60 (= 120 parental situations) in which Toyama selections were both of the parents used; however, no data on pollination numbers were recorded for seven of the mother-only families, two of the father-only families, and 35 (= 70 parental situations) of the both-parents families. For 'North America - old', the number of seeds was more than the recorded number of fruit because data on the number of fruit were missing for several large families. 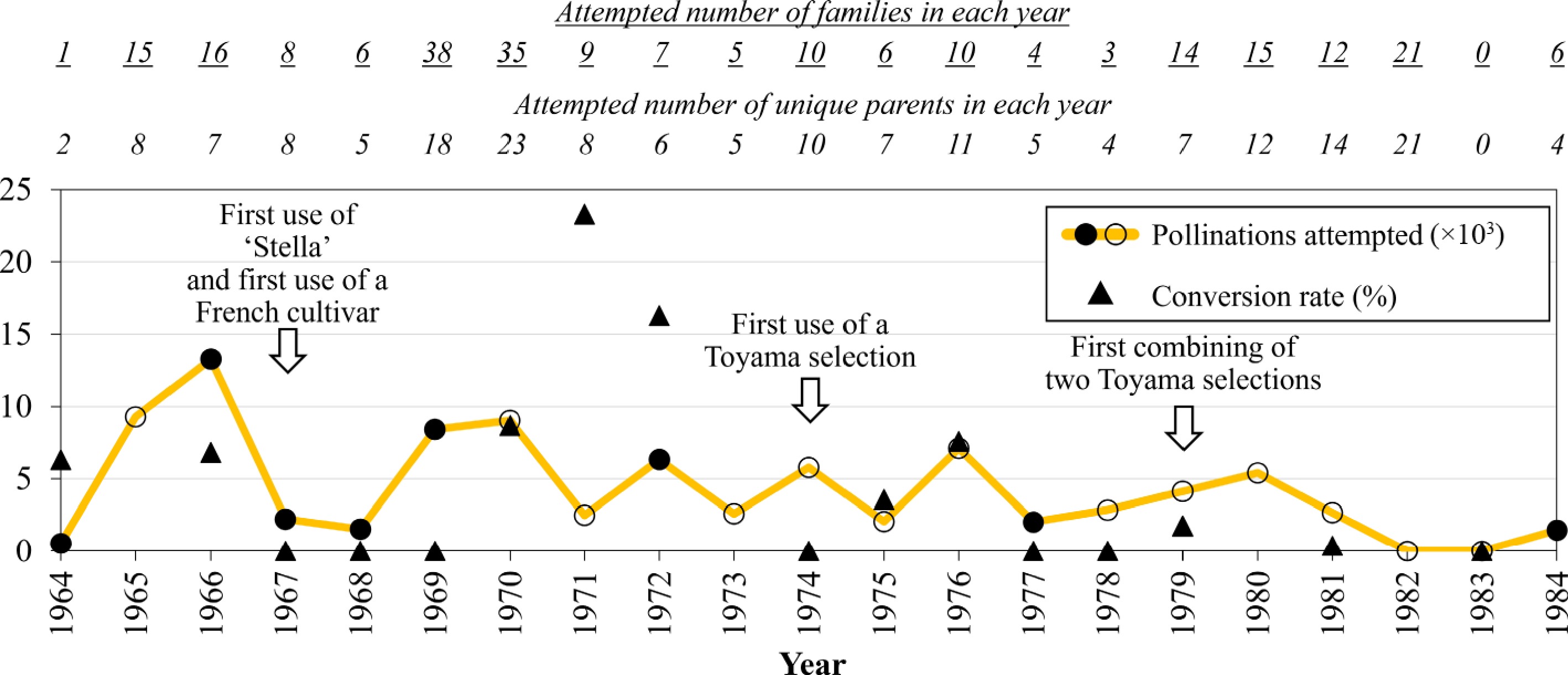
Figure 2.
Summary of crossing input and outcomes by Dr. Thomas Toyama over two decades (1964–1984). Numbers of pollinations attempted shown in open circles are minimums, given that pollination data for some families were not recorded in those years. No germinated seedlings were recorded in 1967–1969, 1974, 1977–1978, and 1983, while no data on germinated seedling numbers were available in 1965, 1973, 1980, 1982, and 1984.
Table 2. Conversion rates (number of germinated seeds obtained to flowers pollinated) for 16 attempted families using seven parents in 1966 by Dr. Thomas Toyama.
Father Total Bing Ebony Sam Star Van Windsor Mother Bing − 10% − − − 0% 6% Ebony − − − − − 11% 11% Lambert − 15% 16% − 23% 32% 23% Sam 4% 0% − 9% − 6% 4% Van 5% 12% 0% 0% − 0% 3% Total 4% 7% 6% 5% 23% 8% 6.8% Across all families for which data were available, the average conversion rates were 22.1% for pollinations to fruit, 75.6% for fruit to seeds, 35.6% for seeds to germinated seeds, and 6.48% of pollinations to germinated seeds (Fig. 3). Maternal origin groups of parents significantly differed for each conversion rate (Fig. 3). 'France − old' had the highest fruit sets (pollinations-to-fruit rates) at 39%, while the few 'France − recent', 'Europe − recent', and 'Europe − old' families had the lowest fruit set at 1%, 7%, and 21%, respectively. The fruit-to-seed conversion rates of 'North America − recent' (86%) and 'Toyama own selections' (79%) were significantly higher than for 'France − old' (62%). Seed viability (seeds-to-germinated seeds) was significantly higher for 'France − old' (56%), 'Germany − old' (51%), 'North America − recent' (38%), and 'Toyama own selections' (24%) than for 'France − recent' and 'Europe, other − old' that were associated with zero viability. For overall conversion rates of flowers pollinated to germinated seeds obtained, 'Germany − old' (12.5%), 'France − old' (11.8%), and 'North America − recent' (6.7%) were significantly higher than the zero rate for 'France − recent' and 'Europe, other − old' (Fig. 3), driven by the success of the previously mentioned 'Dönissens Gelbe Knorpelkirsche', 'Gros Blanc', and 'Stella' in 1971, respectively. The high pollinations-to-germinated-seed rate of 'Lambert' in 1966 (Table 2) was derived from a particularly high pollinations-to-seed rate, which was double that of 1966 families and all families (Supplementary Table S1). Across the entire 21-year period, every mother or father used for two or more years was always associated with at least one family for which no germinated seeds were obtained (Supplementary Table S1).
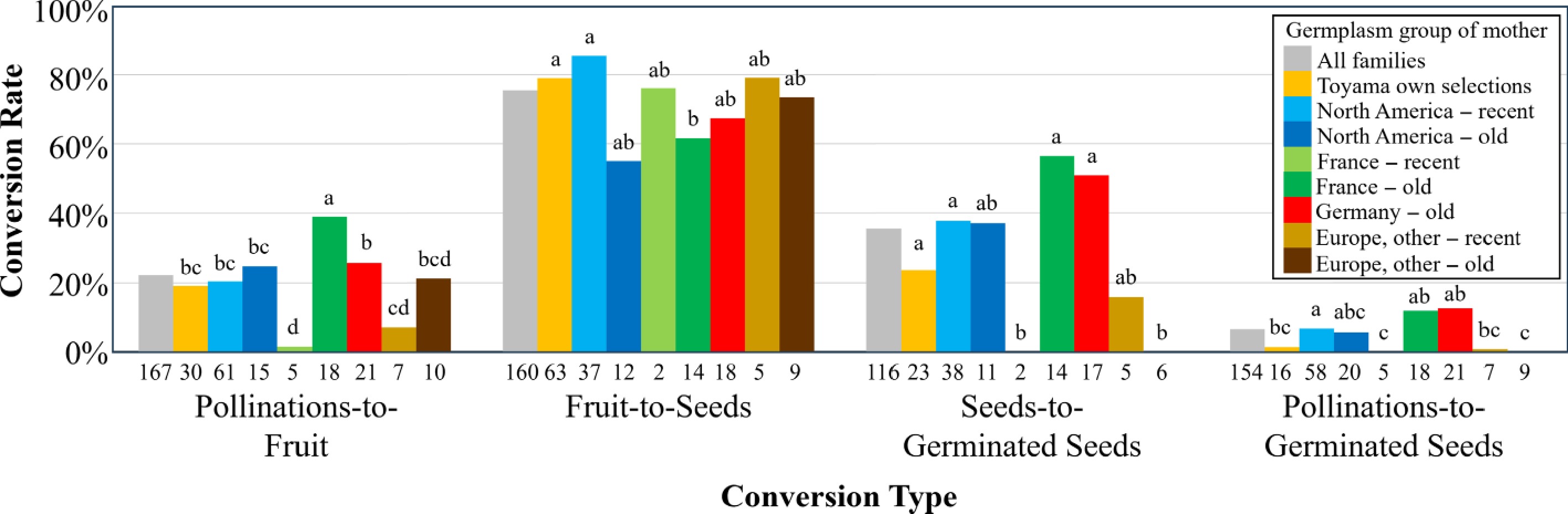
Figure 3.
Conversion rates achieved in all recorded families between pollinations attempted and obtained fruit, seeds, and germinated seeds, considering the germplasm group of the mother tree of each family. The number of families with recorded data for conversion calculations is indicated below each column. Within each conversion type, germplasm groups with different letters are significantly different (p < 0.05).
Parents used
-
Dr. Toyama used a total of 103 parents from various origins (Fig. 4), with up to 23 unique parents used in any specific year (Fig. 2). 'Toyama own selections' were the single largest group used as parents, numerically representing a half of all parents (54/103) (Fig. 4). These selections arose from crosses as early as 1965 and began to be used as parents in 1974. The first intentional crossing of two 'Toyama own selections' was in 1979, and from then on, most crosses were such combinations. 'Stella' ('Lambert' × JI 2420) was the most-used parent in Toyama's crossing scheme: both as mother (25,669 gametes) and father (9,621 gametes) and accounted for 20% of gametes in attempted pollinations (Fig. 4). 'Stella' was similarly highly represented in parental gametes for fruit (22%), seeds (14%), and germinated seeds (24%) (Fig. 4). 'Stella' was correspondingly the most represented parent of germinated seeds: 46% had this cultivar as the maternal parent, paternal parent, or both. The most commonly represented groups in Toyama's crosses were 'North America − recent' (47% of gametes recorded for pollinations, 41% for fruit, 37% for seeds, and 44% for germinated seeds), 'North America − old' (17% of gametes for pollination, 15% for fruit, 24% for seeds and 23% for germinated seeds), and 'Toyama own selections' (19% of gametes recorded for pollinations, 25% for fruit, 23% for seeds, and 11% for germinated seeds) (Table 1; Fig. 4). Besides 'Stella', other prominent parents were 'Bing', 'Lambert', 'Sam', and 'Yellow Glass' (Fig. 4). Almost half (46) of the original known parents used in crossing were represented in the 5,182 germinated seeds.
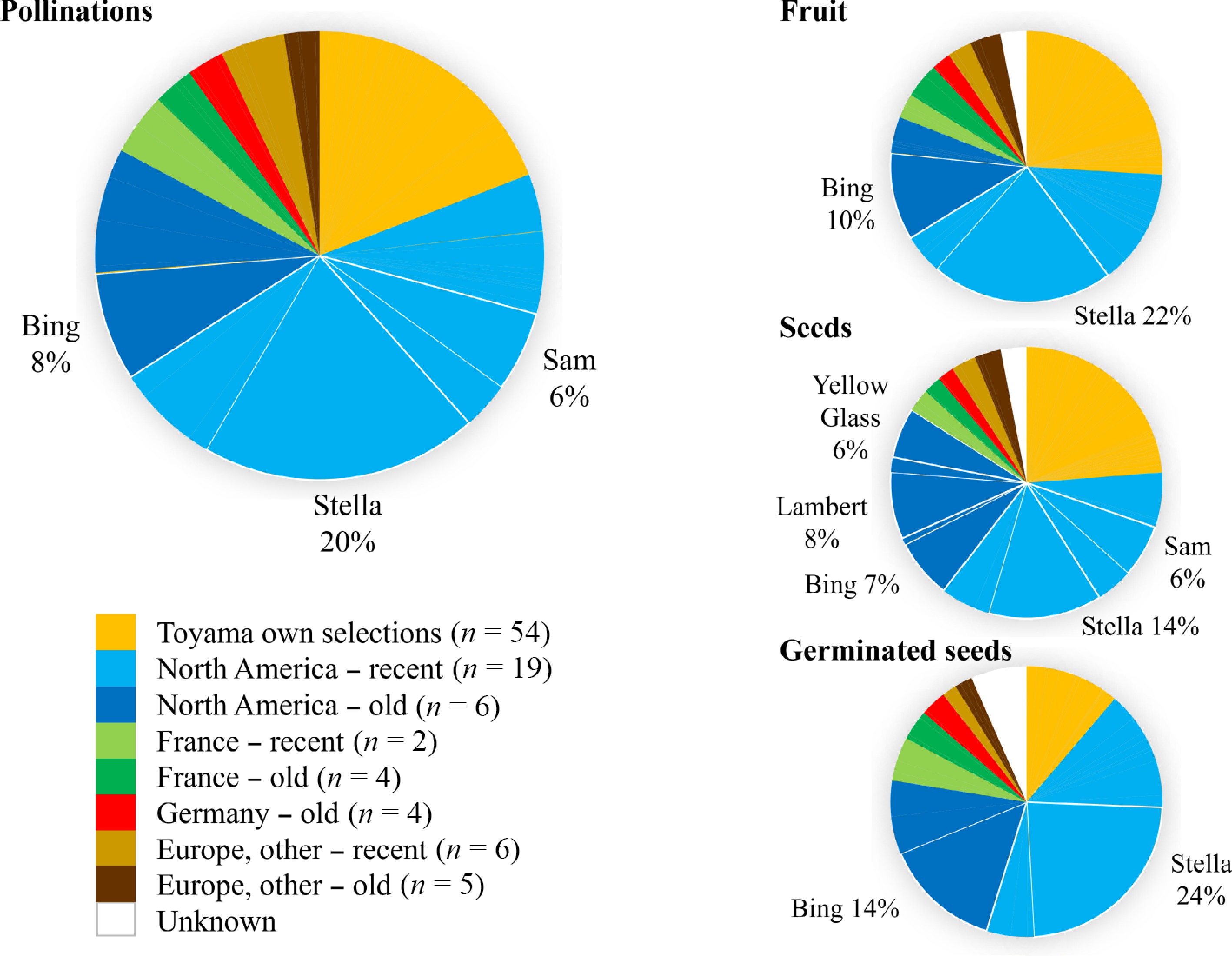
Figure 4.
Frequency of use of parents by Dr. Thomas Toyama for crosses from 1964 to 1984. Use of parents is quantified by the proportion of parental slots (gametes) involved in recorded pollinations, fruit, seeds, and germinated seeds. Parents are grouped according to geographic/temporal origins or 'Unknown' for fathers of open-pollinated families. Parents representing at least 5% of the total are noted and delimited with thicker lines.
Ancestral genetics accessed
-
Toyama's crosses incorporated many ancestors from various geographical and temporal origins, although certain known ancestors were highly represented (Fig. 5). The most common ancestral origin was 'Europe, other − old'. The cultivar Napoleon accounted for 27% of the ancestry of crosses made, especially because it is a grandparent of 'Stella' through both parents and is a parent of 'Bing', but it was also directly used as a parent for nine created families. 'Napoleon' represented the ancestral background of 31%, 29%, and 33% of fruit, seeds, and germinated seeds, respectively (Fig. 5). Unknown ancestry accounted for 7%–12% of germplasm, stemming from the use of various code-labeled selections of unclear origins and some families derived from open pollination.
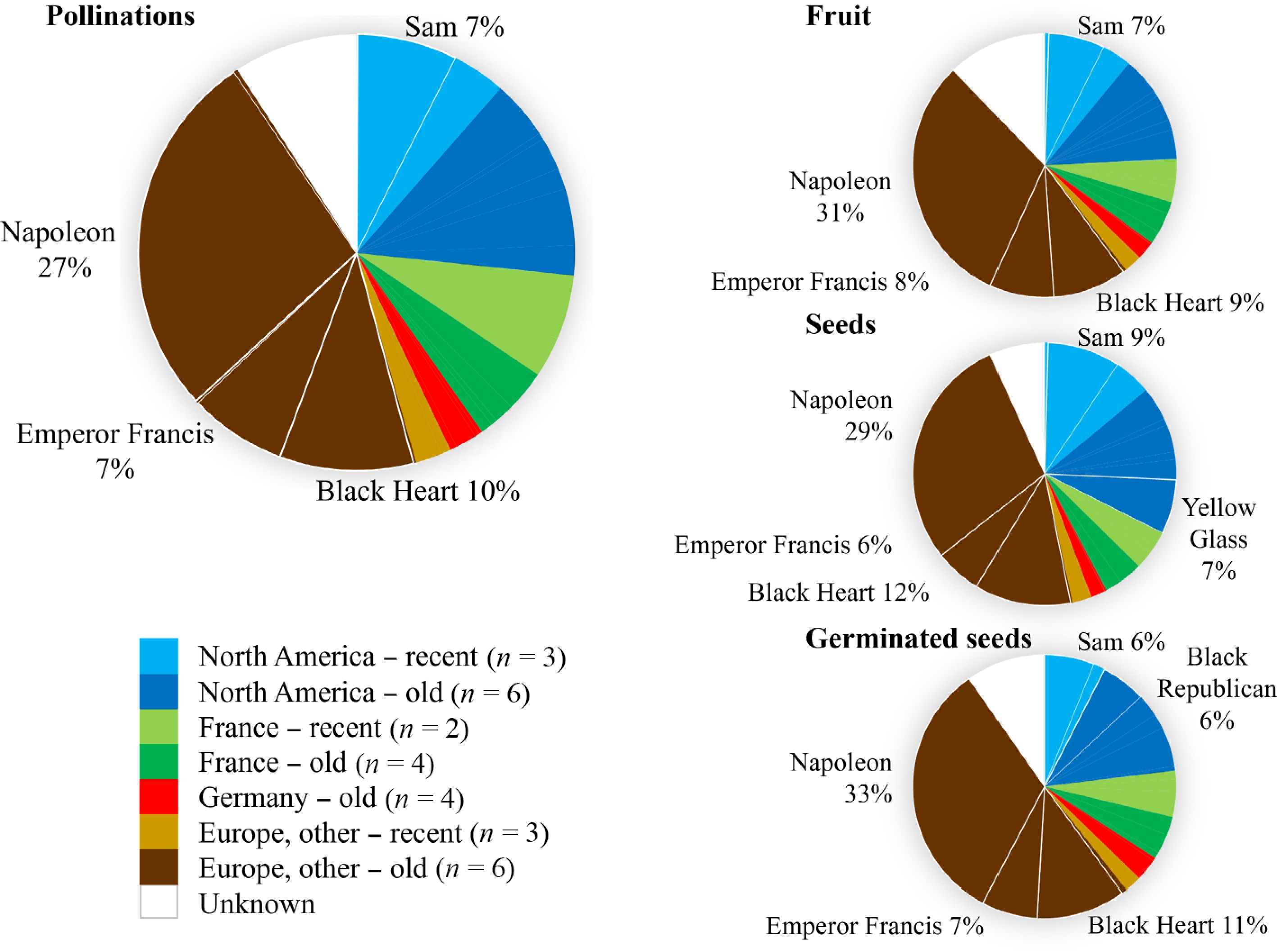
Figure 5.
Frequency of representation of ancestors in crosses made by Dr. Thomas Toyama from 1964 to 1984. Representation of 29 known ancestors is quantified by their estimated genomic contributions to parents used across all parental slots (gametes) involved in recorded pollinations, fruit, seeds, and germinated seeds. Ancestors are grouped according to geographic/temporal origins or 'Unknown' for fathers of open-pollinated families. Ancestors representing at least 5% of the total are noted and delimited with thicker lines.
Toyama's extant selected germplasm: parentage and ancestry
-
The origins of the 20 parents used to create the extant selected Toyama germplasm (11 cultivars and 30 selections; Supplementary Table S2) were skewed more toward Toyama's own selections than for the entire set of crosses made (Fig. 6a compared to Fig. 4). The only parents of European origin for extant germplasm were several individuals from France and 'Hollander' (although according to Toyama's records the parent of two extant selections might be 'Starking Hardy Giant', an old USA cultivar, rather than 'Hollander'). Of the original 103 known parents used in crossing, only 20 were parents of this extant selected germplasm. The most commonly recurring parents of extant Toyama germplasm were 'Stella', two Toyama selections (PC 7147-4 and PC 66138-2), and 'Early Burlat' (Fig. 6a). All extant cultivars except one ('Olympus') had 'Stella' as a parent or grandparent. Similarly, the only extant selections without 'Stella' as a parent or grandparent were three with unknown fathers and one (PMR-1) with both parents being unknown. Most extant cultivars (seven of the 11) were recorded to have 'Stella' and a French cultivar [either 'Early Burlat' or 'Moreau (recorded by Toyama as 'Precoce de Beaulieu')] as parents or grandparents. One extant cultivar resulted from the combination of 'Stella' and 'Hollander' (or 'Starking Hardy Giant'), and another had the latter as a grandparent. The parental/grandparental combination of 'Stella' and a French cultivar was less frequent among the extant selections compared to cultivars (eight of the 30 selections), while the combination of 'Stella' and 'Hollander' (or 'Starking Hardy Giant') was much increased (14 selections) (Supplementary Table S2).
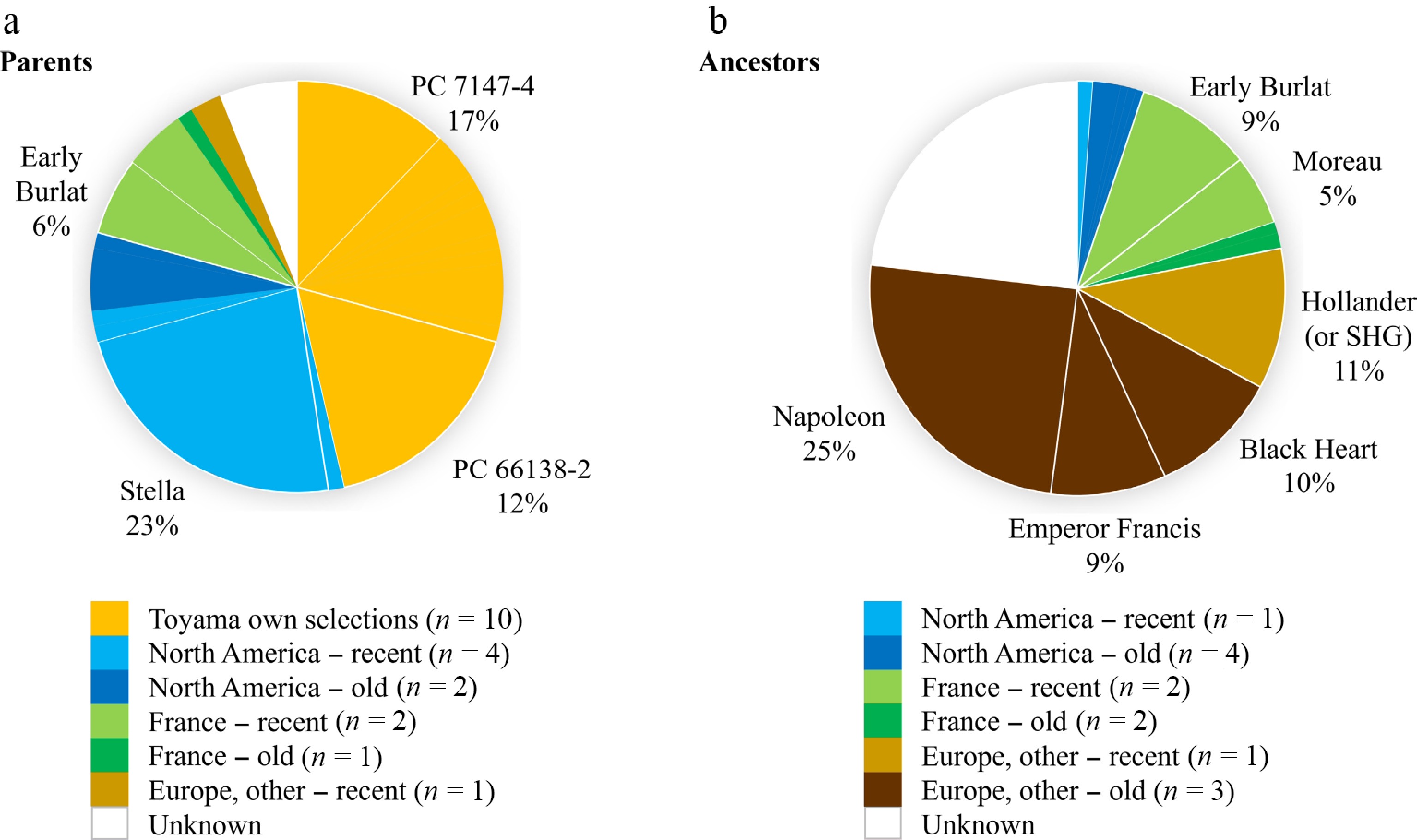
Figure 6.
(a) Frequency of representation of parents among extant Toyama selected germplasm (11 cultivars and 30 selections), grouped according to geographical/temporal origins. (b) Frequency of representation of ancestors of extant Toyama selected germplasm, grouped according to geographical/temporal origins. Within each origin, only those cultivars representing at least 5% of the total are noted and delimited with thicker lines. SHG = 'Starking Hardy Giant'.
The ancestry of this extant selected Toyama germplasm was like that of germplasm stages prior to selection for commercial performance, although there was a greater representation of French cultivars and 'Hollander' (or 'Starking Hardy Giant'), less of North American, and no German (Fig. 6b compared to Fig. 5). Old European cultivars composed almost half the ancestry of extant germplasm, with 'Napoleon' continuing to dominate. Twenty-three percent of the ancestry of Toyama's extant germplasm was unknown (Fig. 6b), which was double that of pre-selected germplasm (Fig. 5), and was because of four selections mentioned above with some unknown parentage and 13 other selections being offspring of PC 66138-2 or PC 66154-1 for which parentage records were not found in the Breeding Records book.
-
Dr. Thomas K. Toyama created a commercially viable germplasm base that is important to current breeding efforts and the supported PNW sweet cherry industry. Through this study, a detailed understanding was obtained of the history, parents, and ancestry of the created germplasm and extant cultivars and selections from the iconic WSU sweet cherry breeding program of 1965–1984. The investigation revealed that Toyama utilized a broad genetic base, but most commonly used the self-fertility source 'Stella' and had a large ancestral contribution from the old European cultivar Napoleon. Extant cultivars from this breeding program were determined to be derived particularly from 'Stella' and often involved the signature complementing of self-fertile 'Stella' with two early-season French cultivars.
Pollinated flowers converted to germinated seeds
-
Toyama's 'global success rate' for converting pollinated flowers to germinated seeds[2] of 5.5% is higher than reports in several other sweet cherry breeding programs in Europe. The Romanian program at Iasi Fruit Research Station made more than 700,000 pollinations to produce 6,493 germinated seeds, from 1981 to 2004[29] – a global success rate of 0.9%. The French program at INRA-Bordeaux reported a global success rate of 1% (almost 270,000 pollinations and 2,625 germinated seeds) from 2008 to 2018[2]. The Spanish program of the Jerte Valley in Extremadura from 2006 to 2008 had a global success rate of 4.9% (almost 37,000 pollinations and more than 1,800 germinated seed[30]) – which was almost as high as Toyama's. Although Toyama's program obtained a relatively high global success rate, large variability was observed among yearly conversion rates. Similar variability among years was noted by the European studies, suggesting that external conditions, such as weather or climate for the longer-running programs, can greatly influence the success rate. Indeed, cherry breeding is renowned for its unpredictability in fruit set and germination rates[2]. Extreme weather events, especially at flowering, to which sweet cherry is sensitive[31], probably explain the complete lack of fruit set for two of the 21 years (1968 and 1977). Quality of pollen used by Toyama appears to have also been affected by yearly weather conditions, with recorded notes mentioning 'fresh unfrozen pollen', 'frozen pollen', and 'pollen scarce' as well as use of '1974 pollen' and '1975 pollen' for some crosses in 1976, indicating unavailability of flowers and thus fresh pollen of the intended fathers. Besides such serendipity, careful management of mother trees and pollen might have helped the success rate of Dr. Toyama's program, as well as his use of a genetically broad parent pool.
Access of a broad germplasm base
-
Several features indicate that the Toyama germplasm base was considerably broad. Although Dr. Toyama relied much on 'Stella', the most popular worldwide source of self-fertility[32], this cultivar represented only 20% of gametes in pollinations, and each of the 102 other parents, from various geographical and temporal origins, represented only 8% or less of gametes in pollination (Fig. 4). Similar to the many and varied parents, diverse ancestors filled the germplasm base, each with small contributions (Fig. 5). From an ancestral perspective, 'Napoleon', arising in Europe in the 1800s[3], was over-represented, although it was calculated to account for only 27%–33% of gametes. The high global success rate of Toyama's program appears to be due at least partially to his use of diverse germplasm. The use of certain female genitors (mothers) can greatly influence fruit set, seed viability, and germination rate[2]. In Toyama's germplasm, some of the variation in conversion rates observed among parents and families (Table 2; Supplementary Table S1) and among the parental origin groups might be due to genetic differences. Genetically influenced traits might have varied among the parents, such as pollen viability, fruit set (yield), and degree of embryo abortion. Similarly, genotype-by-environment effects could have influenced conversion rates, such as if in some years mother trees predisposed to late-blooming escaped late frosts that destroyed pollination attempts on earlier-bloomers. For example, Toyama's breeding records in 1970 noted 'some pistils frozen' for a cross and 'late pollination' for another, while other families that year seemed unaffected, consistent with reports that some cultivars are less sensitive than others to cold temperatures at bloom[33,34]. The low proportional representation of 'Toyama own selections' at the stage of germinated seeds (Fig. 4) might be associated with low seed viability (Fig. 3) that is reportedly attributed to inbreeding among the small number of highly related parents typically used by any program[2,35,36]. Although inbreeding coefficients were not determined here for the Toyama attempted families and extant selected germplasm, after verification of cultivar pedigrees, such information could be informative. Overall, while some of the parents were associated with greater conversion rates on average, given that no parents used for two or more years were consistently associated with conversion rates greater than zero, external factors such as weather variability likely played the greatest role in year-to-year variability in breeding output. However, using a diverse range of mothers and fathers each year and over the years likely helped mitigate those external effects and successfully contributed to a relatively high 'global success rate'.
The complementarity of Toyama's extant selected germplasm
-
Toyama's extant selected germplasm might be the result of tapping into unusual sources of valuable alleles or particular allelic combinations created by joining complementary germplasm. The number and proportion of cultivars arising from a program can only be determined decades after the germplasm is created and can be difficult to assign to a particular period if a program is ongoing. However, the distinct periods of the WSU Toyama program described here (20 years, 1965–1984) and the Romanian program at Iasi Fruit Research Station (24 years, 1981–2004[29]) enable such summaries of commercial output to be made. The Toyama program's achievement of 11 cultivars from 5,182 germinated seeds (0.21% – two per thousand young plants) is perhaps higher but similar to the Romanian commercial output of eight cultivars from 6,493 germinated seeds (0.12% – one in a thousand). With advances in operations regarding germplasm creation, such as the use of protected environments and bumblebees for parents during crossing and in vitro embryo rescue to enhance seed germination[1,2], modern sweet cherry breeding programs often generate thousands of germinated seeds in a single year. The quantity and/or quality of worldwide output of sweet cherry cultivars is therefore expected to concomitantly increase in the coming years.
As demonstrated by this study, successful release and grower adoption of new cultivars is not just 'a numbers game' – it also relies on choices of parents and combinations that provide suitable new allelic combinations for the target commercial deployment environment. A wide germplasm base is often considered a requirement for the commercial viability of sweet cherry breeding[1,36]. Toyama clearly targeted – and successfully achieved – the complementing of certain desirable attributes, especially self-fertility and early season maturity timing, from distinct ancestral sources. 'Stella' appears to be the primary source of valuable alleles for the PNW industry, given its massive representation in Toyama's cultivars. 'Stella' is also the recurring progenitor of cultivars from its originating Canadian breeding program in Summerland (also part of the PNW region) that are commercially prominent in the PNW, such as the self-fertile 'Sumtare', 'Lapins', and 'Skeena'. The combination of 'Stella' with several early-season French cultivars, ancestrally distinct from 'Stella', also appears to be a defining feature of commercially successful Toyama germplasm. Complementary allelic combinations resulting from 'Stella' crossed with the French 'Early Burlat' and 'Moreau' might hold particular genetic value for PNW production, given that most Toyama cultivars were determined to have this background. 'Hollander' (or 'Starking Hardy Giant') might have additional well-suited alleles and/or effective allelic combinations with 'Stella', given its much increased representation in extant selections compared to earlier germplasm stages. This study's findings, therefore, indicate that complementing useful alleles from distinct ancestral sources can generate valuable elite germplasm. It might be worthwhile to access and combine the original sources of self-fertility and early season maturity timing to recreate and even improve upon Toyama's cultivars, especially under different selection regimes. In any case, continued use of Toyama germplasm in the WSU sweet cherry breeding program and beyond, with large numbers of seedlings generated, is expected to result in more and improved cultivars.
The fact that Toyama's 'Precoce de Beaulieu', used as a father in 1970−1978 and ultimately the parent of 'Benton' and the widely grown early-season cultivar 'Chelan', was actually 'Moreau' was previously reported only in passing (in Supplementary Table S1[25]); the implications of this synonymy are worth highlighting here. 'Precoce de Beaulieu' (or shortened to 'Beaulieu') is a parental name that appears to be no longer extant in the world. This name was recorded in 1963 as an accession (PI 131203) of the USDA-ARS National Plant Germplasm System (NPGS) received from France in 1938 and held in USDA stations in California and Oregon[37] – presumably the source of Toyama's material for this and many other parents. The name is otherwise rare in the literature and is no longer in the NPGS cherry collection nor reported in collections elsewhere. In contrast, 'Moreau' (also known as 'Bigarreau Moreau') is widely known and available in collections. The availability of 'Moreau' enables its phenotypic and genotypic evaluation to better understand valuable alleles carried (Vanderzande et al.[38]) and its further use in breeding.
Further research
-
Knowledge improvements are required in parentage and pedigree connections among Toyama's germplasm. Some of the recorded parentages in Toyama's 'Breeding Records' book might not be correct. While genotypic analyses[25,27] have largely confirmed the originally recorded parentage of Toyama's extant cultivars, including for the three ('Cashmere', 'Glacier', and 'Olympus') that Toyama used as parents from 1979, DNA fingerprinting is increasingly revealing that tree fruit breeding records can be incorrect[4,25,39,40]. Also, some parents of extant Toyama cultivars and selections are still unknown or unclear, such as for PMR-1[20,21], and some of the terminal ancestors considered here are probably closely related to each other[41]. An improved understanding of which parents and ancestors more often lead to successful cultivars could guide crossing decisions to make breeding efforts more efficient. Studies could then investigate which chromosomal segments from these parents and ancestors positively contribute to key horticultural traits, to further inform breeding decisions.
The data extracted for this study from Dr. Toyama's notebooks represented only part of the data existing in those notebooks, as they also contain additional columns, some specific notes on reasons or outcomes of certain crosses, other metadata about the sweet cherry breeding program, data for other stone fruit crops bred simultaneously, and other years beyond Dr. Toyama's involvement. The 'Breeding Records' notebooks could be digitized to render their content available for further reuse by researchers, as recommended by Farrell et al.[42] for such analog scientific data.
-
The authors confirm their contributions to the paper as follows: study conception and design: Peace C; data collection, draft manuscript preparation: Caymaz D, Peace C; analysis and interpretation of results: Caymaz D, Peace C, Vanderzande S, Luo F. All authors reviewed the results and approved the final version of the manuscript.
-
All data generated or analyzed during this study are included in this published article and its supplementary information files.
-
The authors declare that they have no conflict of interest.
-
accompanies this paper online at: https://doi.org/10.48130/frures-0026-0010.
- Supplementary Table S1 "Breeding Records" recorded data, and resulting calculations, for Dr. Thomas Toyama's sweet cherry breeding program at Washington State University, 1964−1984.
- Supplementary Table S2 Parentage and ancestry of 41 extant cultivars and selections of Dr. Thomas Toyama's sweet cherry breeding program.
- Copyright: © 2026 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Caymaz D, Vanderzande S, Luo F, Peace C. 2026. Examination of Toyama's historical breeding records reveals distinct sweet cherry germplasm created. Fruit Research 6: e016 doi: 10.48130/frures-0026-0010
Examination of Toyama's historical breeding records reveals distinct sweet cherry germplasm created
- Received: 26 November 2025
- Revised: 03 March 2026
- Accepted: 13 March 2026
- Published online: 27 April 2026
Abstract: The historical Washington State University Sweet Cherry Breeding Program, led by Dr. Thomas Toyama from 1963 to 1985, developed important and distinct sweet cherry germplasm. The program ultimately released 11 cultivars, including 'Chelan', 'Cashmere', 'Glacier', 'Tieton', 'Kiona', and 'Cowiche'. These cultivars and 30 remaining selections contribute significantly to commercial production worldwide and current breeding. However, the origins of this valuable 'Toyama germplasm' are unclear. This study relied on hand-written breeding records of crosses and verified pedigree information to analyze the output of this historical breeding program and characterize the genetic origins of Toyama's germplasm. Dr. Toyama used 103 unique parents from diverse origins to create 241 families, and at least 5,182 germinated seeds formed from at least 88,000 pollinated flowers. Most parents were from North America, especially the most-used 'Stella' that generated half of all germinated seeds. Half of the Toyama germplasm's ancestry was traced to pre-1900 European cultivars, with 'Napoleon' representing 25%–33% of the genetic background. For extant selected germplasm, 'Stella' and two of Toyama's own selections were the most used parents, accounting for 52% of recorded parentages. Similar ancestry contributions for selected and pre-selected germplasm indicate that the diverse germplasm accessed persists in extant breeding parents and commercial cultivars. Exploitation of self-fertility and Toyama's signature complementing of 'Stella' with early-season French cultivars could explain the high success rates of pollinated flowers converted into germinated seeds and eventually into cultivars. DNA-based diagnostics could further reveal the ancestry and valuable alleles of this germplasm to inform future breeding endeavors.
-
Key words:
- Breeding /
- Early-season /
- Pedigree /
- Prunus avium L. /
- Self-fertility /
- Stella













