-
Tea is one of the most consumed nonalcoholic beverages worldwide. It is well known for its charming flavor, leisure characteristics, and potential health benefits, such as antioxidative, antimutagenic, and anticancer effects[1]. Recent international standard (ISO 201715: 2023) shows that tea products can be classified into six types: black, green, oolong, dark, white, and yellow teas. These tea products are made from Camellia sinensis (L.) O. Kuntze tender leaves via different manufacturing processes[2]. As of the end of 2022, green tea production in China occupied 58.8% of the total tea production, while black tea production accounted for about 15.2%.
Interestingly, black tea consumption accounts for 80% of the total tea commerce in the world[3], especially in India, Sri Lanka, Kenya, Turkey, and Bangladesh, where black tea processing is dominant. According to the new international standard, black tea products contain Orthodox black tea, broken black tea, congou black tea, and souchong black tea. Orthodox black tea is a traditional tea product in the international trade market. It comprises twisted strip/wiry leaf, shotty/curly/semi-curly Pekoe, bud tips, small slices, and powdered tea, such as Turkey black tea[4] and Orthodox Keemun black tea[5]. Congou black tea processing usually contains a rolling stage after withering, while Souchong black tea has a particular stage after fermentation. A pine needle or branch flame gives a smoky taste and smell to souchong tea products, such as Lapsang Souchong[6]. Hence, the appearance of Congou and Souchong black tea can be observed as complete buds or leaves, differing from broken black tea[7].
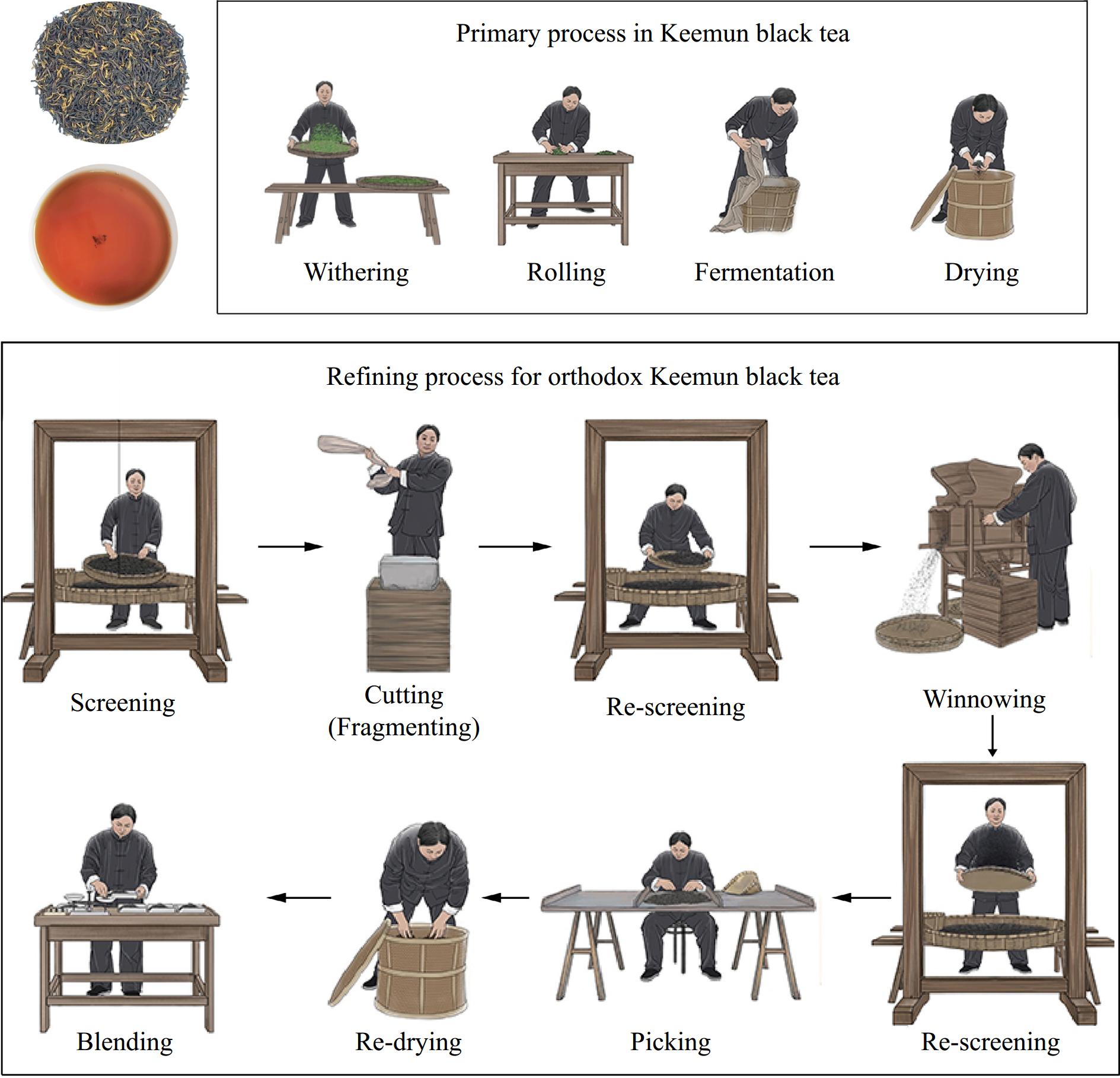
Keemun Congou black tea, like Orthodox black tea, has a different volatile profile from Jinzhen, Maofeng, and Xiangluo Keemun black tea, as described in our previous study[5]. In the initial Keemun black tea production period, refining, and blending stages are necessary for balancing tea quality and yield. A complex manual manufacturing process refined primary black teas processed by different regions, seasons, and maturity (Fig. 1). Based on their experience, tea experts make up different black tea grades according to the size, length, and thickness of the original materials. Tea processing using machines gradually replaced that using manual, and curiosity for tea products of superior quality made tea companies pursue new black tea products, such as Gold Jun Mei black tea[8] and Keemun Jinzhen black tea[5], with the development of economy and tea industry.
These premium black tea products are usually made from tender buds (one bud or one bud and one leaf) before the Qingming Festival (April 5th) and then undergo a simple refining process (screening, winnowing, picking, and blending), starting to flow into tea commerce. However, Keemun Congou black tea (Orthodox Keemun black tea) often undergoes a long-time refining stage (2‒3 months) before occurring in the tea market. Our previous study showed that Keemun Congou black tea with a special refining stage has a high proportion of flowery, fruity, and sweet odorants[5]. Herein, a targeted metabolomic technique with ultra-performance liquid chromatography connected to an ESI-triple quadrupole linear ion trap mass spectrometry system (UPLC–QTRAP–MS/MS) was applied to obtain nonvolatile profiles of Keemun black tea.
The nonvolatile compounds (e.g., amino acids, catechins, and flavonoids) are closely related to taste characteristics. Its profile in black tea is quite different from other tea types. For instance, total amino acid content accounts for only 46% of the original in black tea products, while 75% of that can be detected in green tea products[9]. The total catechin concentration also decreased significantly during black tea processing. In contrast, their polymers, such as theaflavins (TFs), thearubigins (TRs), and theabrownine (TB), were highly concentrated[10,11]. These nonvolatile catechins, caffeine, theaflavins, and amino acids are usually detected using liquid chromatography[12]. In contrast, applying a mass spectrometry system often requires identification of flavonoids, flavonoid glycosides, or other low concentration compounds that contribute to tea taste. A total of 203 flavonoids were identified in Congou black tea using the UPLC–QTRAP–MS/MS technique[13]. The UPLC system, coupled with a time-of-flight mass spectrometer (UPLC–Q–TOF–MS), detects 58 critical nonvolatile metabolites in Keemun black tea[14]. The trans-4-O-p-coumaroylquinic acid, kaempferol-glucoside, quercetin-glucoside, and kaempferol 3-O-rutinoside were identified as dominant taste compounds in Keemun black tea infusions using the mass spectrometry technique[15], especially the trans-4-O-p-coumaroylquinic acid, which gives a strong astringent taste to tea infusion. Theaflavin-glucose adducts were also identified using the UPLC–Q–TOF–MS technique[16].
A previous study showed that Keemun Congou black tea has a different volatile profile from other Keemun black teas[5]. In this study, 48 Keemun black tea samples (12 samples for each black tea type) were comprehensively analyzed by UPLC–QTRAP–MS/MS. The study aimed to (i) profile the nonvolatile metabolites of the four Keemun black tea types and (ii) explore the differential metabolites and effects of the manufacturing process on the nonvolatile profile.
-
Forty-eight black tea samples from the Qimen area (latitude 29°51', longitude 117°43') were kindly gifted by the Qimen Tea Association, including 12 samples for each tea type (Congou, Maofeng, Xiangluo, and Jinzhen Keemun black teas). The manufacturing process of these four black tea types has been elucidated in our previous study[5]. They all were processed by one bud and one leaf or one bud and two leaves from early to mid-April. Tea experts evaluated these tea samples and ensured they had the characteristic Keemun black tea flavor.
Sensory evaluation
-
Six tea experts, including two men and four women, were recruited to conduct the sensory evaluation based on the taste part of the National Standard of the People's Republic of China (GB/T 23776−2018). Tea infusions (3 g of tea samples, 150 mL boiling water, 5 min) were prepared. Taste scoring was followed as 90−99 (with umami, mellow, and umami, or sweet and mellow characters), 80−89 (with mellow character), and 70−79 (with barely mellow character). The umami and sweet taste can be described as having a positive effect on the human tongue (sweet aftertaste); the mellow and thick taste of tea infusions are used to describe the strong, or weak.
Chemicals
-
Catechin standards were purchased from Shanghai Yuanye Bio-Technology Co., Ltd (Shanghai, China) including gallic acid, catechin (C), epicatechin (EC), epigallocatechin (EGC), gallocatechin (GC), gallocatechin gallate (GCG), epigallocatechin gallate (EGCG), epicatechin gallate (ECG), catechin gallate (CG), theaflavin, theaflavin-3-gallate, theaflavin-3'-gallate, and theaflavin-3,3'-di-O-gallate. The mixture of amino acid standards were purchased from Sykam GmbH (Munich, Germany). Flavonoids and other standards were purchased from Shanghai Macklin Biochemical Technology Co., Ltd (Shanghai, China), including rutin, quercetin, vitexin, myricetin, myricetin 3-O-galatoside, kaempferol-3-O-glucoside quercetin-3-O-glucoside, kaempferol-3-O-rutinoside, quercetin-3-O-glucosylrutinoside, proanthocyanidin B1, proanthocyanidin B2, proanthocyanidin B3, proanthocyanidin B4, β-glucogallin, and quinic acid.
Catechins detection by HPLC
-
Catechins were extracted and analyzed according to a previous study[17]. Nine typical samples for each tea type were selected for catechins analysis. A high-performance liquid chromatography system (HITACHI, Tokyo, Japan) coupled with a C18 column (5 μm × 4.6 mm × 250 mm; Phenomenex, CA, USA) was used. The mobile phases, flow rate, detection wavelength, and column temperature were 0.04% (v/v, phosphoric acid/distilled water) phosphoric acid solution (A) and 100% acetonitrile (B), 1.0 mL/min, 280 nm, and 40 °C, respectively. The gradient elution was the same as in a previous study[17]. The identification and quantitation (mg/g, dry weight, DW) of catechins were compared and calculated with their authentic standards and calibration curves. Three technical replicates were conducted.
Amino acid analysis
-
Amino acid extracts were prepared according to a previous study[17]. An automatic amino acid analyzer S-433D (SYKAM, Munich, Germany) coupled to an LCA K07/Li column (SYKAM, Munich, Germany) was applied. The elution and derivation flow rates were maintained at 0.45 and 0.25 mL/min. The mobile phases were purchased from SYKAM, and the gradient elution was the same as in a previous study[17]. The identification of amino acids was conducted by comparing their authentic standards. Quantitation (mg/g, dry weight, DW) was calculated by comparing the sample peak areas and analytical standards. Nine typical samples for each tea type were selected for amino acid analysis and three technical replicates were measured.
Theaflavins, thearubigins, and theabrownine determination
-
Tea-pigment separation and extraction were performed according to previous studies[10,11]. In the present study, nine typical samples for each tea type were selected for pigment analysis.
Tea infusions were prepared (6 g of tea samples, 250 mL of boiling water). After a hot-water bath for 10 min, the aqueous extract was filtered using degreasing cotton. After cooling to room temperature, 25 mL aliquots were put into a separating funnel, and 25 mL ethyl acetate was added. It was gently shaken for 5 min manually and placed still for layering. The lower and upper layers were collected in triangle beakers, respectively.
The A solution comprised 2 mL of the upper layer and was placed into a 25 mL volumetric flask using 95% ethyl alcohol up to 25 mL. The B solution was composed of 2 mL of the lower layer. It was placed into a 25 mL volumetric flask, adding 2 mL oxalic acid solution and 6 mL of distilled water, and then using 95% ethyl alcohol up to 25 mL. The C solution was composed of 15 mL of the upper layer, put into a separating funnel, and added 15 mL of 2.5% sodium bicarbonate solution. It was quickly shaken for 30 s manually and placed still for layering. In this step, 4 mL of the upper layer formed was collected in a 25 mL volumetric flask using 95% ethyl alcohol up to 25 mL. The D solution comprised 25 mL tea infusions, put into a separating funnel and 25 mL of n-butyl alcohol was added. It was manually shaken for 3 min and placed still for layering. 2 mL of the lower layer formed in this step was put into a 25 mL volumetric flask, added 2 mL oxalic acid solution and 6 mL distilled water, and then using 95% ethyl alcohol up to 25 mL. OD values of the four solutions were measured at 380 nm using an ultraviolet spectrophotometer (TU-1810, Persee, Beijing, China) and recorded as EA, EB, EC, and ED. Theaflavins, thearubigins, and theabrownin in tea samples were calculated according to the following formulas: Total theaflavins (%) = 2.25 × EC; Thearubigins (%) = 7.06 × (2EA + 2EB − EC − 2ED); Theabrownin (%) = 2 × ED × 7.06.
Soluble sugar detection
-
Soluble sugar content in tea infusions was analyzed according to anthrone colorimetry. In this step,100 mg of tea powder was put into a 25 mL test tube, added 15 mL of water, and boiled in a water bath for 20 min. After cooling to room temperature, tea infusions were placed into a 100 mL volumetric flask and up to the volume using distilled water. A total of 1 mL aliquots and 5 mL 1 g/L anthrone solution were put into a 25 mL test tube and boiled in a water bath for 10 min. After cooling to room temperature, the solution was measured at 620 nm using an ultraviolet spectrophotometer (TU-1810, Persee, Beijing, China). Soluble sugar concentrations (%) in the tea samples were calculated according to the glucose calibration curves.
Non-volatile compounds detected by UPLC-QTRAP-MS/MS
-
Aqueous infusions were prepared according to a previous study[18]. Tea powder (0.06 g) was extracted with 1.8 mL of 80% methanol solution. The aqueous infusions were centrifuged at 12,000 rpm for 10 min and the residues were re-extracted with 1.8 mL of 80% methanol solution, as above. Aqueous infusions were filtered through a 0.22 μm membrane before the LC-MS analysis. All 48 black tea samples were analyzed, and a triple extraction was performed for each sample.
All samples were analyzed using a Q-Exactive Focus Orbitrap LC-MS/MS System (Thermo Fisher Scientific, USA). The LC column was 3 mm × 150 mm × 2.7 μm C18 (Poroshell 120 SB-C18, Agilent, California, USA). The mobile phases were 0.4% acetic acid (V/V, acetic acid: distilled water, A) and 100% acetonitrile (B). The flow rate, column oven temperature, and injection volume were set at 0.5 mL/min, 30 °C, and 10 μL, respectively. The gradient elution was as follows: 0.1% B (0 min), 7% B (0.01−10 min), 7% B (10.01−22 min), 11% B (22.01−25 min), 12% B (25.01−30 min), 14% B (30.01−31 min), 35% B (31.01−43 min), 80% B (43.01−44 min), 80% B (44.01−46 min), and 0.1% B (46.01−47 min).
The MS operating conditions were 8 L/min drying gas nitrogen flow at 325 °C, 11 L/min sheath gas flow at 350 °C, 45 psi nebulizer pressure, and 3,500 V capillary voltage. A negative ionization mode was performed. The full MS parameters were set as follows: 70,000 resolution, 5e6 AGC target, 200 ms maximum IT, and 80‒1,200 m/z scan range. The dd-MS2 parameters were set as follows: 17,500 resolution, 1e5 AGC target, 60 ms maximum IT, five loop count, and 0.4 m/z isolation. The collision energy was set at 15−40 V. The acquired MS and MS/MS data files were analyzed using Thermo Xcalibur 4.1 and TraceFinder 4.1.
Identification and quantification of nonvolatile compounds
-
The identification of nonvolatile compounds in this study was performed using precursor ions, MS/MS fragments, and retention times. The precursor ion and MS/MS fragment messages of nonvolatile compounds were obtained from previous studies[13,14,18] and standards used in this study. The retention times of 28 compounds described previously wereidentified according to compared with their authentic standards, while other compounds were confirmed in a previous study[18].
A local database was established according to compound name, chemical formula, precursor m/z, polarity, and retention time in Thermo TraceFinder 4.1 software. Compound integration was performed based on this newly established database. Compound quantification was calculated based on the peak areas of myricetin (10 mg/L).
Thermal reaction of MF black tea
-
Among 12 MF tea samples, one of them was selected to perform the thermal reaction. A drying oven (Zhejiang Sunyang Machinery Co., Ltd, Quzhou, China) was used to conduct three thermal treatments (90 °C, 2 h; 130 °C, 2 h; 150 °C, 1 h). After thermal treatment, the above black tea samples were analyzed by UPLC-QTRAP-MS/MS.
Black teas processed by tea buds, leaves, and stems
-
Fresh shoots (one bud and two leaves) of tea variety (Camellia sinensis var. sinensis cv. Fuzao2) were plucked in the middle of April, and then the tea bud, leaf, and stem were separated manually. The three fresh materials were processed into black tea, respectively. The withering stage was performed at 28−30 °C for 6−7 h, and a rolling stage was conducted using a rolling machine for 55 min. The fermentation procedure was performed at 23−25 °C for 2−3 h. Drying was performed at 110 °C for 5−7 min, room temperature for 20−30 min, and 85 °C for 20−30 min.
Statistical analysis
-
Significant differences (p < 0.05) between tea samples were conducted using one-way analysis of variance (ANOVA) and Duncan's multiple range tests in SPSS 20.0 (SPSS Inc., USA). A principal component analysis (PCA) and variable importance for the projection (VIP) plot were determined using SIMCA 14.1 (Umetrics Corporation, Sweden).
-
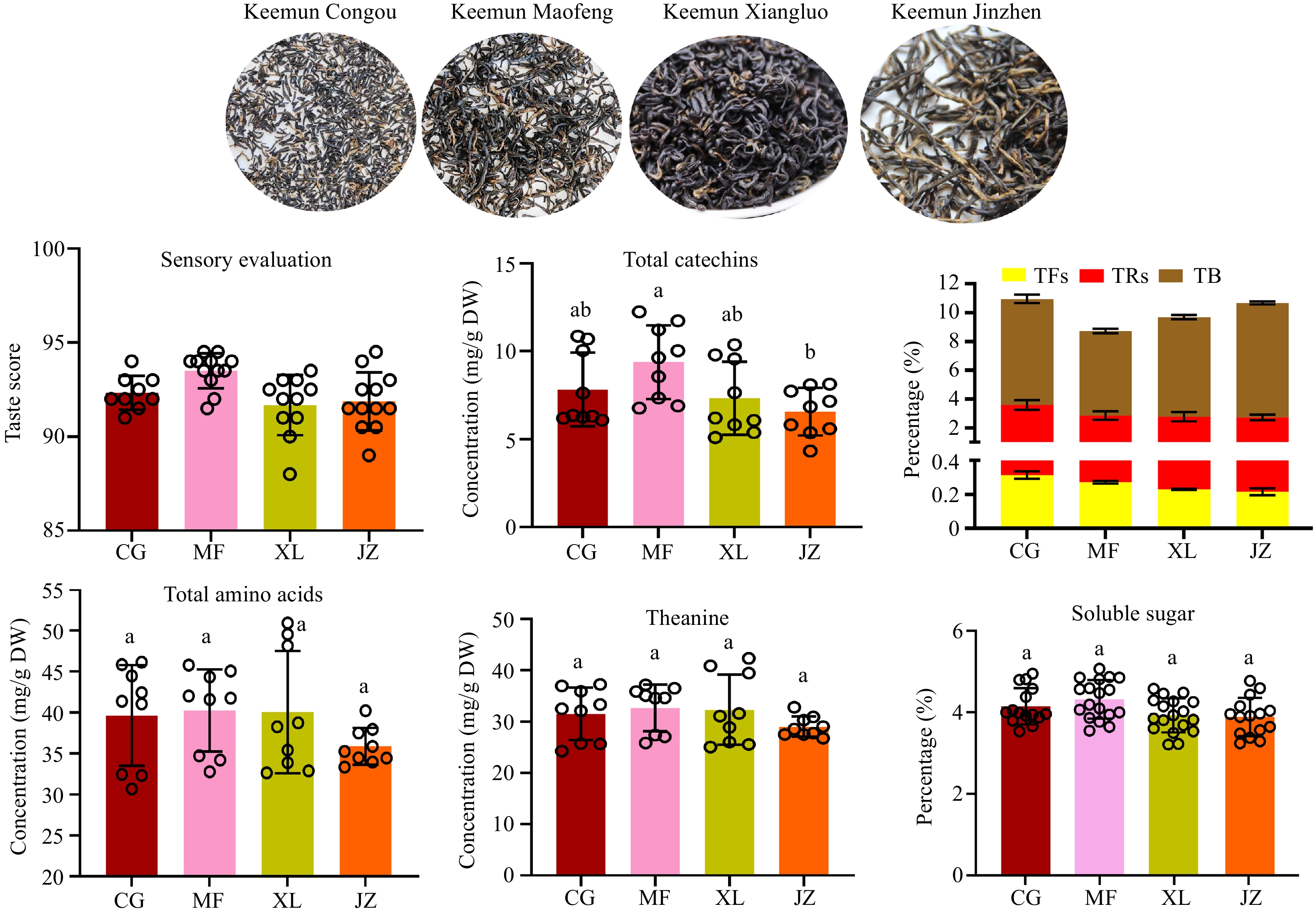
Orthodox Keemun black tea (Keemun Congou) had a different appearance from the other three Keemun teas (Fig. 2). Based on sensory evaluation, Keemun Maofeng black teas had a high intensity of sweet-after-taste and reached the most elevated taste score (93.5 ± 1.8), followed by Keemun Congou (92.2 ± 1.5), Jinzhen (91.8 ± 1.5), and Xiangluo ( 91.8 ± 1.6).
Tea catechins, as the main nonvolatile compounds in tea plants, are easily oxidized to form polymeric polyphenols during black tea processing, such as TFs, theasinensins, TRs, and TB[19]. Total catechins concentration showed no significant difference among CG, MF, and XL. Still their concentration in JZ was significantly lower than in MF (Fig. 2). Catechins were highly concentrated in Maofeng Keemun teas, while their polymer, tea pigments were less concentrated. The relative content of theaflavins in JZ and XL was lower than in CG and MF black teas, while the TRs content had no significant difference in MF, XL, and JZ, except for CG. The TRs/TFs ratio ranging from 10−12 is a good index for black tea quality[20]. The TRs/TFs ratio in four Keemun black teas (CG, 10.5; MF, 9.6; XL, 11.1; JZ, 11.6) were different, but all within the good index ranges, suggesting that all four Keemun black tea types are good quality. Theaflavins and D-glucose can produce adducts in thermal reactions[16]. To shape the curly or straight appearance, a long thermal response occurs in the manufacturing process of Jinzhen and Xiangluo black teas, decreasing theaflavins concentration. Furthermore, the TB/TRs ratio in Jinzhen (3.2) and Xiangluo (2.7) was higher than in Congou (2.2) and Maofeng (2.3) black teas, suggesting that the polymerization of catechins was also the reason for the decrease of theaflavins concentration.
Total amino acid concentration showed no significant difference between the four Keemun black tea types except for Jinzhen black teas. Theanine is the most abundant non-protein amino acid in tea and is an important contributor to the umami taste of the tea. It also contributes to a tea-roasted peanutty aroma when it reacts with sugar-producing methylpyrazine and 2,5-dimethylpyazine[21]. Theanine concentrations in JZ teas were significantly lower than in the others (Fig. 2). Given that the shaping stage of Jinzhen black tea processing requires a thermal reaction, amino acids and carbohydrates undergo a Maillard reaction, leading to a decrease in amino acid concentrations[22]. Subsequently, the soluble sugar content in tea infusions was measured. The soluble sugar content in JZ significantly decreased compared to MF teas. The high temperature shaping stage (110−130 °C) has been suggested to result in a Maillard reaction between theanine and glucose in JZ tea processing. This could also explain why MF teas had a high intensity of sweet-after taste in sensory evaluation.
A previous study showed that catechins, theobromine, and theaflavins concentrations were below their corresponding threshold concentrations in Keemun black tea infusions (W/V, 1:50). Still, caffeine had a high dose over the threshold (DoT) value (2−3)[15]. The DoT value of nonvolatile is greater than one, indicating that this compound may contribute more to the taste of tea infusion. Caffeine concentration showed no significant difference between the four Keemun black tea types, in which caffeine concentration ranged from 39.5 to 41.1 mg/g. Flavonoid glycosides give a mouth-drying and velvety-like astringency to the human tongue[22], especially quercetin-3-O-rhamnoyranosyl-β-D-glucopyranoside (rutin) with a 0.00115 μM threshold value[23]. Hence, UPLC-QTRAP-MS/MS was applied to analyze flavonoids and their glycosides in the current study.
Flavonoids and their glycosides analysis by LC-MS/MS
-
In negative ionization mode, nonvolatiles detected in this study produced an intense signal of [M‒H]‒ ion. Compared with the [M‒H]‒ precursor ion, MS/MS fragment, retention time, and 87 metabolites were identified, including 28 metabolites having authentic standards and 59 nonvolatiles identified by previous studies[13,14,18]. They comprised 12 simple catechins, 12 catechins dimers or polymers, 11 organic acids, 37 flavonoids and their glycosides, five proanthocyanidins, and 10 theaflavins. Their concentration was calculated by comparing the myricetin (external standard) peak area. Supplemental Table S1 lists the relative concentration of 87 compounds in tea samples. Their total relative concentration was different in the four Keemun black tea types. Maofeng teas have the highest average content (4,523 mg/kg), followed by Congou (4,393 mg/kg), Xiangluo (3,659 mg/kg), and Jinzhen (3,688 mg/kg).The ANOVA results showed that the total relative concentration in the Congou and Maofeng groups was significantly higher than in the Jinzhen and Xiangluo groups (p < 0.05).
As shown in Fig. 3, most nonvolatiles showed a decreasing tendency in the Xiangluo and Jinzhen black teas. Only 11 compounds in the relative concentration had no significant differences between groups, namely, EC, ECG, epiafzelechin gallate, galloyl acid, β-glucogallin, quercetin-3-O-rutinose, kaempferol-3-O-galactoside, kaempferol-3-O-galactosylrutinoside, kaempferol-3-O-glucoside, kaempferol-3-O-rutinoside, and kaempferol-3-O-rutinoside isomer. Among them, kaempferol glycosides were dominant, suggesting that the manufacturing process weakly influenced kaempferol glycosides. Kaempferol-3-O-glucoside and kaempferol-3-O-galactoside are both highly concentrated in young leaves[24], and the former has a high DoT value (3.16‒7.73) in Keemun black tea infusions[15].
The remaining 76 compounds had a significant difference between groups. Simple catechin results by UPLC-QTRAP-MS/MS were consistent with HPLC data (Fig. 2). They were highly concentrated in Keemun Maofeng, except for the non-epi catechins CG and GCG. During the thermal reaction, epi- and non-epi catechins could convert to each other[25]. The epimerization conversion rates of gallated and non-gallated are quite different. For example, the conversion rate of EC to C is about 56.1% at 90 °C, while EGCG to GCG is about 27.4%[25]. As described in our previous study[5], Maofeng black tea has a simple manufacturing process compared with other Keemun black teas, that benefit from the residue of simple or dimeric catechins. Catechin dimers EGC-EGC or EC-EGCG also have a decreased tendency in JZ and XL, suggesting that a long-time thermal procedure harms catechin dimer concentration and enhances the polymerization reaction of catechins, such as the increasing accumulation of theabrownin (Fig. 2). Theacitrins A is a characteristic yellow pigment oxidated from catechins[26], and its isomer concentration also decreased in Keemun Jinzhen and Xiangluo teas.
Organic acids have an important impact on tea taste quality. A previous study mentioned turbidity analysis as a powerful tool for identifying astringent compounds and n-butanol extracts compared to petroleum ether or dichloromethane extracts with the highest turbidity value. With the application of UPLC-Q-TOF-MS, n-butanol extracts consisted of hydroxycinnamoyl quinic acids and flavonoid glycosides, such as 3-O-p-coumaroylquinic acid, 4-O-p-coumaroylquinic acid, and 5-O-p-coumaroylquinic acid[15]. Many p-hydroxycinnamoyl quinic acids in roasted coffee have been identified as strong astringent compounds[27]. Maofeng and Congou black teas contained more hydroxycinnamoyl quinic acids than Jinzhen and Xiangluo black teas (Fig. 3). According to sensory evaluation, the astringent intensity of MF and CG black tea infusions was not stronger than that of JZ and XL. Still, the thickness of the tea infusions was stronger in MF and CG than in JZ and XL. With an astringent character, these compounds might give a complex sense to the human tongue.
Flavonoids (e.g., flavanol, flavonol, flavanone, and flavanonol) and their glycosides are important classes of taste compounds in food[28]. The mono-, di-, tri-, and tetra-glycoside forms of flavonoids have been comprehensively identified in plant tissues, and flavonoid glycosides are mono- or di-glycoside types[29]. The astringent taste is often classified into two types, namely, puckering/rough astringency and velvety/silky astringency. The former provides a negative sensation to tea assessors, while the latter positively impacts on human sensation[28]. Simple catechins and procyanidins are the puckering astringent type, whereas flavonol glycosides are the velvety astringent type, such as kaempferol 3-O-glucoside, quercetin 3-O-galactoside, and myricetin 3-O-glucoside[28]. Among the flavonoids and their glycosides, 31 compounds showed a significant difference between the four Keemun teas. Most flavonoids and their glycosides had a high concentration in Keemun Maofeng and Congou, except for kaempferol-3-O-di-p-coumaroylhexosides and kaempferol (Supplemental Table S1). The kaempferol was highly concentrated in JZ teas, attributed to the decomposition of the glycoside.
Theaflavins were highly concentrated in Keemun Maofeng and Congou, consistent with the tea pigment results. Theaflavins and glucose in thermal conditions could yield theaflavin-3,3′-digallate-glucose and theaflavin-gallate-di-glucose[16]. Based on mass data from previous studies, the [M‒H]‒ precursor ion was restricted with 1,039.2344 (theaflavin-gallate + 2glucose-2H2O), 1,029.1926 (theaflavin-3,3'-digallate-glucose), and 1,011.1811 (theaflavin-3,3'-digallate-glucose-2H2O). The results showed that 1,039.2344 and 1,029.1926 m/z hardly extracted any peaks from MS data, whereas the extraction of 1,011.1811 m/z reached an exceptionally low response value (Supplemental Fig. S1). The response of 1,011.1811 m/z in JZ teas was not stronger than in MF, CG, and XL, whereas the soluble sugar content in JZ teas was lower than in other teas. Our data suggested that theaflavins-glucose conjugates could be detected in Keemun black teas with a trace content, and the decrease of theaflavins and sugars concentration in JZ and XL teas might be attributed to polymerization of theaflavins and the Maillard reaction. For example, theadibenzotropolone A (C50H38O21), a pigment is oxidated from (−)-epicatechin, (−)-epigallocatechin gallate, and theaflavin 3-gallate[30]. We also searched this compound using its [M‒H]‒ precursor ion (973.1833 m/z). The results showed that the 973.1833 m/z has an extremely low response in MS data (about 7e4), below the set detection value (Supplemental Fig. S2). The composition of theabrownin still needs to be elucidated.
Differential metabolites
-
Partial least squares discriminant (PLS-DA) and hierarchical clustering analysis (HCA) results showed that four Keemun black tea types were classified into two groups, the Maofeng-Congou group and the Jinzhen-Xiangluo group (Fig. 4a & b). Orthogonal partial least squares discriminant analysis (OPLS-DA) was used to identify the differential metabolites between the two groups. Sixteen differential metabolites (VIP value ≥ 1) and 12 compounds with a p-value of Student's t-test ≤ 0.05 were identified between MF and JZ teas (Fig. 4c & d).
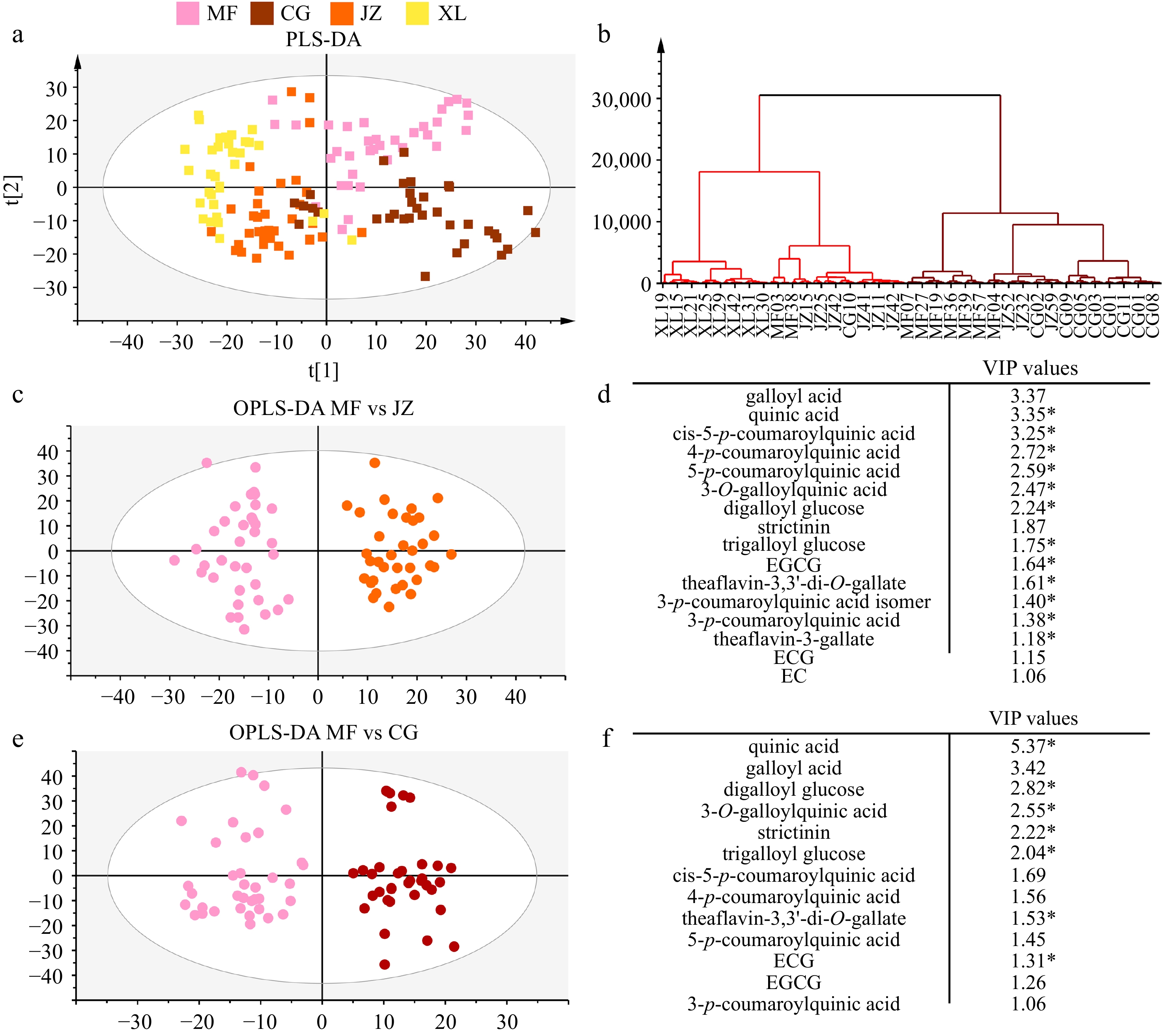
Figure 4.
Identification of differential nonvolatiles between four Keemun black tea types. (a) Principal component analysis (PCA); (b) hierarchical clustering analysis; (c) and (d) orthogonal partial least squares discriminant analysis (OPLS-DA) and differential metabolites between MF and JZ teas; (e) and (f) OPLS-DA analysis and differential metabolites between MF and CG teas.
Among these, p-coumaroylquinic acids were dominant. Coffee roasting experiments showed that 11% of p-coumaroylquinic acids were lost after 210 °C treatment for 20 min, especially 5-p-coumaroylquinic acid[31]. Slight differences in taste flavor between MF-CG and XL-JZ groups are attributed to the shaping stage, which gives a thermal reaction and causes the loss of soluble sugars, p-coumaroylquinic acids, catechins, and theaflavins. Three thermal treatments on Maofeng were performed to elucidate the change mechanism of nonvolatiles.
PCA results showed that the thermal reaction at different temperatures has an important influence on the nonvolatile profile (Fig. 5a). Twenty-six differential metabolites with a VIP value ≥ 1 were identified (Fig. 5b). The p-coumaroylquinic acids increased at 90 °C treatment but decreased significantly at 130−150 °C treatments. Especially 3-p-coumaroylquinic acid, its abundance reduced by 84% at 130 °C for 2 h and was hardly detected after 150 °C treatment. In addition, the epimerization and polymerization of catechins were observed after thermal treatments. The pigment, theadibenzotropolone A, increased as the temperature moved up. A previous study showed that oxidation of EGCG/GCG and ECG/CG yielded gallic acid, de-galloyl flavanols, and corresponding o-quinone derivatives[32].
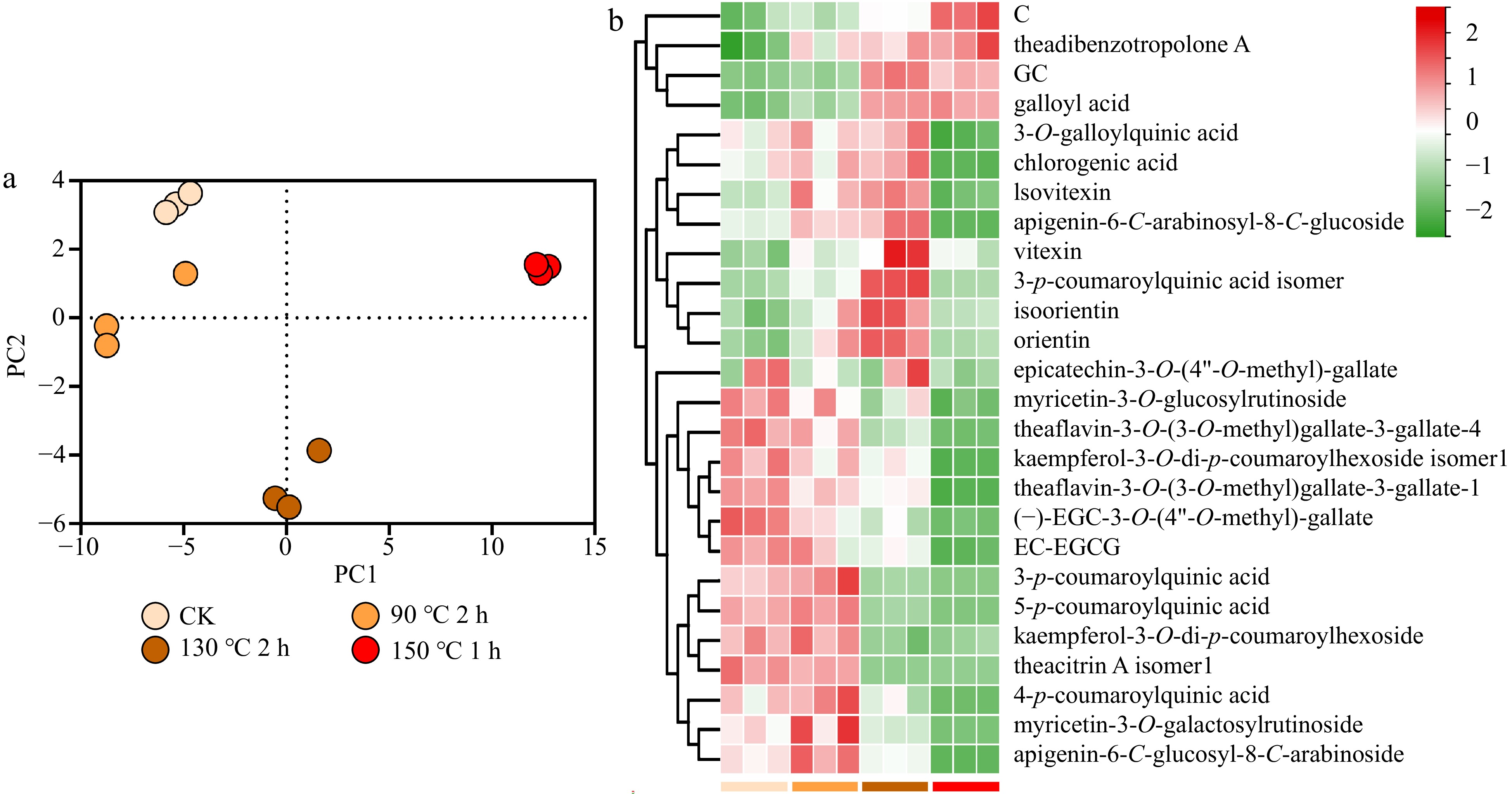
Figure 5.
Thermal treatments of MF black tea samples. (a) PCA; (b) differential nonvolatiles (VIP ≥ 1).
Thirteen differential metabolites (VIP value ≥ 1) and seven compounds with a p-value of Student's t-test ≤ 0.05 were identified between Maofeng and Congou teas (Fig. 4e & f). Most differential compound concentrations were higher in MF teas, except for quinic acid. Quinic acid was significantly negatively correlated with sweet taste[14]. This might explain why the sweetness intensity of Congou was slightly lower than that of Maofeng black tea infusions. As described in a previous study[5], Maofeng teas could be recognized as the primary materials for Congou teas. Maofeng black teas contain complete tender shoots: one bud and one leaf or one bud and two leaves. After the refining and blending stage, Keemun Congou teas contain twisted strip/wiry leaves, shotty/curly/semi-curly Pekoe, bud tips, small slices, and powdered tea. Our previous study separated tea tender shoots into buds, leaves, and stems, and processed them into black teas[33]. Their nonvolatile profiles were analyzed using UPLC-QTRAP-MS/MS in this study.
Metabolite profiles of black teas processed by tea bud, leaf, and stem
-
The metabolite profile in bud, leaf, and stem black teas differed considerably, combined with our previous study[33]. A total of 57% amino acids were concentrated in stem black tea. In contrast, only 14% of catechins were detected (Fig. 6a). Tea pigments, such as TF, TRs, and TB, were also lowly concentrated in stem black tea because of the low concentration of catechins. The total volatile concentration was higher in bud black tea than in stem black tea. Still, benzaldehyde and benzeneacetaldehyde odorants (with a strong honey-like aroma) are highly concentrated in stem black tea[33].
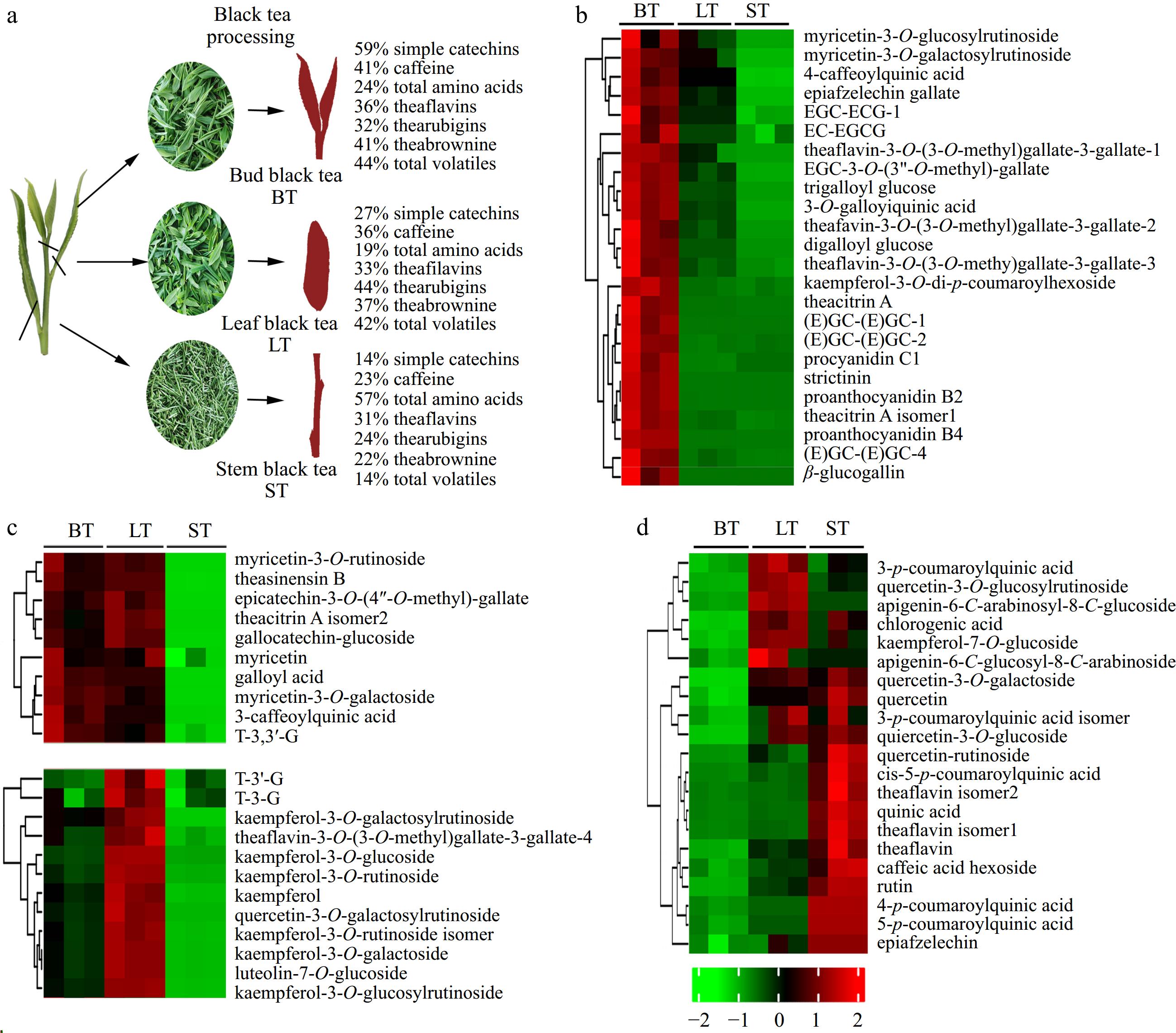
Figure 6.
Metabolite profiles of bud (BT), leaf (LT), and stem (ST) black tea. (a) Concentration proportion of nonvolatile and volatile compounds in BT, LT and ST; (b) nonvolatiles with a high concentration in BT; (c) nonvolatiles with a high concentration in BT and LT; (d) nonvolatiles with a high concentration in LT an ST.
The LC-MS/MS results showed that the majority of nonvolatiles were lowly concentrated in stem black tea (Fig. 6b & c). Catechin dimers, proanthocyanidins, β-glucogallin, and galloyl glucoses were highly concentrated in bud black tea (Fig. 6b). Myricetin, myricetin glycosides, kaempferol, kaempferol glycosides, and galloyl theaflavins were more abundant in bud and leaf black teas (Fig. 6c). In contrast, quercetin, quercetin glycosides, theaflavin, quinic acid, and p-coumaroylquinic acids were more abundant in leaf and stem black teas (Fig. 6d). The accumulation pattern of flavonoids and its glycosides varies significantly with organs of tea plant[34]. Flavonol mono-, di-, and tri-glycoside mostly accumulated in young tea shoots[24]; young leaf had higher concentrations of kaempferol glycosides, while young stem had higher concentrations of quercetin glycosides. A recent study also showed that the nonvolatile profiles of black tea samples made by different tenderness of tea fresh leaves differed, contributing to their distinguished flavor[35].
Supplemental Figure S3 shows a blending principle for traditional Keemun black tea, and this recipe is only prepared for the third grade of Keemun Congou black tea. S1‒S4 refers to the four screening stages; each stage has six sifters of different sizes. M is the last batch of Keemun black tea samples. Hence, according to the scientific recipe, the percentage of twisted strip/wiry leaf, shotty/curly/semi-curly Pekoe, bud tips, small slices and powdered tea is scientifically assigned during the Keemun Congou black tea blending stage. Our previous study showed that Congou black teas contained a highly volatile concentration compared to the other three Keemun black tea types[5]. In the current study, even though Maofeng black tea had a high taste score and abundant nonvolatiles, its aroma was inferior to Keemun Congou black tea. We suggest that the experienced recipe for Keemun Congou gives multiple and coordinated flavors to human sensation.
-
In the current study, four Keemun black tea types were analyzed comprehensively, including taste characteristics, nonvolatile profiles, and differential metabolites. Maofeng black teas without a shaping process reduced the thermal reaction and maintained more catechins, theaflavins, soluble sugars, flavonoids, and glycosides. Jinzhen and Xiangluo, black teas with a shaping stage, contained more polymerized nonvolatile compounds, such as theabrownine and kaempferol-3-O-di-p-coumaroylhexosides. In contrast, soluble sugars, theanine, and amino acids were lowly concentrated in them. The nonvolatile and volatile profiles are differentiated in bud, leaf, and stem black teas. Keemun Congou black teas have complicated refining and blending processes, and the scientific proportion of tea buds, stems, and leaf pieces give a coordinated quality to it. This study indicates that manufacturing processes change the metabolite profile and give customers an alterable flavor and consumption feature.
-
The authors confirm contributions to the paper as follows: study conception and design: Zhou H, Lei P; material preparation and data collection: Xu Y, Wu Q, Yang J; data analysis: Zhou H, Xu Y; draft manuscript preparation and revision: Zhou H, Lei P; partial funds and consultation: Zhou H, Lei P. All authors read and approved the final manuscript.
-
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
This work was supported by the Second Level Youth Development Fund (QNYC-202119) from Anhui Academy of Agricultural Sciences, Anhui Science and Technology Major Program (202003a06020019), and the National Natural Science Foundation of China (Grant Number 32002096). We are grateful to Anhui Keemun Black Tea Development Co., Ltd for providing the flow picture of traditional Keemun black tea processing in Fig. 1.
-
The authors declare that they have no conflict of interest.
- Supplemental Table S1 Identification of 87 nonvolatile compounds in this study.
- Supplemental Fig. S1 The 1011.1811 m/z ion chromatogram extracted from MS data of the four Keemun black tea infusions.
- Supplemental Fig. S2 The ion chromatogram of theadibenzotropolone A (C50H38O21) using its precursor ion (973.1833).
- Supplemental Fig. S3 A blending principle for the third grade of Orthodox Keemun black tea.
- Copyright: © 2024 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Zhou H, Xu Y, Wu Q, Yang J, Lei P. 2024. Identification and comparison of nonvolatile profiles of the four Keemun black tea types. Beverage Plant Research 4: e036 doi: 10.48130/bpr-0024-0025
Identification and comparison of nonvolatile profiles of the four Keemun black tea types
- Received: 13 May 2024
- Revised: 11 June 2024
- Accepted: 21 June 2024
- Published online: 10 October 2024
Abstract: Nonvolatile profiles of four Keemun black tea types (12 samples for each tea type), including Congou (CG), Maofeng (MF), Jinzhen (JZ), and Xiangluo (XL), were comprehensively analyzed using liquid chromatography coupled with a mass spectrometry system (UPLC-QTRAP-MS/MS). MF black teas had the highest taste score, followed by CG. Catechins and amino acids were lowly concentrated in JZ teas, while the theabrownine content was high. The TRs/TFs ratio in CG, MF, JZ, and XL were 10.5, 9.6, 11.6, and 11.1, respectively, all within adequate indexes. Eighty-seven nonvolatiles were identified and quantified, of which the total relative concentration was high in MF and CG. Flavonoid glycosides and hydroxycinnamoyl quinic acids decreased significantly in JZ and XL. Still, their conjugates kaempferol-3-O-di-p-coumaroylhexosides increased in JZ teas. Thermal treatments showed that hydroxycinnamoyl quinic acids decreased significantly as drying temperature moved up and high drying temperature enhanced the epimerization and polymerization of catechins. Furthermore, the metabolite profiles of bud, leaf, and stem black teas differed, and the refining procedure of CG teas balanced these taste and aroma compounds. The present study showed that the difference in manufacturing process changed metabolite profiles of Keemun black tea, and provided customers with an alterable flavor and consumption feature.
-
Key words:
- Keemun black tea /
- Tea processing /
- Nonvolatile profile /
- LC-MS/MS /
- Refining process