-
Oyster mushrooms are the second most cultivated mushroom species in the world, with about 19% of market share[1]. The grey oyster mushroom (Pleurotus ostreatus) is among the most versatile and robust mushroom species, as it can be cultivated on an extensive range of agricultural residues without the need for complete substrate sterilization[2,3]. Even though it is possible to grow oyster mushrooms on wood or straw, many mushroom farmers use substrate ingredients, which could alternatively be used as feed, like cotton seed hulls or bran, which is a commonly recommended substrate supplement[4,5]. While supplementation with nitrogen-rich, edible ingredients can increase mushroom yields[2], the use of substrates which cannot be used as food or feed is more sustainable in the context of food security and agroecology[6]. Also, little is known about the influence of different substrates on carbon emissions during the mushroom cultivation process. Since mushrooms, like animals, are heterotroph organisms, it is important to investigate how their cultivation could contribute to climate change. Another aspect of sustainability in mushroom production is the crop rotation system from which the substrates are sourced, since sustainable crop rotation can reduce the need for fertilizers. Legumes are an important part of crop rotation due to their ability to increase soil nitrogen, and can increase the productivity of cereal crops that are cultivated on the same plot afterwards[7,8]. Cereal crops are rich in carbohydrates and provide a lot of calories, while legumes are rich in proteins and fats, so a combination of the two can provide most of the macronutrients needed for human nourishment[9]. Oyster mushrooms are rich in vitamins and minerals[10] and are a good meat substitute due to their amino acid profile and high protein content[11−13], which makes them a valuable addition to a diet mostly based on cereals and legumes. Mushroom production does not compete with other food production on farmland, as it is nearly 'landless'[6]. However, it can compete with animal husbandry if feed-grade substrates are used. We stipulate that the ideal straw for oyster mushroom cultivation is so nutrient-poor that it provides too little metabolic energy (ME) and digestible protein for feeding a 60 kg goat. A 60 kg goat needs 9.7 MJ of metabolizable energy (ME) and 70 g of digestible protein for its maintenance needs, which it has to draw from a maximum of 1.4 kg feed (dry matter = DM) which it can eat per day[14]. Therefore, 6.93 MJ of ME and 50 g of digestible protein must be available per kg of feed DM. With this baseline, it is guaranteed that the substrate has very little value as feed and for most other agricultural use cases apart from organic matter in the soil. The crops providing the straws for our study were chosen partly due to their high relevance as staple crops and partly to represent typical legumes and cereal crops from temperate climate zones (faba bean and wheat) and from more tropical regions (soy bean and maize). With the experiment we conducted, using these straws to cultivate oyster mushrooms, we want to answer the following research questions:
(1) What is the nutrient composition and feed quality of the different straws?
(2) What is the oyster mushroom production potential of the different straws?
(3) How much carbon and nitrogen are retained in the spent mushroom substrate (SMS) and what are the implications for the need of fertilizer or compost use on the field?
(4) How much carbon is emitted in the process of mushroom cultivation?
-
Straw from two cereal crops and two legumes was used in the experiment: maize (Zea mays, variety Saludo), wheat (Triticum aestivum, variety Faustus), soy bean (Glycine max, a mix of varieties Merlin, GL Melanie, Marquise, Aurelina, ES Favor, RGT Sphinxa, ES Comandor, Amarok, and Arcadia) and faba bean (Vicia faba, variety Tiffany). All straws were produced with certified organic farming practices[15], which ensures that there are no remains of fungicides on the material, which could influence mushroom growth. Maize, faba bean, and wheat were cultivated in 2019 under scientifically controlled conditions at the experimental station of the Thünen-Institute of Organic Farming in northern Germany. To get a nutrient-poor maize straw despite using a feed variety of this crop (sweet maize varieties cannot be cultivated in cold, northern German climate), the maize was left standing in the field for four months after harvest season before cutting it, allowing nutrients to leach back into the soil. Soy was grown at the organic experimental station Gladbacher Hof of the University Giessen in central Germany. The different straws included all the above-ground parts of the plant, except the grain, and the cobs in the case of maize. All the straws were chopped (< 2 cm) and dried for 5 d at 40 °C for storage.
Grain spawns with the mycelium of P. ostreatus ((Jacq.: Fr.) P. Kumm, strain number: P10001, type of grain: wheat) was used to inoculate the straws for mushroom cultivation.
Experimental design
-
The experiment consisted of four treatments (wheat, maize, soy, and faba) with eight replicates. Each replicate consisted of a polypropylene mushroom grow bag with a micropore filter (50 cm × 8 cm × 0.1 cm, Hemoton brand®) filled with 800 g of moist, pasteurized substrate (200 g dry matter and 600 g water) and 20 g of mushroom grain spawn (6.6 g dry matter). Hot air pasteurization at 100 °C for 3 h was used to pasteurize the substrate. Grain spawn was added to the substrate after letting it cool down, ensuring that the spawn was well distributed by twisting and shaking the bags. The mushrooms were cultivated under controlled conditions in the laboratories of the Thünen-Institute, at 21 °C and 90% humidity in a grow-box (HOMEbox Vista Medium), as depicted in Fig. 1.

Figure 1.
Different substrates are used for mushroom cultivation in a random replication approach in a grow chamber.
Since it was intended to use the same amount of dry matter and water in the different treatments, but the straws had different water-holding capacities, the replicates were hung in a way that allowed excess water to drip from a small opening at the bottom.
Data collection
-
Up to three flushes were harvested from each replicate. The fresh weight of the harvested mushrooms was determined immediately after harvest. The dry yield was determined after drying at 105 °C for 24 h. From this data, the biological efficiency (BE, percentage of dry matter of substrate converted to fresh matter of mushrooms[2] and the biomass conversion rate (BCR, percentage of dry matter of substrate converted to dry matter of mushroom) were calculated. The visually discernible occurrence of bacteria or molds in each replicate was checked and the mycelial growth (from 0% of substrate colonized to 100%) was estimated weekly during the first three weeks of the experiment. After the cultivation period, the substrate was removed from the bags, crushed, and dried at 105 °C for 24 h, to determine the dry weight of each replicate and to take samples for chemical analyses. All raw data on mushroom yield and the composition of SMS is made available in the Supplemental Table S1 to be published with this study.
Chemical analyses, estimations of protein content and carbon emissions
-
The nitrogen (N) and carbon (C) content of the spawn, straw, SMS and mushrooms were analyzed with the DUMAS-method[16]. The protein content (XP) of the straw and mushroom was estimated by multiplying the nitrogen content with the factor 6.25, which is commonly used in the analyses of feed[17]. Carbon emissions through respiration of the fungal mycelium are estimated by subtracting the amount of carbon found in the SMS and the mushrooms from the amount present in the straw before cultivation. The crude fiber (XF) of the straw was determined with the Weender-van Soest analysis[18]. The number of analyses were focused on SMS. While only one collective sample from the straw, spawn and mushrooms were taken, a separate sample from the SMS of each replicate was analyzed. To assess whether the type of straw influences the protein composition of the harvested mushrooms, three more samples from mushrooms cultivated on the same straw under the same conditions were analyzed a year later and are included in the results of this study. All analyses were carried out in the laboratory of the Thünen-Institute of Organic Farming.
Estimations of feed quality
-
To calculate the metabolizable energy (ME) of the different straws for ruminants, the following formula was used[19]:
ME (MJ) = 0.0312 * digestible fat (g) + 0.0136 * digestible fiber (g) + 0.0147 * (digestible organic matter (g) – digestible fat (g) – digestible fiber (g)) + 0.00234 * raw protein (g)
The data on the digestibility of the different fractions of the maize and wheat straw were taken from the publications of the German association for agriculture[19], while the data on faba bean straw was taken from the Dutch central feedstuff databank[20]. For soy bean straw, only incomplete data could be found. Information for the digestibility of organic matter and of protein of soy straw was found on the Feedipedia database[21]. To fill in the missing values on the digestibility of lipids and fibers in soy straw, we used the data on straw from a similar legume, namely the pea, Pisum sativum, from the DLG[19]. The compiled digestibility data can be found in Table 1.
Table 1. Digestibility of the different macronutrient fraction in the different straw types: Digestible organic matter (DOM), digestible protein (DP), digestible lipids (DL) and digestible fiber (DF).
Straw type DOM (%) DP (%) DL (%) DF (%) Wheat 47 20 49 53 Faba bean 52 46 53 42 Maize 72 50 64 68 Soy bean 52 54 55 42 Statistical analyses
-
For statistical analysis, Microsoft Excel and the freeware R-studio (version 4.0.3) were used. One-way analysis of variance (ANOVA) and the posthoc Tukey's test were used to compare different treatments and show significant differences. To test for the assumptions of normal distribution of the data and the residuals, histograms, and qq-plots were assessed and the Shapiro-Wilk test was used.
-
The chemical analysis (Table 2) showed large differences between the types of straw with regard to the nitrogen content, while the carbon content was almost the same. The wheat straw contained the least nitrogen and faba bean contained the most. The carbon/nitrogen-ratio (C/N-ratio) of the straws varied from 45 to 130. The content of crude fiber content (XF) was lowest in maize straw followed by soy bean, wheat, and faba bean. The mushroom spawn had a notably higher nitrogen content than the straws with a C/N-ratio of 15.6.
Table 2. Chemical composition of the dry matter (DM) of different straws and mushroom spawn used for oyster mushroom cultivation.
Sample C (% DM) N (% DM) C/N-ratio XF (% DM) Wheat straw 47.12 0.36 130.37 47.97 Faba bean straw 47.12 1.05 44.75 49.86 Maize straw 47.07 0.68 69.72 38.03 Soy bean straw 47.18 0.59 79.91 43.86 Mushroom spawn 46.33 2.96 15.63 x In combination with the data in Table 1, the digestible protein and metabolizable energy were calculated. As Table 3 shows, wheat straw had the least digestible protein, while faba bean had the most. Maize straw, according to these calculations, had the most metabolizable energy.
Table 3. Protein, digestible protein (DP) and metabolic energy (ME) of the different straws for 1 kg dry matter and 1.4 kg dry matter (estimated daily feed intake of a goat).
Sample Protein DP ME g/kg
DMg/kg
DMg/1.4 kg
DMMJ/kg
DMMJ/1.4 kg
DMWheat straw 19.4 3.9 5.4 6.3 8.8 Faba bean straw 63.0 29 40.5 7 9.8 Maize straw 38.9 19.5 27.2 9.8 13.7 Soy bean straw 33.7 18.2 27.4 7.2 10.1 Mushroom production
-
In the 56 days of the experiments, most replicates produced two mushroom harvests, with a few replicates producing only one or even three harvests. No clear pattern was discernible between treatments in terms of number of harvests. The occurrence of green mold was limited to one replicate of the wheat straw treatment and two replicates of the maize straw treatment. Since these replicates failed to produce mushrooms, they were taken out of the experiment, reducing the number of replicates in wheat straw to seven and in maize straw to six. Mycelial growth was the fastest in the soy bean straw treatment, where all replicates were fully colonized after 13 d, while the replicates in other treatments were fully colonized after 21 d.
Wheat straw produced significantly lower yields than all other treatments, both in terms of fresh matter (BE) and dry matter (BCR). Maize straw produced significantly more mushrooms than all other treatments in terms of fresh yield but not significantly more than soy straw in terms of dry yield (Table 4).
Table 4. Fresh and dry yield.
Treatment Fresh matter BE (%) Dry matter BCR (%) Wheat straw 58b (12.5) 3.8c (0.8) Faba bean straw 76bc (17.3) 6.6b (1.4) Maize straw 114a (10.2) 9.2a (0.9) Soy bean straw 89.1b (14.7) 8.6a (1.3) Biological efficiency (BE) and biomass conversion rate (BCR) of the different treatments. Significant differences between treatments are marked by letters above the data. Standard deviation given in brackets behind the mean. While wheat, faba bean, and soy bean straw produced more than 75% of the total dry yield in the first harvest, maize straw on average produced more than 50% of the mushrooms in the second and third harvests (Fig. 2).
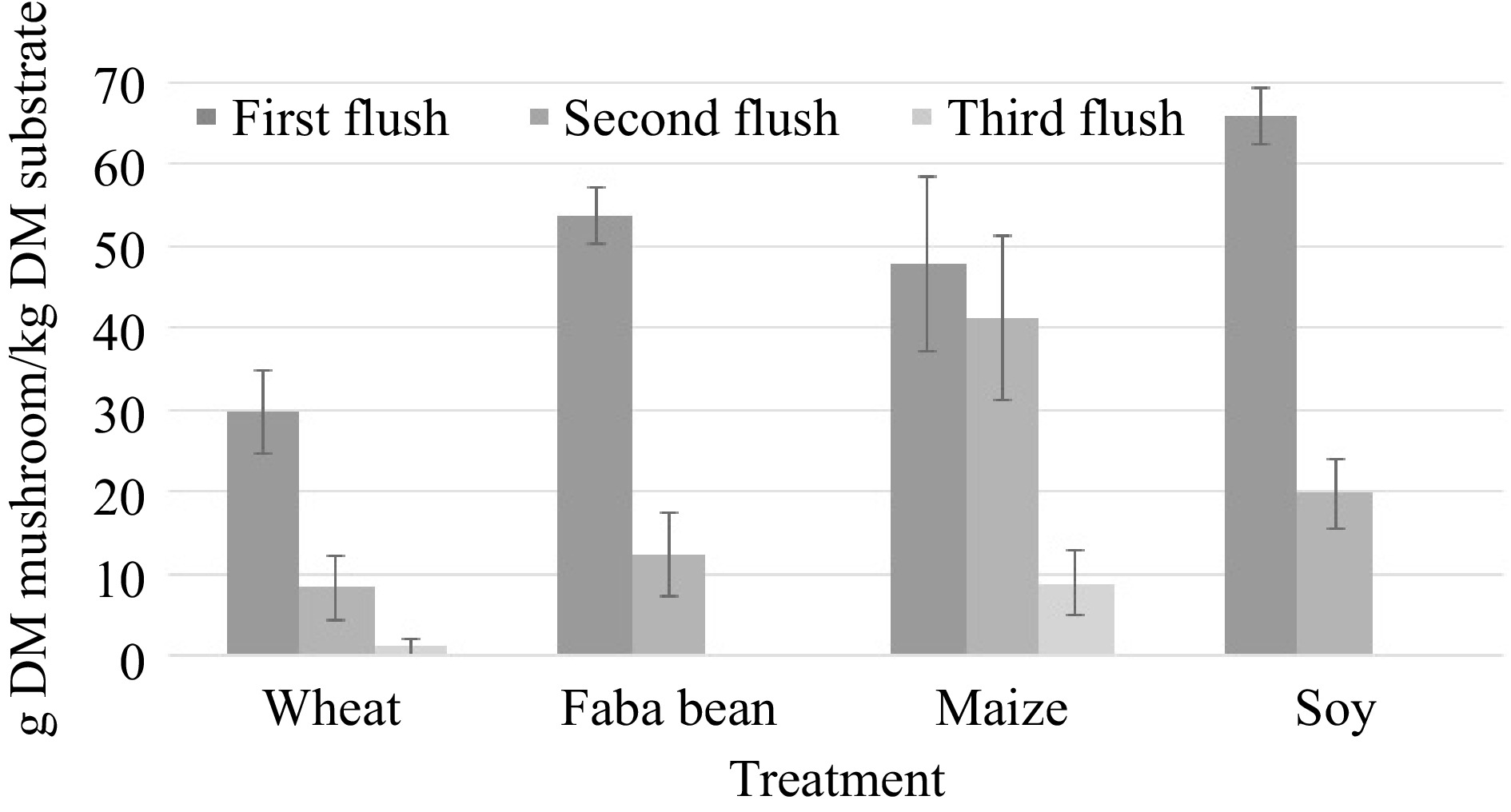
Figure 2.
Average dry yield per dry substrate distributed over different harvest flushes in the different treatments. Error bars show standard deviation.
The composition of mushrooms from the different treatments is presented in Table 5. Mushrooms cultivated on faba bean straw contained significantly more nitrogen and thus protein than mushrooms from the other treatment.
Table 5. Chemical composition of the mushrooms from different treatments.
Treatment C (%) N (%) C/N Protein (%) Wheat straw 44.7a (1.1) 2.7b (0.3) 16.8b 16.8 (1.6) Faba bean straw 45.4a (1.2) 3.7a (0.2) 12.2a 23.2 (1.3) Maize straw 45.1a (0.8) 3b (0.2) 14.9b 19 (1.4) Soy bean straw 45.6a (0.6) 2.8b (0.2) 16.2b 17.7 (1.6) Standard deviation given in brackets behind the mean. Significant differences between treatments are marked by letters above the data. By synthesizing the yield data and the chemical analysis, one can estimate the amount of protein produced per kg of straw. On wheat straw, 6.4 g protein were produced per kg of straw, on soy bean straw 15.2 g, on faba bean straw 15.4 g and on maize straw 17.4 g.
Nitrogen and carbon flow
-
The change in nitrogen and carbon content of the straws after cultivation differed notably between the different treatments. While the C/N ratios of wheat, maize, and soy bean straw were decreased by mushroom cultivation, it slightly increased on faba bean straw (Table 6).
Table 6. Chemical composition of the spent mushroom substrate (SMS).
SMS type C (% DM) N (% DM) C/N C/N ratio change Wheat straw 45.5 (0.9) 0.4 (0) 106.0 −24.4 Faba bean straw 45.6 (0.4) 1 (0) 47.3 2.6 Maize straw 43.8 (0.4) 0.7 (0) 62.7 −7.0 Soy bean straw 43.9 (0.4) 0.6 (0) 71.7 −8.3 Standard deviation of carbon and nitrogen content given in brackets after the mean. C/N change is the difference in the C/N ratio in comparison to the ratio of the straw before mushroom cultivation. The amount of dry matter, carbon, and nitrogen from the straw that remained in the SMS after cultivation is presented in Table 7. Dry matter, carbon and nitrogen reduction was notably lower in wheat straw than in the other treatments. The strongest reduction in all these metrics occurred in the treatment with maize straw. In terms of emissions per unit of mushrooms, wheat straw emitted the most carbon, with 3.5 kg per 1 kg of dry mushrooms, while soy straw emitted the least, with 2.6 kg.
Table 7. Mass transfer from straw to spent mushroom substrate in the different treatments.
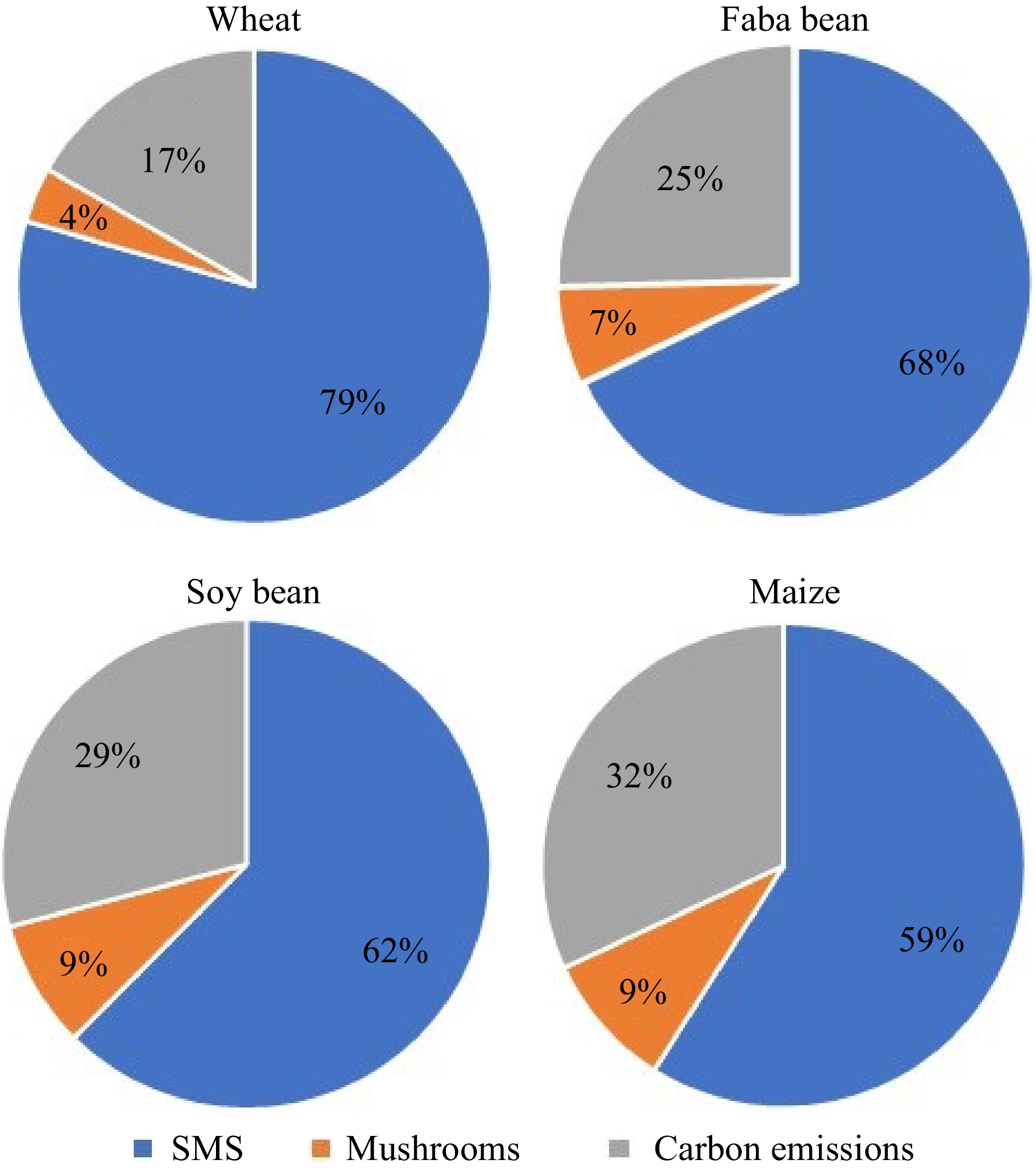
Treatment Dry matter (%) C (%) N (%) Wheat straw 82.2 (6.4) 79.5 (5.6) 79.3 (12.3) Faba bean straw 70.1 (5) 67.9 (4.6) 60.5 (3.9) Maize straw 63.4 (1.9) 59 (1.7) 59 (3.9) Soy bean straw 67.1 (1.9) 62.4 (1.6) 61.5 (4) Standard deviation given in brackets behind the mean. By subtracting the amount of dry matter in the SMS (Table 7) and in the mushrooms (Table 5) from the amount of dry matter in each replicate at the beginning of the experiment (200 g straw + 6.6 g spawn), the unaccounted rest was calculated. This rest is the amount of carbon that was lost to respiration by the mushroom. As can be seen in Fig. 3, in the wheat treatment, 17% of the dry matter are estimated to be carbon emissions, 25% in the faba bean treatment, 32% in the maize treatment and 29% in the soy treatment.
-
None of the straws used for mushroom cultivation in this study can be considered as good feed for goats or other ruminants, since none would provide both enough energy and protein for metabolic basic needs. Therefore, the use for mushroom production as supplementary food for humans is a sustainable choice, as long as the SMS is brought back to the soil for organic matter.
Mushroom production potential
-
The best-performing straws in the experiment were maize and soy straw, which were almost evenly matched in terms of dry yield. There were however interesting differences between these two treatments, which could be relevant to mushroom producers. On one hand, maize straw produced more fresh yield than soy straw, which may be related to differences in crude fiber contents and the physical structure of the two materials. While soy straw is coarse and hard, maize straw is very soft and sponge-like and could thus better provide the mycelium with water and air, as for example, Stamets discusses[2]. Interestingly, soy produced more mushrooms than maize in the first flush, indicating that the nutrients were more accessible to the mushroom. Unlike the other substrates maize straw produced more than half of its total dry yield in the second and third harvests. From an economic and space-use efficiency standpoint, it could thus be beneficial for mushroom producers to harvest only once from all straws except maize. Maize yields were high in comparison to other studies[22−24]. Soy bean straw produced comparable results to another study, which used different species of oyster mushrooms[25]. No studies were found utilizing faba bean straw. The results in this study concerning yield from wheat straw, were in the range of other studies, with some having higher yields[26] and some lower[27]. The low nitrogen content of the wheat straw used in this study surely contributed to the low productivity.
If we take the view of Stamets that an oyster mushroom cultivator should operate at around 100% BE or more, to be economical[2], the faba bean and wheat straw can be said to be un-economical if used as pure substrates. However, the validity of this view may differ in different markets and depending on the profit made through the use of SMS. Looking at the nutrient composition of the different straws, it is notable that faba bean straw produced relatively few mushrooms despite its high nitrogen content. This is supported by the fact that the C/N ratio increased in faba bean straw as a result of mushroom cultivation, indicating that there was more nitrogen available than necessary for the mushroom. The significantly increased protein content of mushrooms produced on faba bean straw in comparison to the other treatments is also most likely a result of this effect. This substrate may be more efficiently utilized in oyster mushrooms production, if mixed with a more carbon rich material.
Mass, nitrogen, and carbon flow
-
The removal of dry biomass from the different straws during the cultivation process, displayed in Fig. 3, was in the range of other studies[26]. However, the distributions that were found are roughly equivalent to that presented by Stamets[2], who writes that around 10% of the biomass of a substrate is turned into mushrooms and 70% into SMS.
Significant amounts of nitrogen were removed from the straw by the mushrooms in all treatments, with the lowest removal found in wheat straw and the highest in maize straw. While the value of the SMS as fertilizer is higher than that of straw due to the decreased C/N ratio, this nevertheless means that more nitrogen would be removed from the field than if the straw was used as mulch. Therefore, the removal of straw from fields for mushroom cultivation might make it necessary for farmers to return nitrogen through application of fertilizers. It would also be possible to mix SMS with other materials like dung and compost them, to solve this problem.
Carbon removal from the straw by the respiration of oyster mushroom mycelium was also significant and roughly in the same range as nitrogen. Large differences were found between the treatments in terms of the carbon that was emitted during mushroom cultivation. Wheat straw emitted the least carbon. However, in terms of emissions per unit of mushrooms, wheat straw emitted the most carbon, while soy straw emitted least. In terms of greenhouse gas emissions, mushroom production on legume straw was more sustainable than on cereal straw. However, to get a complete picture, a full live cycle assessment has to be conducted and the emissions caused by pasteurizing the substrate and during the cultivation phase also have to be accounted for. Nevertheless, the carbon emissions associated with mushroom cultivation will most likely be quite low compared to other protein sources such as goat meat or other animal products[28]. Further research into this topic should be conducted.
-
Using nutrient-poor straw from cereals and legumes for oyster mushroom cultivation is an efficient and sustainable way of producing protein-rich food in very little space. Even substrates that have almost no value as feed can be used in this way, which can increase the economic return of cropland, if unused straw will be used for mushroom production. Maize stover was found to be the best straw, followed by soy straw, which also produced high yields. While wheat straw produced inferior yield, faba bean straw had mediocre yields, but produced mushrooms with a higher protein content. Since significant amounts of carbon and nitrogen are removed from the straw through mushroom production, it is necessary to increase fertilizer or compost application to the field where straw was removed, even if the SMS is returned as an organic amendment.
-
The authors confirm contribution to the paper as follows: study conception and design: Grimm D, Sonntag E, Rahmann G; data collection: Grimm D; analysis and interpretation of results: Grimm D, Rahmann G; draft manuscript preparation: Grimm D. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
-
The authors declare that they have no conflict of interest.
- Supplemental Table S1 Basic data from mushroom cultivation experiment on four different straws.
- Copyright: © 2024 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Grimm D, Sonntag E, Rahmann G. 2024. Oyster mushroom cultivation on cereal and legume straw of poor feed quality. Studies in Fungi 9: e010 doi: 10.48130/sif-0024-0010
Oyster mushroom cultivation on cereal and legume straw of poor feed quality
- Received: 31 May 2024
- Revised: 05 July 2024
- Accepted: 09 July 2024
- Published online: 12 August 2024
Abstract: This study explores the viability of cultivating oyster mushrooms on cereal and legume straw of poor feed quality, investigating oyster mushroom productivity, and the implications for mass-, nitrogen- and carbon flows within the agricultural system. Four types of straw (wheat, maize, faba bean, and soy bean) were utilized as substrates for mushroom cultivation. Fresh yields varied widely, from 114% biological efficiency on maize straw to 58% on wheat straw, while dry yields ranged from 9.2% biomass conversion rate on maize straw to 3.8% on wheat straw. The protein content of mushrooms varied between 16.8% on wheat straw and 23.2% on faba bean straw, correlating with the nitrogen content of the straw. Furthermore, results revealed significant variations in carbon emissions, ranging from an estimated 3.5 kg (on wheat straw) to 2.6 kg (on soy straw) emitted per kg of dry mushroom produced. These findings underscore the importance of substrate selection in mushroom cultivation, with implications for agricultural resource management and food production. Depending on the focus, different substrates may be considered as optimal. While maize straw produced most mushrooms in this study, soy bean straw emitted the least carbon in relation to yield, faba bean straw produced mushrooms with higher protein content, and wheat straw retained the most nitrogen in the spent mushroom substrate.
-
Key words:
- Mushrooms /
- Straws /
- Cultivation /
- Cereals /
- Legumes /
- Circular Economy