-
Saline-alkali soil is widely distributed, and is generally characterized by high salt content, poor structure and nutrients, suppresses plant growth, even causes plant death and seriously threatens agricultural production[1, 2]. Saline-alkali stress is the most significant factor influencing the sustainable development of agriculture, which urgently needs improvement and utilization[3, 4]. Extensive studies have been carried out on salinized soil from different perspectives and progress has been made[5, 6]. In general, the improvements of saline-alkali land mainly include physical, chemical and biological measures[7−9]. The physical improvements, which are generally based on irrigation or soil change, require a large amount of capital investment, thus are not suitable for large-scale promotion. Chemical improvement measures[10], usually using gypsum, zeolite, sulfuric acid, citric acid and other chemicals to offset the salt in soil, can achieve enhancement effects in the short term. However, the inorganic amendments are complex and costly, and can also cause secondary pollution to the water and soil environments. Biological fertility improvement measures are mainly adopted to reduce soil salt content by planting highly salt-tolerant plants, which are characterized by strong operability, sustainable development and broad application prospects[11].
The cultivation of salt-tolerant green manure, with both productive and ecological value, is considered to be an effective measure for utilization and sustainable development of saline-alkali land[12]. For productive value, green manure cultivation could largely improve the buffering of alkaline soil, prevent dramatic changes in soil pH value due to excessive alkaline substances, and reduce the accumulation of soluble salt in the soil surface. Besides, the huge plant roots can effectively improve the soil structure, enhance the ability to retain water and fertilizer and improve yields[13]. A large number of studies have demonstrated the advantages of planting forage legumes on improving soil properties[14−16]. For ecological value, the introducing of salt-tolerant plants is a good approach to develop the carbon (C) sequestration potential of saline-alkali land, which can rapidly increase the vegetation cover on the surface, improve greenhouse gas absorption and store up aboveground C, contributing to modification of the global C cycle[17].
In recent decades, more attention has been paid to the cultivation of saline-tolerant forage species. Alfalfa, ryegrass, forage rape, sweet sorghum and sesbania are the potential forages that grow in saline soil[18,19]. Brassica campestris can accumulate more soluble sugars, amino acids and other similar compounds to improve the concentration of cell fluid, thus normally absorbing water and nutrients from the saline soil solution with higher concentration and avoiding salt-alkali damage[2]. Previous studies have demonstrated that Medicago sativa, an excellent perennial leguminous forage, has strong adaptability and can withstand a certain degree of saline-alkali stress[20]. Italian ryegrass has a large and developed root system, along with strong reproduction, wide distribution, rich germplasm resources, and good stress resistance[21]. Feng et al.[22] investigated the salt tolerance ability of two Italian ryegrass cultivars, and demonstrated that high salt tolerance was partly due to the prevention of plants from ionic homeostasis disruption. Therefore, it is of great significance to improve soil quality and agricultural productivity by using salt-tolerant green manure in saline-alkali land.
It is well known that utilization of green manure, to a large extent, is helpful for reducing the dependence on mineral fertilizers[23, 24]. The green manure returned to the field contributes to the generation of a good growth environment, with increase of soil organic matter and soil nutrient content, as well as improvement of soil aggregate structure, soil microbial community, and micro-ecological environment[25,26]. Accumulated evidence has demonstrated that green manure application could increase soil nutrient cycling and utilization efficiency, depending on the positive effects of soil microbes in the decomposition process[27,28]. Mwafulirwa et al.[29] reported that ryegrass shoot residue addition resulted in higher residue C mineralization rates, accelerated soil microbial activity, and increased soil organic matter priming, as compared with that of root residues, particularly in the early days. Understanding the impacts of different green manure returning to the field on saline alkali soil quality improvement is of benefit to the sustainability of agricultural production.
Soil fertility quality is an essential component of soil quality, which directly affects plant growth, agricultural production structure, distribution and benefits[30]. Appropriate evaluation methods and appropriate soil fertility indicators should be the most important considerations, which have a significant impact on soil fertility quality outcomes[31]. The calculation of soil fertility quality index is the core issue of soil quality evaluation, which is a widely employed way to evaluate the relationship between certain soil factors and soil productivity using fuzzy mathematical methods[32,33]. In this study, we employed different green manure species (Medicago sativa, Lolium multiflorum and Brassica campestris) with eight varieties, using a field experiment, aiming to measure their effects of cultivation for aboveground carbon store and returning to the field to improve saline alkali soil quality.
-
We employed four Medicago sativa varieties (Aurora, Sanditi, Eureka+, Sardi), Lolium multiflorum, and three Brassica campestris varieties (Huayouza 82, Huayouza 158, Huayouza 62) in our experiment. The Medicago sativa and Lolium multiflorum seeds were provided by Zhengzhou kaiyuan Grass Industry Technology Co., Ltd (China), and Brassica campestris seeds were provided by Zhengzhou Huafeng Grass Industry Technology Co., Ltd (China), with all germination rate > 85%.
Field site description
-
Our field experiment was carried out in Tiaozini, Dongtai, Jiangsu Province, China (32°51' N, 120°56' E). This region is a transition zone between subtropical and warm temperate zone with distinct seasons. The average annual temperature in this area is 15 °C, with the coldest month being January (mean monthly temperature 0.8 °C). July is the hottest month with an average monthly temperature of 27 °C. The average annual rainfall is 1,061 mm. The annual rainfall from June to September accounts for 63% of the whole year. The soil properties were as follows: pH 8.20, salt content 1.83 ‰, soil organic matter 7.92 g·kg−1, total N 0.48 g·kg−1, total P 0.65 g·kg−1, total K 18.76 g·kg−1, soil available N 45.92 mg·kg−1, available P 18.31 mg·kg−1, available K 187.07 mg·kg−1, Ca2+ content 41.84 g·kg−1, Mg2+ content 13.64 g·kg−1.
Experimental design
-
The field experiment, with a random design, included eight green manure varieties. We defined each variety as a treatment and a non-cultivation as control (CK). Each treatment had three replicates (plots), and the size of each plot was 2 m × 2 m. The cultivation was performed in late October, 2021. The sowing density of Medicago sativa, Lolium multiflorum and Brassica campestris was 2.5 g·m−2, 2.5 g·m−2 and 1 g·m−2, respectively. The sowing depth was 3 cm and row spacing was 25 cm. No fertilization was applied during the growth of green manure. Irrigation and weeding were the same as in routine management.
Collection of aboveground and soil samples
-
The green manure biomass was recorded at harvest (May 5, 2022). We took random subplots (0.5 × 0.5 m) within each plot and destructively harvested, and the shoots were cut at the soil surface. At the same time, we collected soil samples. Five cores (5 cm diameter and 0−15 cm depth), on the rows, were randomly sampled and sufficiently mixed to yield one representative sample. After sampling, the rest of the green manure in each plot were crushed and returned to the field with the returning depth of 20 cm. Thirty days later, soil samples were collected as previously. All collected soil samples (54 samples in total: 9 treatments × 3 replications × 2) were sieved (2.0 mm mesh) and homogenized for soil physicochemical property analysis.
Determination of aboveground samples
-
The collected green manure aboveground samples were oven dried at 65 °C for 72 h to a constant weight before weighing. The shoot dry weights were expressed as total aboveground biomass per m2. Then the dry shoots were ground through 20 mesh in a Wiley mill. The prepared aboveground samples were measured by an Elementar Analyzer (Vario EL III, Germany) for total carbon (C).
Determination of soil physicochemical properties and soil fertility evaluation
-
Soil pH was determined with soil-water slurry (1:5, w/v) by a PB-10 pH meter (Sartorius, Germany). Soil electrical conductivity (EC) was measured by a conductivity meter (B-173; HORIBA, Kyoto, Japan). Soil organic C (SOC) was measured by an Elementar Analyzer (Vario EL III, Germany). The total N was determined using a Kjeltec Analyser (FOSS Tecator, Hoganas, Sweden). The determination of soil available nitrogen (N) was measured according to Shi[34]. The soil fertility evaluation was calculated according to previously described methods[35, 36].
Statistical analyses
-
The data in our study were log-transformed when necessary to meet the criteria for a normal distribution. We employed SPSS 22.0 (IBM, Armonk, NY, USA) software for statistical analysis of all parameters. The data from each treatment were analyzed using one-way analysis of variance (ANOVA), and Duncan’s multiple range tests (P < 0.05) were performed for multiple comparisons. The Mann-Whitney U test method was used to test soil fertility index differences between non-cultivation and cultivation groups after returning to the field.
For soil fertility evaluation, we employed five soil fertility evaluation parameters, including pH, EC, SOM, TN and AN. Then we chose appropriate function curves and turning points to determine values of each soil fertility parameter, according to our data characteristics. The 'optimum' curve equation is employed for pH and EC, while the 'more is better' curve equation is used for SOM, TN and AN[33, 35, 36]. The equation for the scoring curve as follows:
(a) The 'optimum' curve equation:
$ f(x)=\left\{\begin{array}{ll} 0.1 & x \leqslant L, x \geqslant U \\ 0.1+0.9(x-L) /\left(O_{1}-L\right) & L \lt x \lt O_{1} \\ 1.0 & O_{1} \leqslant x \leqslant O_{2} \\ 1.0-0.9\left(x-O_{2}\right) /\left(U-O_{2}\right) & O_{2} \lt x \lt U \end{array}\right. $ (b) The 'more is better' curve equation:
$ f(x)=\left\{\begin{array}{ll} 1.0 & x \geqslant U \\ 0.1+0.9(x-L) /(U-L) & L \lt x \lt U \\ 0.1 & x \leqslant L \end{array}\right. $ where x is the monitoring value of the parameter; f(x) is the score of the parameters ranging between 0.1 and 1.0; U and L are the upper and the lower threshold values of the parameters, respectively. O1 and O2 are the best values of the variables.
We employed partial least squares path modeling (PLS-PM), based on 'plspm' (1000 bootstraps) package in R software (v.4.0.0), to determine the complex multivariable relationships among green manure varieties, edaphic variables, plant C content, biomass and soil fertility. Then we tested the model architectures from simple to complex (direct and indirect links, previous effects)[37]. Based on the determination coefficient (R2) of the explained latent variables and goodness of fit (GoF), we selected the corresponding architecture.
-
There were significant (P < 0.05) differences among Medicago sativa, Lolium multiflorum and Brassica campestris cultivation treatments for biomass at harvest (Fig. 1). In total, the largest green manure biomass was observed in Lolium multiflorum cultivation treatment (895.11 g·m−2). Lolium multiflorum cultivation treatment increased the biomass by 2.48 and 0.79 times compared with Medicago sativa and Brassica campestris cultivation treatments, respectively. Additionally, we found significant (P < 0.05) differences among the varieties of each green manure. For Medicago sativa, the biomass of Eureka+ was significantly (P < 0.05) increased by 10.38%, 8.29% and 14.88%, compared with that of Aurora, Sanditi, Sardi, respectively. For the biomass of Brassica campestris, there was no significant difference between Huayouza 82 and Huayouza 158, while they were both significantly (P < 0.05) higher than that of Huayouza 62, with increasing by 60.66% and 71.29%, respectively.
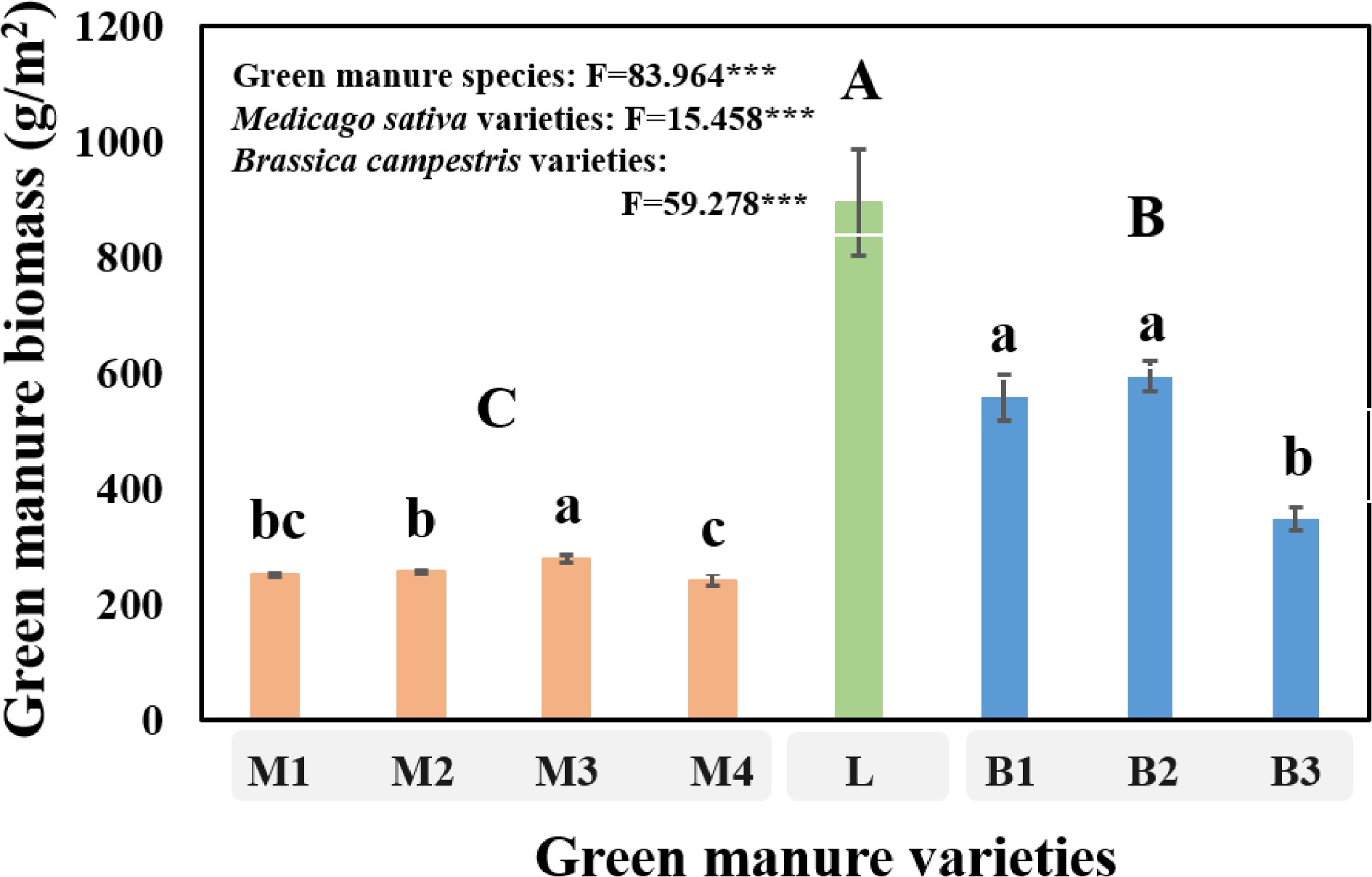
Figure 1.
The green manure biomass of different varieties cultivation at harvest in saline alkali soil field experiment. 'M1, M2, M3 and M4' represent Aurora, Sanditi, Eureka+, Sardi, respectively, which all belong to Medicago sativa. 'L' refers to Lolium multiflorum. 'B1, B2 and B3' represent Huayouza 82, Huayouza 158, Huayouza 62, respectively, which all belong to Brassica campestris. One-way analysis of variance (ANOVA) was used to assess the differences among treatments. Bars represent the mean values of three replicates ± SD. Values that do not share the same lower case letter are significantly different (P < 0.05) among green manure varieties. Values that do not share the same uppercase letter are significantly different (P < 0.05) at different green manure species (Medicago sativa, Lolium multiflorum, Brassica campestris). *** refers to P < 0.001.
Green manure carbon sequestration of different varieties cultivation
-
The carbon content was lowest in Medicago sativa cultivation treatment (401.58 g·kg−1), however, no significant difference was observed among Medicago sativa, Lolium multiflorum and Brassica campestris cultivation treatments (Fig. 2a). For Medicago sativa, the carbon content of Aurora (364.25 g kg−1) was significantly (P < 0.05) lower than that of Eureka+. For Brassica campestris, the carbon content of Huayouza 82 was significantly (P < 0.05) higher than that of Huayouza 158 and Huayouza 62 by 12.55% and 12.09%, respectively. In addition, different green manure cultivation led to a significant (P < 0.05) difference in aboveground carbon store (Fig. 2b). The carbon store in Lolium multiflorum cultivation (400.58 g·m−2) was significantly (P < 0.05) higher than that of Medicago sativa and Brassica campestris cultivation treatments by 2.87 and 0.91 times, respectively. For Medicago sativa, the carbon store of Eureka+ was significantly (P < 0.05) higher by 33.88%, 25.71% and 20.36% than that of Aurora, Sanditi and Sardi, respectively. Additionally, for Brassica campestris, the carbon store of Huayouza 62 (139.41 g·m−2) was largely significantly (P < 0.05) lower than that of Huayouza 82 (251.31 g·m−2) and Huayouza 158 (237.78 g·m−2), respectively.
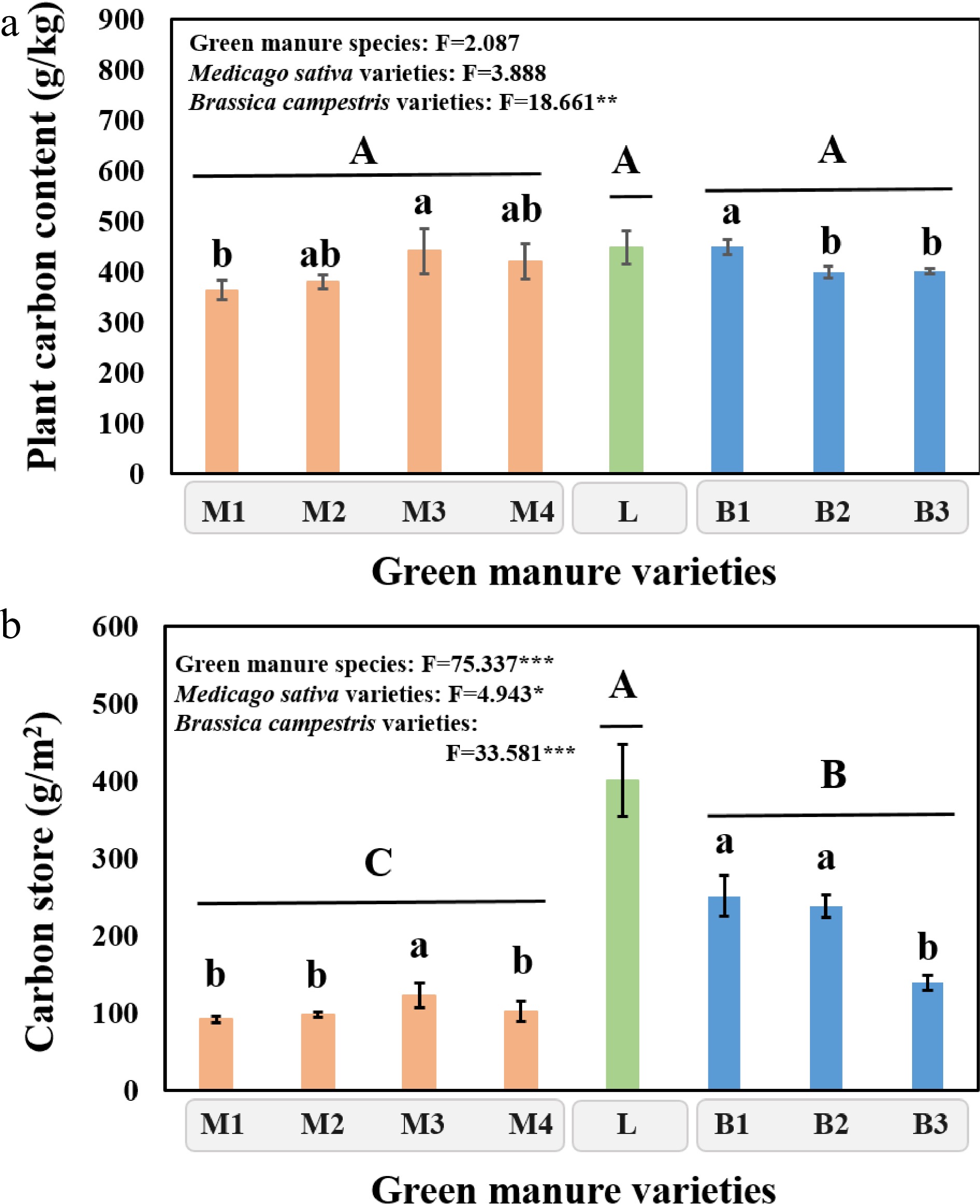
Figure 2.
(a) Carbon content and (b) carbon store of different varieties cultivated at harvest in saline alkali soil field experiment. 'M1, M2, M3 and M4' represent Aurora, Sanditi, Eureka+, Sardi, respectively, which all belong to Medicago sativa. 'L' refers to Lolium multiflorum. 'B1, B2 and B3' represent Huayouza 82, Huayouza 158, Huayouza 62, respectively, which all belong to Brassica campestris. One-way analysis of variance (ANOVA) was used to assess the differences among treatments. Bars represent the mean values of three replicates ± SD. Values that do not share the same lower case letter are significantly different (P < 0.05) among green manure varieties. Values that do not share the same uppercase letter are significantly different (P < 0.05) at different green manure species (Medicago sativa, Lolium multiflorum, Brassica campestris). * refers to P < 0.05; ** refers to P < 0.01; *** refers to P < 0.001.
Soil physicochemical properties in different cultivation treatment at green manure harvest
-
The green manure cultivation generated significantly different physicochemical properties in saline alkali soil (Table 1, Supplemental Table S1). The cultivation of Medicago sativa (Aurora, Sanditi, Eureka+) and Lolium multiflorum resulted in a significant (P < 0.05) lower pH by 6.32%, 3.67%, 1.69% and 4.03% than that of CK. Moreover, compared with CK, the EC in Medicago sativa (except for Sanditi) and Brassica campestris were also significantly (P < 0.05) declined. For soil organic matter, only Eureka+ and Huayouza 62 were significantly (P < 0.05) higher than that of CK. Additionally, soil total N in Sardi and Huayouza 62 were significantly (P < 0.05) lower than that of CK. For available N, Aurora, Sanditi, Sardi and Lolium multiflorum were significantly (P < 0.05) higher by 33.08%, 27.53%, 29.70%, 38.49%, when compared with CK.
Table 1. Soil properties measured under different treatment at green manure harvest in saline alkali soil field experiment.
Green manure cultivation Treatment pH EC
(us·cm−1)Soil organic matter
(g·kg−1)Total N
(g·kg−1)Available N
(mg·kg−1)No-cultivation CK 9.04 ± 0.16abc 496.33 ± 19.50b 5.27 ± 0.14bc 0.58 ± 0.05abc 38.76 ± 2.03bc Medicago sativa M1 8.50 ± 0.13f 429.00 ± 26.21cd 4.41 ± 0.78c 0.58 ± 0.01ab 51.58 ± 2.66a M2 8.72 ± 0.02e 470.33 ± 13.32bc 5.11 ± 0.42bc 0.54 ± 0.01cd 49.43 ± 4.35a M3 8.89 ± 0.03d 356.67 ± 6.43ef 5.63 ± 0.66a 0.54 ± 0.02bcd 41.89 ± 4.57b M4 8.93 ± 0.03cd 391.00 ± 4.36de 4.33 ± 0.24c 0.53 ± 0.02d 50.27 ± 1.61a Lolium multiflorum L 8.69 ± 0.04e 567.67 ± 52.88a 5.33 ± 0.46bc 0.60 ± 0.01a 53.68 ± 3.79a Brassica campestris B1 9.02 ± 0.04bc 363.00 ± 26.91ef 5.13 ± 0.11bc 0.51 ± 0.01de 37.53 ± 3.16bc B2 9.11 ± 0.02ab 330.67 ± 12.66f 5.07 ± 1.32bc 0.51 ± 0.02de 38.87 ± 2.33bc B3 9.17 ± 0.01a 328.00 ± 28.69f 6.00 ± 0.21a 0.49 ± 0.01e 34.97 ± 5.50c CK: no cultivation. 'M1, M2, M3 and M4' represent Aurora, Sanditi, Eureka+, Sardi, respectively, which all belong to Medicago sativa. 'L' refers to Lolium multiflorum. 'B1, B2 and B3' represent Huayouza 82, Huayouza 158, Huayouza 62, respectively, which all belong to Brassica campestris. Data are the mean values of three replicates. Numbers followed by '±' are the standard deviations (SDs). Within a column, values that do not share the same letter are significantly different (P < 0.05). Variation of soil physicochemical properties in different treatment after green manure returning to field for 30 days
-
The soil properties were measured after green manure was returned to the saline alkali soil field for 30 d. In general, the soil properties after green manure return (Table 2, Supplemental Table S2) were generally improved when compared with previous (Table 1). The returning of Medicago sativa (only Sardi), Lolium multiflorum and Brassica campestris (Huayouza 82, Huayouza 158, Huayouza 62) had significant (P < 0.05) reduction effects on soil pH, which reduced by 2.04%, 8.30%, 4.90%, 4.77% and 4.17%, respectively, when compared to no-returning (CK). Moreover, as compared with CK, only returning of Sanditi and Eureka+ significantly (P < 0.05) decreased soil EC content. In addition, the green manure return led to significant (P < 0.05) improvement on soil organic matter. Similarly, significant (P < 0.05) promoting effects on soil total N were also observed, expect for Sanditi, as compared to CK. The soil available N in Sardi, Lolium multiflorum, and Brassica campestris (Huayouza 82) were significantly (P < 0.05) increased by 30.55%, 57.24%, 23.22% than that of CK.
Table 2. Soil properties measured under different treatment after green manure return to saline alkali soil field for 30 d.
Green manure return Treatment pH EC
(us·cm−1)Soil organic matter
(g·kg−1)Total N
(g·kg−1)Available N
(mg·kg−1)No-returning CK 9.00 ± 0.14ab 591.00 ± 56.45bcd 5.84 ± 0.74b 0.52 ± 0.01c 39.80 ± 1.88d Medicago sativa M1 8.89 ± 0.07bc 536.33 ± 32.59d 7.92 ± 1.24a 0.60 ± 0.01b 41.99 ± 4.37cd M2 9.11 ± 0.03a 407.33 ± 43.25e 8.55 ± 0.80a 0.54 ± 0.05c 45.70 ± 4.68bcd M3 9.00 ± 0.02ab 438.33 ± 49.08e 8.36 ± 0.59a 0.60 ± 0.02b 47.24 ± 4.26bcd M4 8.82 ± 0.02c 549.67 ± 15.28cd 7.76 ± 0.55a 0.63 ± 0.02b 51.96 ± 1.87b Lolium multiflorum L 8.31 ± 0.12e 519.00 ± 14.53d 7.72 ± 0.88a 0.71 ± 0.02a 62.58 ± 5.98a Brassica campestris B1 8.58 ± 0.05d 621.67 ± 5.13bc 7.75 ± 0.44a 0.62 ± 0.02b 49.04 ± 8.57bc B2 8.59 ± 0.07d 701.33 ± 71.44a 7.30 ± 0.46a 0.61 ± 0.01b 42.43 ± 3.22cd B3 8.64 ± 0.01d 627.67 ± 35.53b 7.61 ± 0.28a 0.68 ± 0.03a 48.57 ± 3.73bcd CK: no cultivation. 'M1, M2, M3 and M4' represent Aurora, Sanditi, Eureka+, Sardi, respectively, which all belong to Medicago sativa. 'L' refers to Lolium multiflorum. 'B1, B2 and B3' represent Huayouza 82, Huayouza 158, Huayouza 62, respectively, which all belong to Brassica campestris. Data are the mean values of three replicates. Numbers followed by '±' are the standard deviations (SDs). Within a column, values that do not share the same letter are significantly different (P < 0.05). Effects of different returning treatment to improve soil quality in saline alkali field experiment
-
The descriptions of detailed scoring function values and weights assigned to the selected soil fertility parameters are available in Table 3. The weights of pH, EC, soil organic matter, total N and available N were 0.25, 0.23. 0.07, 0.23 and 0.22, respectively. Based on these, the soil fertility index in each returning treatment was calculated. As shown in Fig. 3, we observed significant (P < 0.001) difference between the non-cultivation and cultivation group. In the cultivation group, returning Lolium multiflorum to the field had the best soil fertility enhancing effect (0.56), which significantly (P < 0.05) improved soil fertility by 55.56% and 33.33% compared with Medicago sativa and Brassica campestris. For Medicago sativa, the soil fertility index of Sardi was significantly (P < 0.05) higher than that of the other three varieties. However, there was no significant difference among Brassica campestris varieties.
Table 3. Scoring function values and weights assigned to selected soil fertility parameters.
pH (H2O) EC
(us·cm−1)Soil organic matter
(SOM, g·kg−1)Total nitrogen
(TN, g·kg−1)Available nitrogen
(AN, mg·kg−1)Scoring curve# a a b b b Turning point U 9 1,500 15 1.2 120 L 6.0 100 5 0.5 30 O1 6.5 300 O2 8 400 weight 0.25 0.23 0.07 0.23 0.22 'a' Refers to the 'optimum' curve equation; 'b' refers to the 'more is better' curve equation. 
Figure 3.
Soil fertility index of different treatment that returning to the field in saline alkali soil experiment. One-way analysis of variance (ANOVA) was used to assess the differences among treatments. Bars represent the mean values of three replicates ± SD. Values that do not share the same lower case letter are significantly different (P < 0.05) among green manure varieties. Values that do not share the same uppercase letter are significantly different (P < 0.05) at different green manure species (Medicago sativa, Lolium multiflorum, Brassica campestris). ** refers to P < 0.01; *** refers to P < 0.001.
Contributions of green manure varieties, soil physicochemical properties, plant C, green manure biomass on soil fertility improvement
-
PLS-PM analysis was employed to identify direct and indirect effects of different green manure cultivation and returning to the field on saline alkali soil fertility improvement (Fig. 4a). Green manure varieties significantly (P < 0.05) positively affected biomass (0.76 of the direct effects) and then positively (P < 0.001) affected soil fertility (0.72). Similarly, green manure varieties significantly (P < 0.001) positively affected slow-changing soil properties (0.78), then positively (P < 0.05) affected plant C (0.81), followed with affecting biomass (0.55), and finally affected soil fertility. Furthermore, we observed fast-changing soil properties could significantly (P < 0.01) and directly affect biomass (0.91), while slow-changing soil properties had no significant effect on biomass.
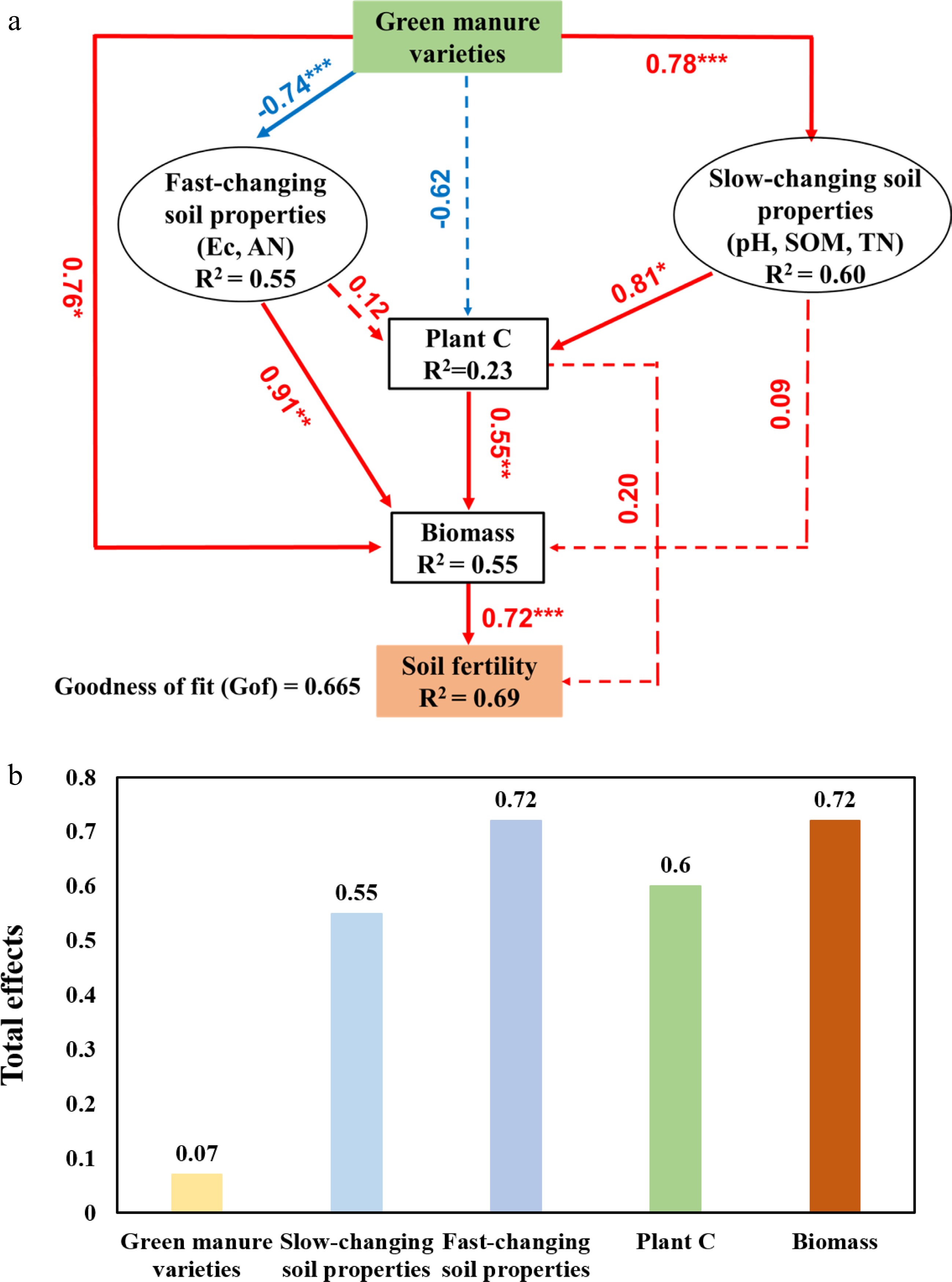
Figure 4.
Cascading relationships of soil fertility with green manure and soil physicochemical properties. (a) Partial least squares path modelling (PLS-PM) disentangling major pathways of the influences of green manure varieties, soil physicochemical properties, plant C, green manure biomass on soil fertility. Red and blue arrows indicate positive and negative flows of causality, respectively. Solid and dashed lines indicate significant (*, P < 0.05; **, P < 0.01; ***, P < 0.001) and nonsignificant (P > 0.05) levels, respectively. Values on arrows indicate significant standardized path coefficients. R2 indicates the variance of dependent variable explained by the model. (b) Total effects of soil fertility in the PLSPM models for green manure varieties, fast-changing soil properties, slow-changing soil properties, plant C, and green manure biomass.
Overall, we found that all variates (green manure varieties, slow-changing soil properties, fast-changing soil properties, plant C and biomass) had positive impacts on soil fertility improvement. Among these, both fast-changing soil properties and biomass exhibited the greatest positive impacts (0.72 of the total effects). In addition, the total effects of fast-changing soil properties (0.552) was greater than slow-changing soil properties (0.55). Plant C also largely contribute to saline alkali soil fertility improvement (0.60). Green manure varieties had the lowest but also positive impact (0.07) (Fig. 4b).
-
Previous studies[38−41] have demonstrated that green manure, with positive effects on soil improvement, is a main and feasible approach for sustainable crop production. In the present study, we conducted green manure varieties cultivation experiments in saline-alkali soil. Here we observed that the biomass of different salt-tolerant green manure was significantly different, and there were also certain differences among different varieties of the same green manure, which indicated that both green manure species and cultivars contributed to biomass. Son[42] revealed that the biomass of green manure crop was the highest in ryegrass, which was consistent with our result. Monirifar el al.[43] assessed the effects of cultivar on alfalfa yield in saline conditions, and found that cultivar selections could largely influence the yield. Meza et al.[44] also demonstrated that total plant biomass, aboveground biomass, root biomass were all influenced by forage species. These all supported our results. Establishment of plants provides an important opportunity to store C in both plant biomass and the soil to mitigate climate change[45]. Thus, the plant carbon store was closely correlated with biomass and C content. It is a good way to improve carbon sequestration by increasing plant biomass, especially when there is no difference among plant C contents. Besides, biomass also significantly affected the deposition of photosynthetically-fixed C into the plant-soil system[16]. Although aboveground C store largely contribute to the whole carbon pool, it’s better to evaluate the aboveground and belowground together. We should pay attention to carbon sequestration from a more holistic perspective in our further research.
Soluble salt content and pH value can reflect the degree of soil salinization, which are important evaluation indexes of saline-alkali soil[46]. Gelaye et al.[47] employed rhodes grass, alfalfa, sudangrass and blue panicgrass to cultivate them in saline field plots, and found they affected pH and mitigated soil salinity and soluble saline ion concentrations. Our study showed that the green manure cultivation reduced soil pH, among which, Medicago sativa and Lolium multiflorum had a better effect on reducing soil pH than Brassica campestris. Additionally, except Lolium multiflorum, salt-tolerant plant cultivation could effectively reduce soil EC. The planting of salt-tolerant green manure had an obvious inhibition effect on the surface soil salinity of soil, which might be due to the planting reducing the surface evaporation and keeping the salt in the subsoil or some salt was taken up and carried out of the soil by salt-tolerant grasses. This is consistent with the results obtained by Liu et al.[48] that planting salt-tolerant forage grass can reduce soil salinity and pH value. For soil nutrients, certain cultivation treatments increased soil organic matter (Eureka+, Huayouza 62) and available nitrogen contents (Aurora, Sanditi, Sardi, Lolium multiflorum), which was consistent with Jing et al.[49] that the planting of M. sativa, S. sudanense, S. bicolor, and P. frumentum had significant enhancement on soil organic matter and available nutrient contents in inland saline soil. Astier et al.[50] also demonstrated that when vetch and oat were planted, soil organic matter and total N increased. The growth of legumes does not always increase the content of total nitrogen in the soil[25], which supported our result that the total soil nitrogen after Medicago sativa and Brassica campestris cultivation decreased to different degrees, as compared with non-cultivation, mainly due to the fact that green manure needs to absorb a large amount of a variety of nutrients from soil during its growth and development, and its consumption of nitrogen increases, resulting in a decrease in total nitrogen content.
After the green manure was returned to the saline alkali field, the pH of the majority treatments were decreased, which might be related to the different dry matter composition of different green manure varieties and their different degradation and transformation rates in soil[51, 52]. Cao et al.[53] also found significant reduction in pH was determined under field conditions of 4 years of alfalfa cultivation in salt-affected soils. The content of soil organic matter, total nitrogen and available nitrogen in cultivation treatments were increased to different degrees compared with the control, indicating that returning green manure to the field had an obvious effect on soil fertility improvement. For Lolium multiflorum and Brassica campestris, their biomass was large, thus the nutrients that carried and were released into the soil were also correspondingly large. For Medicago sativa, except the biomass, a large amount of nitrogen was returned to the soil based on the nitrogen fixation effect to increase the nitrogen content in the soil. Previous studies[46, 54] demonstrated that a variety of soluble organic matter would be produced in the decomposition process, which could promote the efficient cycling of soil nutrients and regulate the soil nutrient balance. The comprehensive soil fertility quality index is a quantifiable index, which indicates a soils ability to perform specific ecological functions[55]. The difference of soil fertility index among different treatments indicates that returning green manure to the field is a good way to improve saline alkali soil quality. Also, the biomass amount of green manure species largely contributed to the soil fertility improvement during the process of returning to the field. Ma et al.[56] generated a comprehensive evaluation of green manure on soil properties based on a meta-analysis and showed their significant improvement effects on soil quality, which was consistent with our research.
According to our PLS-PM analysis, green manure varieties, slow-changing soil properties, fast-changing soil properties, plant C and biomass make a good explaination for soil fertility improvement after plants are returned to the field. We summarized two ways from the process of cultivation to returning to the field and found plant biomass was the core variate to improve soil fertility. Moreover, plant biomass had the greatest influence on soil fertility, suggesting that the increase of soil fertility mainly depended on the biomass of green fertilizer returned to the field. Thus, we concluded that Lolium multiflorum that has a large aboveground biomass was the best potential variety to improve saline alkali soil fertility. Except for plant biomass, fast-changing soil properties, mainly available N, also had an indirect, but large, influence on soil fertility. Feng et al.[57] found that high soil nutrient availability has a positive effects on the alfalfa biomass. Accumulated evidence suggests that fertilization and biomass are strongly correlated in agricultural ecosystems[58]. Zhang et al.[59] demonstrated that N fertilizer application directly promoted ryegrass yield. Therefore, we should pay more attention to improve aboveground biomass to ameliorate soil quality in saline alkali soil in the future.
-
We employed different green manure variates to measure the effects of cultivation for aboveground carbon sequestration and returning to the field to ameliorate soil quality in saline alkali soil, based on a field experiment. We determined that plant carbon store was positively correlated with aboveground biomass. Green manure varieties, slow-changing soil properties, fast-changing soil properties, plant C and biomass all contributed to soil fertility improvement after aboveground returning to the field. The biomass production was a determining factor contributing to soil fertility, and variety with higher biomass production would more effectively improve soil quality in saline alkali soil. Our study can provide crucial theoretical support and a feasible way for green and sustainable development of saline-alkali agriculture.
We thank Zhibo Zhou for his assistance in writing and data analysis. This research was financially supported by the Key Research & Development Plan of Jiangsu Province (BE2021365) and Jiangsu Science and Technology Project (SZ-SQ2021060).
-
The authors declare that they have no conflict of interest.
- Supplemental Table S1 ANOVA F-values of soil properties measured under different treatment at green manure harvest in saline alkali soil field experiment
- Supplemental Table S2 ANOVA F-values of Soil properties measured under different treatment after green manure returning to saline alkali soil field for 30 days
- Copyright: © 2023 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Zhang F, Han Y, Shang H, Ding Y. 2023. Effects of green manure cultivation for aboveground carbon store and returning to the field to ameliorate soil quality in saline alkali soil. Grass Research 3:1 doi: 10.48130/GR-2023-0001
Effects of green manure cultivation for aboveground carbon store and returning to the field to ameliorate soil quality in saline alkali soil
- Received: 15 November 2022
- Accepted: 27 December 2022
- Published online: 18 January 2023
Abstract: The utilization of saline-alkali land together with the consideration of the productive value (improving soil productivity) and ecological value (increasing carbon store ability) has rarely been reported. We conducted a field experiment to investigate the impact of green manure cultivation for aboveground carbon (C) store and then returning this to field to improve soil quality in saline alkali soil. The biomass in Lolium multiflorum cultivation treatment was significantly (P < 0.05) higher than that of Medicago sativa and Brassica campestris cultivation. A similar tendency was observed in aboveground C store. Green manure cultivation resulted in largely different physicochemical properties at time of harvest. Returning the green manure to the field could significantly (P < 0.001) improve soil fertility. Moreover, the soil fertility index of Lolium multiflorum treatment was significantly (P < 0.05) enhanced by 55.56% and 33.33%, as compared with Medicago sativa and Brassica campestris treatments. Based on PLS-PM analysis, both fast-changing soil properties and biomass exhibited the greatest positive impacts (0.72 of the total effects) on soil fertility improvement after aboveground returning to the field. Our research provides evidence that Lolium multiflorum is the best potential variety to improve saline alkali soil fertility. Additionally, green manure cultivation in saline-alkali soil is an important way to store carbon in plants, then returning to the field is a feasible approach to improve saline alkali soil quality, which is beneficial for the green and sustainable development of saline-alkali agriculture.













