-
Rice (Oryza sativa L.) is one of the most important food crops playing a crucial role in addressing human food challenges worldwide. It is cultivated in more than 140 countries across the globe[1]. China ranks as the second largest country in terms of rice planting area and has the largest production globally, with over 60% of its residents relying on rice as their staple food[2,3]. With the advancement of industrialization and intensive agriculture, since the 1980s, a significant quantity of fertilizers and pesticides have been extensively to guarantee high yields[4]. As a farmland ecosystem characterized by significant water usage, fertilizer application, and pesticide use, most paddy fields exhibit varying degrees of soil compaction, acidification, and heavy metal pollution as a result of years of cultivation, prolonged flooding, and excessive application of chemical fertilizers and pesticides. The phenomenon has affected the soil micro-ecosystem, resulting in the degradation of soil texture, accelerated the decline of soil fertility in paddy fields, and contributed to pollution from excessive levels of N, P, and pesticide residues due to the overuse of chemical fertilizers and pesticides in the planting area. These factors pose hidden risks to the sustainable and safe production of rice and environmental safety. As a farmland ecosystem utilizes large quantities of water, fertilizer and pesticides, most of the paddy fields exhibit varing degrees of soil compaction, acidification, and heavy metal pollution due to years of cultivation, prolonged flooding, and imbalance application of chemical fertilizers and pesticides. Under the premise of ensuring food security, agriculture is evolving into a model characterized by low input, high yield, low pollution, and high quality[5]. It is imperative to explore methods for ensuring the supply of nutrients required for rice growth and yield while reducing or eliminating the input of fertilizers.
Agricultural Jiaosu is produced by microbial fermentation using plant-based organic waste as raw material, with or without additives. It contains nutrients, specific microbial active ingredients, and beneficial microbial communities making it suitable for use in agriculture, animal husbandry, and soil improvement[6,7]. According to different preparation processes, agricultural Jiaosu fertilizers can be categorized into various types such as soil regulators and liquid level regulators. Agricultural Jiaosu contains organic acids, mineral nutrients, plant hormones, enzymes, and other components. There components can reduce the content of heavy metals in agricultural products, increase the content of soil organic matter and soil aeration[8], regulate soil acidity and alkalinity ensuring optimal conditions for crop growth[9,10] . Concurrently, Jiaosu used in agriculture enhances the utilization of resources from organic waste. Their deployment presents a feasible approach for realizing circular agriculture objectives. The adoption of circular agriculture practices lead to improved economic gains in agriculture, environmental conservation, and the assurance of a secure and stable food supply system[11].
Although studies have shown that agricultural Jiaosu can promote rice seedling emergence, improve the quality of seedlings and prevent overgrowth[12], increase yield, improve rice quality and soil properties[12,13]. The application of Jiaosu is mostly in the form of Jiaosu microbial fertilizer combined with organic fertilizer or chemical fertilizer, However, the effect of plant-derived agricultural Jiaosu on rice growth and production has not been explored. Therefore, in this study, conventional fertilizer treatment is used as the control, and different combinations of soil liquid agricultural Jiaosu, soil solid agricultural Jiaosu, and leaf liquid agricultural Jiaosu are employed to explore their effects on rice growth dynamics, yield, and soil available nitrogen, phosphorus, and potassium. This research will provide a theoretical and practical basis for the application of different plant-derived agricultural Jiaosu combinations in rice growth and production.
-
Yichun City, an important rice producing area of Jiangxi Province of China features a humid subtropical monsoon climate with four distinct seasons, mild climate, abundant rainfall, and sufficient sunshine. The annual average temperature ranges from 16.2 to 17.7 °C with approximately 1,740 h of sunshine. The effective accumulated temperature ranges from 5,050 to 5,644 °C, and the average annual precipitation ranges from 1,555.8 to 1,740.2 mm[14]. The pot experiment was carried out in Binjiang Town, Yuanzhou District (27.78° N, 114.54° E), which is a typical paddy field area with favorable natural conditions for rice growth. The soil type in this area is gleyed paddy soil, developed from red soil.
Experimental design
-
To obtain typical soil for pot experiments, five 2 m × 2 m plots were selected randomly from paddy fields. Soil samples were collected from the 0-20 cm depth of each plot. Large plant residues, gravel, and debris were removed from the collected sample. The soil samples from all five plots were thoroughly mixed and then placed into the pots for the experiments. To ensure consistent root growth with an equal amount of soil in each pot, the soil each the pot is measured to a depth of 30 cm. In the pot experiment, there were four treatments: conventional fertilizer (F); soil solid agricultural Jiaosu + soil liquid agricultural Jiaosu + leaf liquid agricultural Jiaosu (JA); soil solid agricultural Jiaosu + soil liquid agricultural Jiaosu (JT) and soil solid agricultural Jiaosu + leaf liquid agricultural Jiaosu (JY). These treatments were replicated four times.
The tested agricultural Jiaosu include soil solid agricultural Jiaosu, soil liquid agricultural Jiaosu, and leaf liquid agricultural Jiaosu. Three types of Jiaosu (Shanxi agricultural fertilizer (2018) No. 2150) are all produced by Weinan Shuntian agricultural Jiaosu Technology Company. The conventional fertilizer was rice-specific fertilizer (Gan agricultural fertilizer (2018) No. 0019) produced by Jiangxi Woerde New Fertilizer Technology Company. The basic information on the fertilizer and agricultural Jiaosu is shown in Table 1. Late rice (Chunliangyou 121) was planted using the root bag method.
Table 1. Basic information on different treatments and fertilizers.
Variety pH Soil organic N P2O5 K2O Producer Chemical fertilizer − − 15% 15% 15% Jiangxi Woerde New Fertilizer Technology Co., LTD Solid agricultural Jiaosu for soil 5.50 93.09% 3.31% 1.57% 1.74% Weinan Shuntian Agricultural JiaoSu Technology Co., LTD Liquid agricultural Jiaosu for soil 4.46
(1:250 dilution)31.60 g·L−1 1.62 g·L−1 3.44 g·L−1 8.31 g·L−1 Liquid agricultural Jiaosu for leaf surface 3.96
(1:250 dilution)39.04 g·L−1 − − − F − − 18.00 g·pot−1 18.00 g·pot−1 18.00 g·pot−1 − JA − 140.01 g·pot−1 4.98 g·pot−1 2.40 g·pot−1 2.73 g·pot−1 − JT − 139.97 g·pot−1 4.98 g·pot−1 2.40 g·pot−1 2.73 g·pot−1 − JY − 139.51 g·pot−1 4.96 g·pot−1 2.35 g·pot−1 2.61 g·pot−1 − F is treatment of conventional fertilizer, JA treatment is solid agricultural Jiaosu for soil + liquid agricultural Jiaosu for soil + liquid agricultural Jiaosu for leaf surface; JT is solid agricultural Jiaosu for soil + liquid agricultural Jiaosu for soil; JY is solid agricultural Jiaosu for soil + liquid agricultural Jiaosu for leaf surface. Based on the amount of fertilizer applied in the field (600 kg·ha−1 base fertilizer, 300 kg·ha−1 topdressing) and the experience of using agricultural Jiaosu in the field (solid agricultural Jiaosu: 1800 kg·ha−1, soil liquid agricultural Jiaosu: 150 L·ha−1, leaf liquid agricultural Jiaosu: 15 L·ha−1), the amount of materials used for different treatments is calculated according to the potted plant area (Table 1). Fertilization was carried out as follows: 15 mL of soil liquid agricultural Jiaosu was added to each replicate of JA and JT treatments. Water was then added to cover the soil surface to a depth of 3 cm of the soil surface, stirred evenly, and kept flooded for 3 d. The pots treated with JY and F were added with water to 3 cm on the soil surface, stirred evenly, and flooded for 3 d. After 3 d of drainage, 120 g of rice special fertilizer was added to each pot of F treatment as base fertilizer, and 150 g soil solid agricultural Jiaosu was added to each pot of JA, JT, and JY. On the 16th day of transplanting, the pots treated with JA and JY were sprayed with 1 mL of leaf liquid agricultural Jiaosu of each pot, and the dilution ratio was 1:1000. At the same time, F and JT were sprayed synchronously with 1,000 ml water. To avoid the impact of seedlings variability on the treatment, rice seedlings (Chunliangyou 121) with consistent plant height and growth conditions were selected for transplanting. Each root bag was filled with 200 g soil for single plant transplanting. The root bags were planted according to a standard row spacing of 10 cm (plant spacing) and 15 cm (row spacing).
Sample collection
-
The aboveground parts and roots of the rice plants were collected on the 16th, 27th, 48th, and 100th d (harvest period) after rice transplantation, respectively. All plant samples had their enzymes deactivated at 105 °C for 15 min and dried at 40 °C until constant weight to measure the biomass. While monitoring plant height and aboveground biomass, the soil inside the root bag is collected for measuring soil ammonia nitrogen, available phosphorus, and available potassium.
Rice monitoring indicators and methods
-
On the 16th, 27th, 48th, and 100th (harvest period) day after rice transplantation, one plant (avoiding significantly abnormal rice growth) was randomly selected from each replicate of each treatment for measuring rice plant height (the distance from the ground to the highest point of the rice plant).
During the harvest period, the effective panicle, the plant height of each normally growing rice plant, and the panicle length of each plant were measured. After threshing, the yield of each plant was measured, and the yield was expressed as drying quality. The specific measurement method was as follows:
(1) 1000-grain weight: The rice grains in each pot were mixed respectively, part of the rice was then taken out using the quartering method, and 1,000 grains were counted, dried and weighed.
(2) Seed setting rate: The empty and filled grains of each rice plant were counted and calculated respectively.
$ R=\dfrac{A}{T}\times 100 $ (1) Where, R: rice seed setting rate, %; A: actual number of grains per plant; T: total number of grains per plant.
(3) Effective panicles: the number of rice grains per plant is greater than 5 tillers.
(4) Panicle length: The rice panicles of each rice plant were measured by a ruler to measure the length of each rice panicle from the panicle stem node to the top of the panicle, accurate to 0.1 mm.
Soil monitoring indicator analysis
-
The soil in the root bag was taken out, the roots were removed, and all were passed through a 2 mm sieve. One part of the soil was used to determine the soil ammonia nitrogen, and the other part of the soil was dried to determine the soil's available phosphorus and potassium. Determination of soil ammonia nitrogen: 5.00 g fresh soil was put into the extraction bottle and 25 mL 2 mol·L−1 potassium chloride solution was added. After shaking for 1 h, the solution was filtered and determined by a continuous flow analyzer. Determination of available phosphorus in soil: 5.00 g of air-dried soil with 2 mm sieve was weighed in the extraction bottle, and 50 mL of extractant containing 0.03 mol·L−1 ammonium fluoride and 0.025 mol·L−1 hydrochloric acid was added. After 5 min of oscillation, it was filtered and determined by continuous flow analyzer. Determination of soil available potassium: 5.00 g air-dried soil with a 2 mm sieve was weighed in the extraction bottle, and 1 mol·L−1 ammonium acetate solution (pH = 7.0) was added. After 30 min of oscillation, the soil was filtered and determined by an inductively coupled plasma emission spectrometer.
Data analysis
-
$ HR=\dfrac{{H}_{2}-{H}_{1}}{{T}_{2}-{T}_{1}} $ (2) Where, HR: height growth rate, cm·d−1; H1: starting plant height, cm; H2: ending plant height, cm; T1: starting day, day; T2: ending day, day.
$ BR=\dfrac{{B}_{2}-{B}_{1}}{{T}_{2}-{T}_{1}} $ (3) Where, BR: biomass growth rate, g·d−1; B2: starting biomass growth, g; B1: ending biomass growth, g; T2: starting day, day; T1: ending day, day.
Calculate the proportion of changes in yield indicators for different treatments.
$ V=\dfrac{T-F}{F}\times 100 $ (4) Where, V: Variation rate, %; T: treatment; F: conventional fertilizer.
Generally, increasing plant height also increases biomass. To evaluate the sensitivity of biomass increase to plant height, this study will calculate the multiple of biomass increase for every 10cm increase in plant height based on the exponential correlation between plant height and biomass (Q10).
$ B=a\cdot {e}^{b\times H} $ (5) $ {Q}_{10}={e}^{10\times b} $ (6) where, B: aboveground plant biomass; H: plant height.
All data were analyzed for variance using SPSS 25.0. Multiple comparisons were conducted using Duncan's method for significance testing of differences (p = 0.05), all data were expressed as means ± standard errors (SE). The influencing factors of rice yield were based on the path analysis of AMOS 21.0, and plots were generated using SigmaPlot 14.0.
-
After 27 d of transplanting, the plant height of rice treated with the three Jiaosu treatments was similar to that of rice treated with conventional fertilizers. After 48 d of transplanting, the plant height of rice treated with fertilizers was higher than that of the JY treatment (p < 0.05). During the harvest period, the rice plant height of the three Jiaosu treatments was higher than that of the conventional fertilizer treatment. (Fig. 1a). The plant height of JT was significantly higher than that of the fertilizer treatment (p < 0.05) and the plant height of JT, JY, and JA increased by 7.01%, 4.18%, and 0.24% respectively compared to the conventional fertilizer treatment. After 16 d, the growth rate of plant height in each treatment was equivalent. From 16 to 27 d after transplantation, the growth rate of plant height in the three Jiaosu treatments was higher than that of fertilizer, with JT treatments significantly higher than the fertilizer treatment. However, from the 27th to the 48th day of transplanting, the plant height growth rate of fertilizer treatment was the highest (1.5 ± 0.18 cm·d−1), while JT had the lowest, only 1.1 ± 0.16 cm·d−1. From the 48th to the 100th day of transplanting (harvest period), the plant height growth rate of fertilizer treatment was the lowest, significantly lower than the JT and JY, and slightly lower than the JA treatment (Fig. 1b).
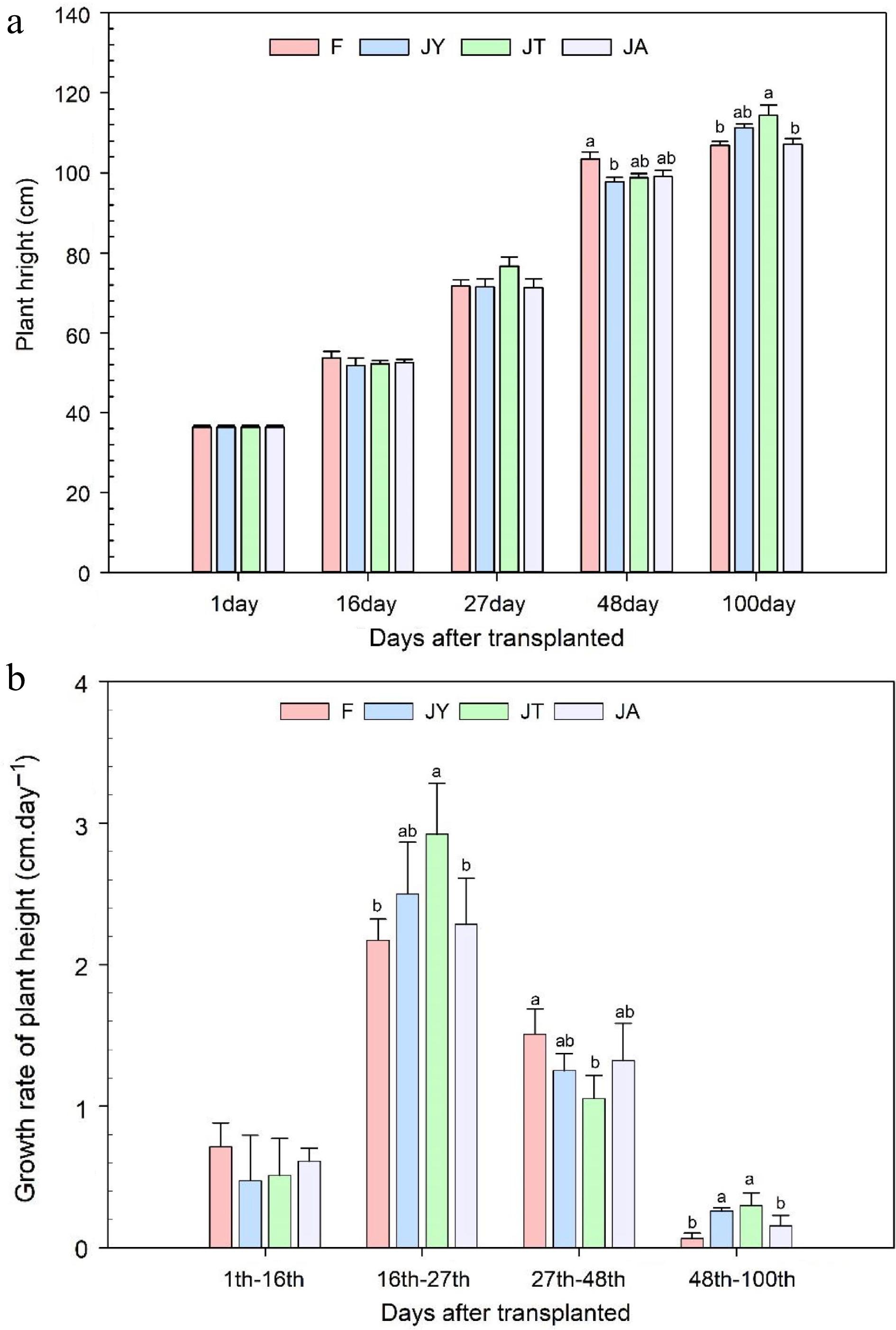
Figure 1.
(a) Rice plant height at the time of transplanting and after 16, 27, 48, and 100 d of transplanting and (b) growth rate during these growing periods. Note: F is chemical fertilizer treatment; JA is the liquid Jiaosu for soil treatment, liquid Jiaosu for leaf treatment, and solid Jiaosu for soil treatment; JT is the treatment of the liquid and solid Jiaosu for soil treatment; JY is liquid Jiaosu for leaf treatment and solid Jiaosu for soil treatment.
Effects of agricultural Jiaosu on rice biomass
-
After 48 d of transplanting, the aboveground biomass of rice treated with the three Jiaosu was similar to that of rice treated with conventional fertilizers. However, after 100 d of transplanting (harvest period), the conventional fertilizer treatment showed significantly higher biomass than the three agricultural Jiaosu treatments (Fig. 2a), with F (154.66 ± 14.05 g) > JA (134.91 ± 5.61 g) > JT (132.52 ± 8.94 g) > JY (119.98 ± 16.16 g) in descending order. The growth rate of aboveground biomass was similar for all four treatments after 48 d of transplanting. The growth rate of aboveground biomass in conventional fertilizer treatment was significantly higher than that in Jiaosu treatment from 48 d to 100 d after transplanting (Fig. 2b). The biomass growth rate of fertilizer treatment showed a gradually increasing trend from transplanting to 100 d after transplanting, while the biomass growth rates of JT and JA treatments were the lowest between transplanting to transplanting for 16 d (Fig. 2b; Supplemental Table S1). The biomass growth rate of the JY treatment was the lowest between 16 d of transplantation, followed by the period between 16 to 27 d after transplantation and then between 48 to 100 d after transplantation. The highest biomass growth rate was observed between 27 to 48 d after transplantation. Between 48 and 100 d of transplanting, the biomass growth rates of the JA and JT treatments in the Jiaosu treatment was similar, while JY treatment had the lowest growth rate.
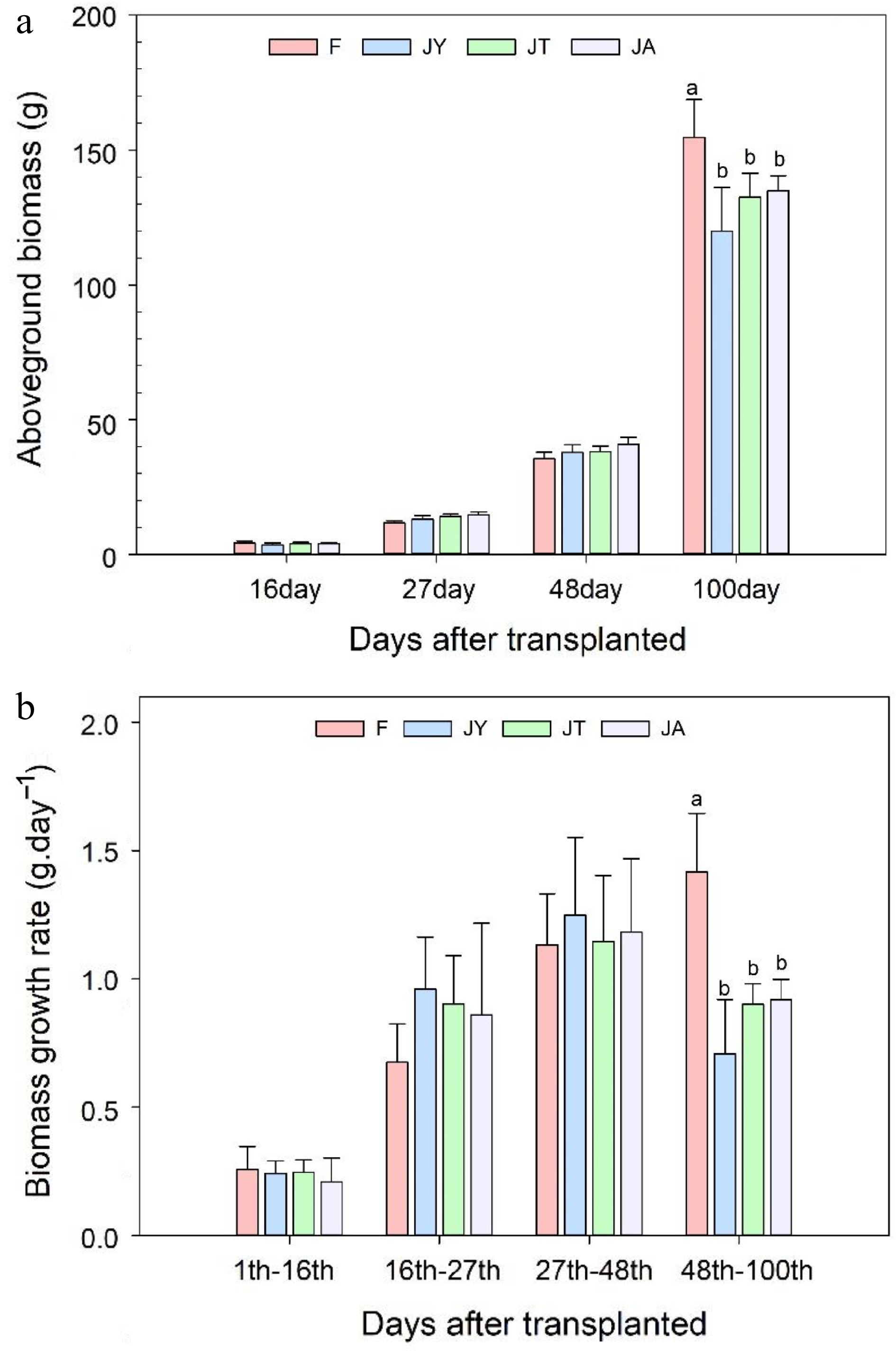
Figure 2.
(a) Aboveground biomass of rice after 16, 27, 48, and 100 d of transplanting and (b) biomass growth rates during these growth periods. Note: F is chemical fertilizer treatment; JA is the liquid Jiaosu for soil treatment, liquid Jiaosu for leaf treatment, and solid Jiaosu for soil treatment; JT is the treatment of the liquid and solid Jiaosu for soil treatment; JY is liquid Jiaosu for leaf treatment and solid Jiaosu for soil treatment.
After 100 d of transplanting, the underground biomass rank was as follows: JA (11.8 ± 1.3 g), F (11.4 ± 0.62 g), JT (9.9 ± 1.7 g), JY (8.4 ± 1.6 g) and (F = 5.24, p < 0.05, Fig. 3a). Compared with conventional fertilizer treatment, JA (0.0875 ± 0.01) and JT (0.0749 ± 0.01) treatments had a higher root-to-shoot ratio in rice compared to the F (0.0746 ± 0.01) treatment, while the JY (0.0708 ± 0.01) treatment was slightly lower than the F treatment (Fig. 3b). Both grain harvest index and grain-straw ratio was the lowest in fertilizer treatment (Fig. 3c, d). for grain harvest index the highest was in the JA (0.37 ± 0.02), followed by JT which was similar to JY, while the lowest was in the F treatment (F = 6.01, p < 0.05). The grain-straw ratio ranged from high to low as follows: JA, JT, JY, and F (F = 6.10, p < 0.01, Fig. 3d).
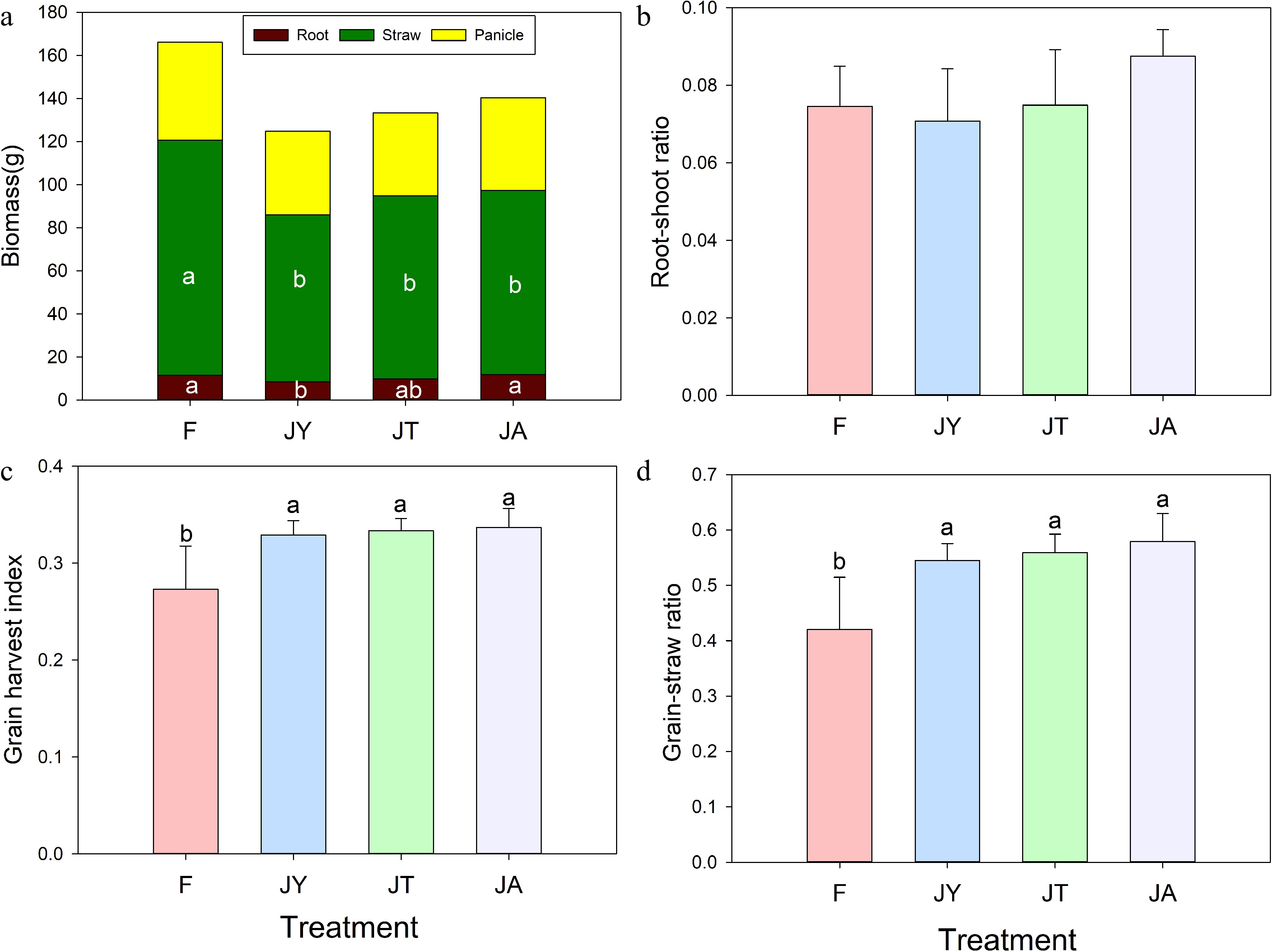
Figure 3.
(a) Total plant biomass, (b) root-shoot ratio, (c) grain harvest index, (d) grain-straw ratio at harvest stage of rice under different treatments. Note: F is chemical fertilizer treatment; JA is the liquid Jiaosu for soil treatment, liquid Jiaosu for leaf treatment, and solid Jiaosu for soil treatment; JT is the treatment of the liquid and solid Jiaosu for soil treatment; JY is liquid Jiaosu for leaf treatment and solid Jiaosu for soil treatment.
The correlation analysis showed that the plant height dynamics of the four treatments had a significant positive exponential growth relationship with aboveground biomass (Fig. 4). As the plant height increased, the JA (Q10 = 2.52 ± 0.10) treatment had the highest rate of aboveground biomass variation, followed by F (Q10 = 2.49 ± 0.09), JY (Q10 = 2.44 ± 0.08), and JT (Q10 = 2.37 ± 0.11).
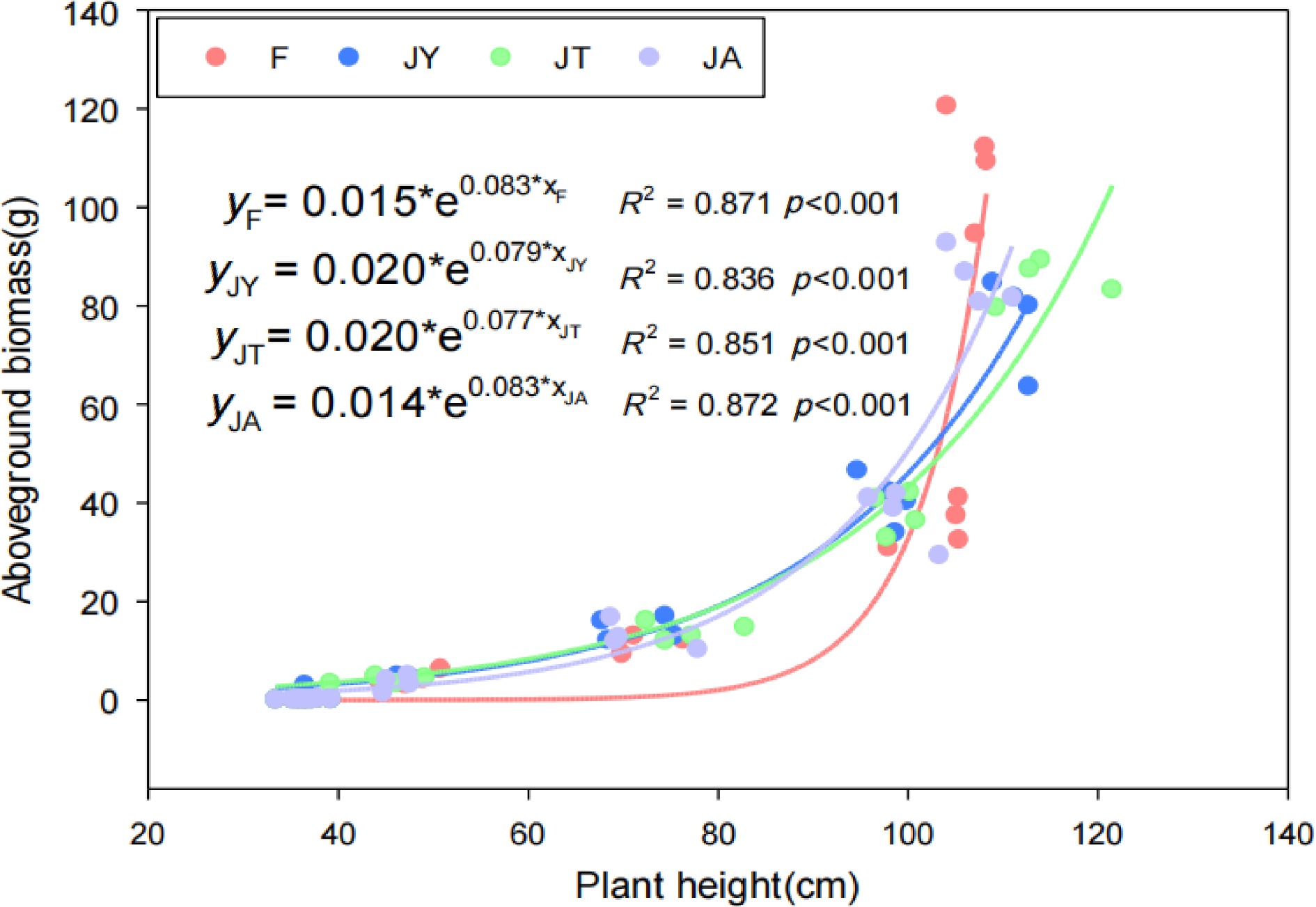
Figure 4.
Correlation between aboveground biomass and plant height. Note: F is chemical fertilizer treatment; JA is the liquid Jiaosu for soil treatment, liquid Jiaosu for leaf treatment, and solid Jiaosu for soil treatment; JT is the treatment of the liquid and solid Jiaosu for soil treatment; JY is liquid Jiaosu for leaf treatment and solid Jiaosu for soil treatment.
Effects of agricultural Jiaosu on the yield factors of rice
-
The actual yield of rice under JA treatment was the highest (49.35 ± 2.43 g·palnt−1), which increased by 8.51% compared to the conventional fertilizer treatment (45.47 ± 9.35 g·palnt−1). Compared with the yield per plant of rice under the fertilizer treatment, the JT treatment (47.56 ± 4.83 g·palnt−1) showed a slightly increased, while the JY treatment (42.40 ± 6.83 g·palnt−1) showed a slight decrease (Fig. 5a). The performance of yield components in each treatment is not consistent (Fig. 5b−f). The effective number of spikes in the three Jiaosu treatments was lower than that in the fertilizer treatment (19 spikes, F = 14.53, p < 0.01). Compared to the fertilizer treatment (145 grains), the number of grains per panicle increased in all three Jiaosu treatments, with JY (188 grains, 29.66%) showing the largest increase. The seed setting rate of the JA treatment (88.35%) is 5.93% higher than that of the fertilizer treatment. The spike length, grain density, and thousand-grain weight of the three agricultural Jiaosu treatments were higher than that of fertilizer treatment, but there was no significant difference. The correlation analysis and path analysis showed that effective panicles, thousand-grain weight, and number of grains per panicle have a significant positive effect on the yield of rice (Fig. 6). Effective panicles have the highest contribution rate to the yield of rice (r = 0.76, p < 0.001), while panicle length has a negative effect on the yield of rice (Fig. 6).
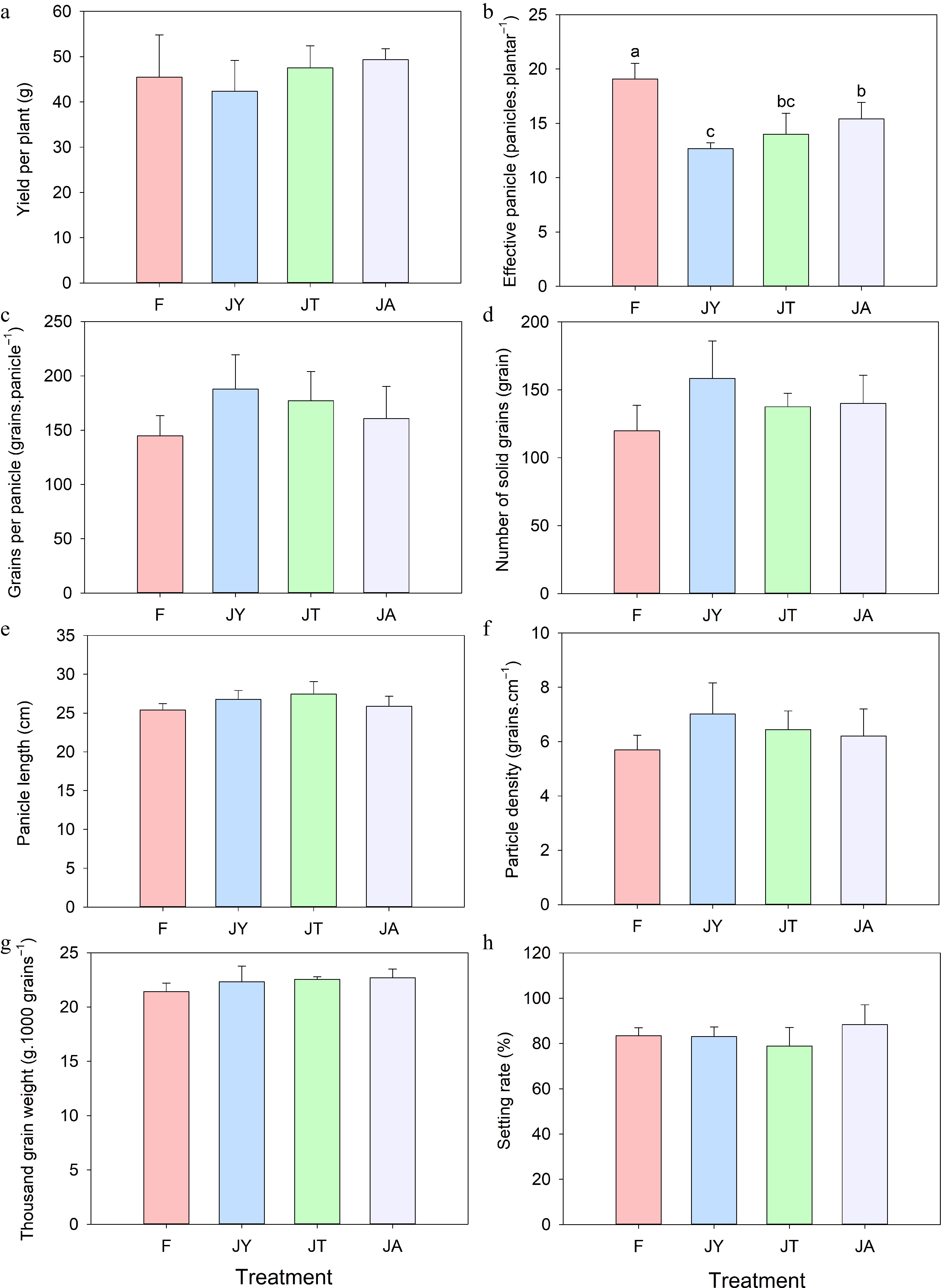
Figure 5.
Components of rice yield under different treatments: (a) Yield per plant, (b) effective panicle, (c) grains per panicle, (d) number of solid grains, (e) panicle length, (f) particle density, (g) thousand-grain weight, (h) setting rate. Note: F is chemical fertilizer treatment; JA is the liquid Jiaosu for soil treatment, liquid Jiaosu for leaf treatment, and solid Jiaosu for soil treatment; JT is the treatment of the liquid and solid Jiaosu for soil treatment; JY is liquid Jiaosu for leaf treatment and solid Jiaosu for soil treatment.
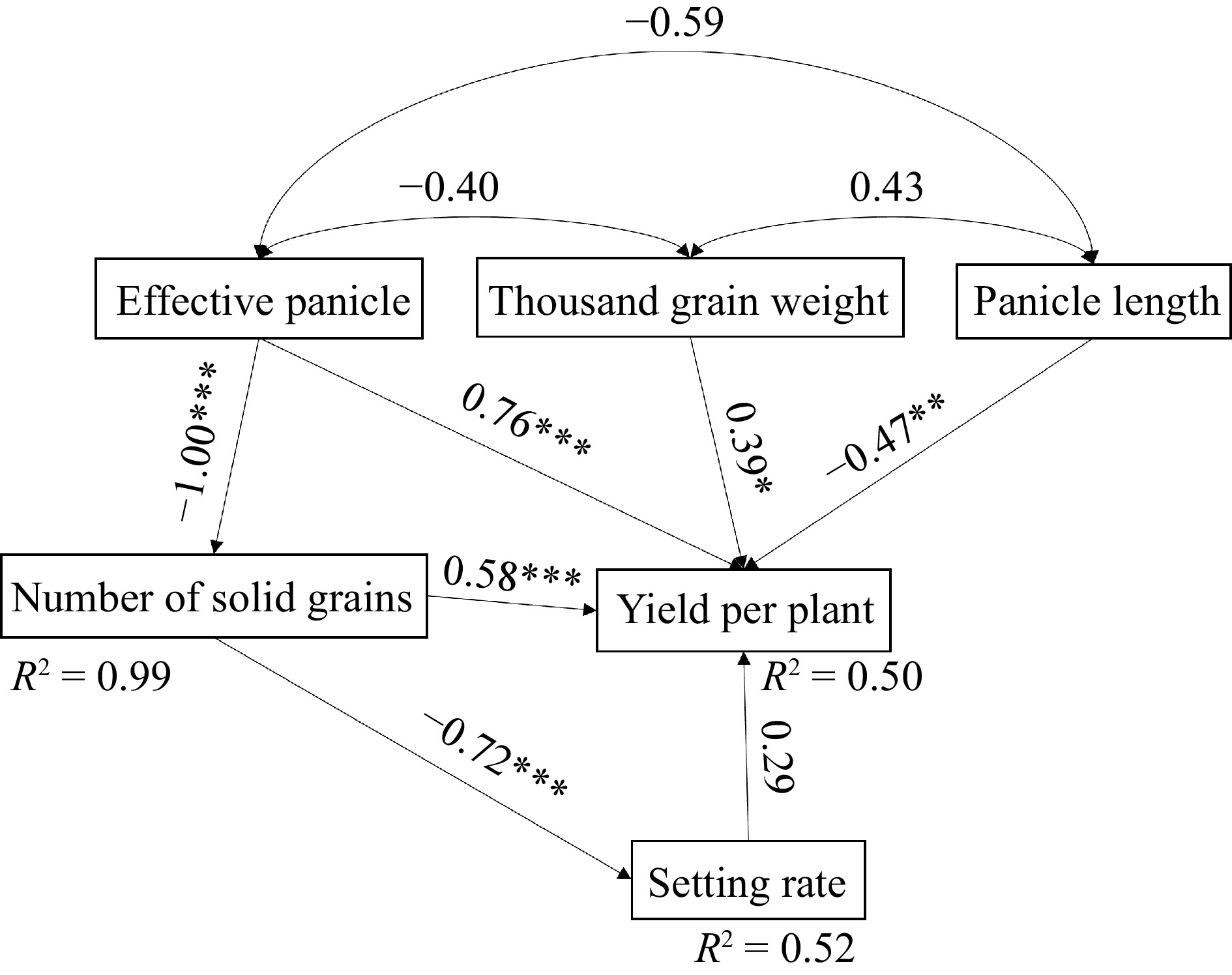
Figure 6.
Effect of yield components on yield per plant. Note: R² represent the proportion of variance explained by the variables in the model, and the arrow numbers represent the standard path coefficients. *, **, and *** indicate the statistically significant, highly significant, and extremely significant at p < 0.01, p < 0.05 and p < 0.001 level respectively.
Effects of agricultural Jiaosu on soil nutrient content
-
The soil available nitrogen content of conventional fertilizer treatment decreased from transplanting to 48 d transplanting, and then increased from 48 to 100 d transplanting. In contrast, the JA and JT treatments decreased from transplanting to 27 d after transplanting and then increased from 27 to 100 d after transplanting. The JY treatment only showed a decrease from transplanting to 16 to 27 d after transplanting, with slight increases in other growth stages (Fig. 7a). During transplantation, the soil available nitrogen content in the fertilizer treatment was twice that of the Jiaosu treatment. After 100 d of transplantation, the soil available nitrogen content in the fertilizer treatment was similar to that in the JA treatment, but lower than that in the JY and JT treatments (Fig. 7a). Along the stages of rice development, the available phosphorus content in soil treated with agricultural Jiaosu was relatively stable. Between 16 and 100 d of transplantation, the available phosphorus content in soil treated with Jiaosu was similar to that of conventional fertilizer treatment (Fig. 7b). At different stages of rice development, the soil available potassium content of the agricultural Jiaosu treatment was lower than that of the conventional fertilizer treatment, but its change trend was similar to that of the conventional fertilizer treatment. There was a decreasing trend in the period from self-transplantation to 48 d after transplantation, followed by a slight increase from 48 to 100 d after self-transplantation. Among the three Jiaosu treatments, the JT treatment had the highest soil-available potassium content (Fig. 7c).
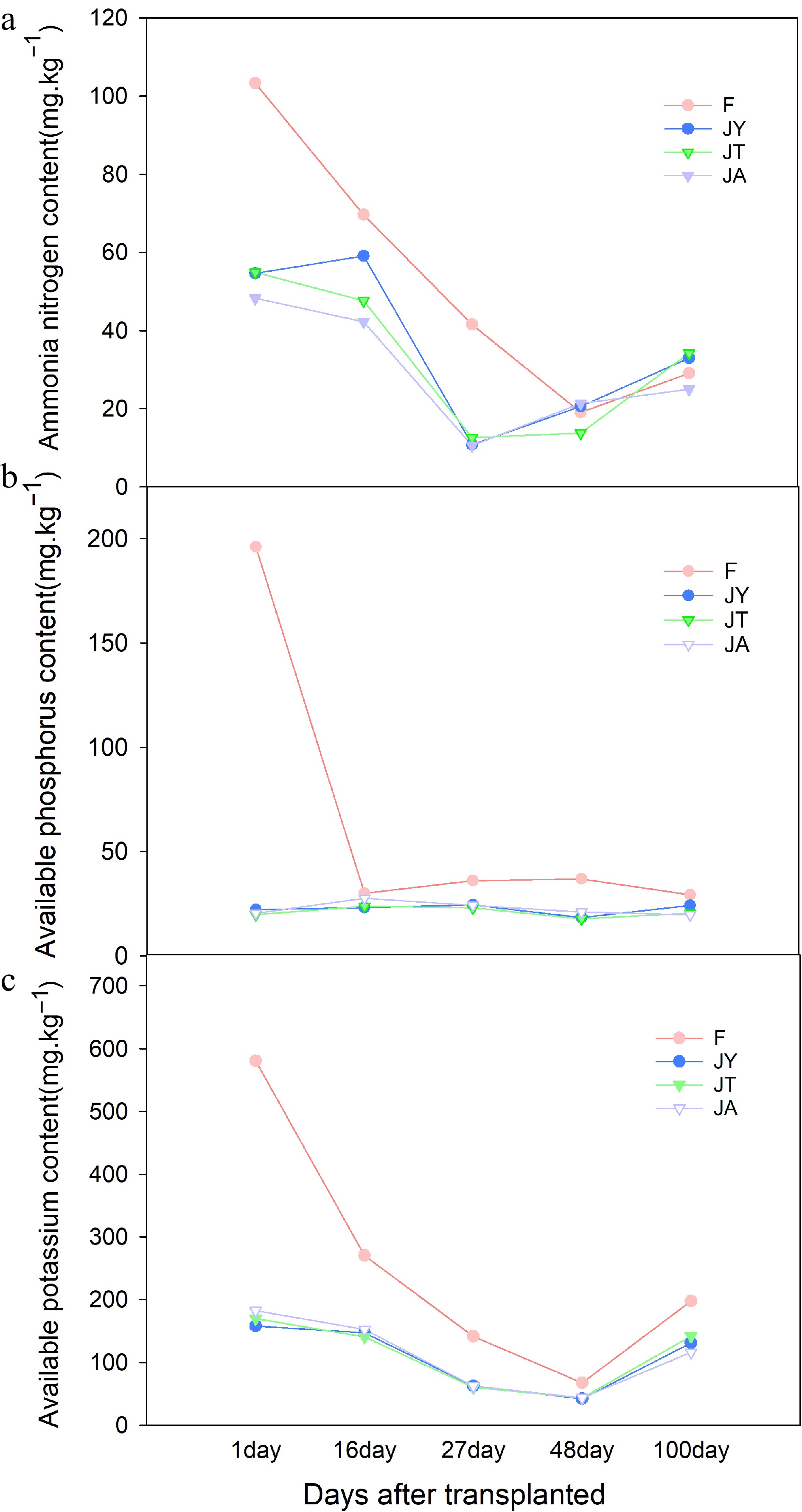
Figure 7.
Differences of soil available (a) nitrogen, (b) phosphorus, and (c) potassium content in different growth stages. Note: F is chemical fertilizer treatment; JA is the liquid Jiaosu for soil treatment, liquid Jiaosu for leaf treatment, and solid Jiaosu for soil treatment; JT is the treatment of the liquid and solid Jiaosu for soil treatment; JY is liquid Jiaosu for leaf treatment and solid Jiaosu for soil treatment.
Correlation between soil available nutrients and growth and yield
-
There was a negative correlation between soil available nitrogen content and plant height throughout the entire growth period (r = −0.460, p < 0.001). Soil available phosphorus content was significantly positively correlated with plant height after 48 d of transplanting (r = 0.690, p = 0.019). There was a significant negative correlation between available potassium and aboveground biomass at 48 d after transplanting (r = −0.760, p = 0.07), a significant positive correlation with aboveground biomass after 100 d of transplanting (r = 0.505, p = 0.046), and a negative correlation with plant height during the whole growth period (r = −0.292, p = 0.025).
The correlation between soil available nutrients and yield factors and biomass indicators during the harvest period (100 d after transplanting) was not uniform in each stage. There is a weak negative correlation between individual plant yield and soil available potassium after 48 and 100 d of transplantation (p < 0.01). The soil available nitrogen, phosphorus, and potassium content at the time of transplantation and 27 d of transplantation, the soil available potassium content at 16 d after transplantation, and the phosphorus and potassium content at 48 d after transplantation is significantly positively correlated with the number of effective panicles. There is a significant negative correlation between the total number of grains and the available nitrogen and phosphorus in the soil at the time of transplantation, the available phosphorus after 27 d of transplantation, and the available phosphorus and available potassium in the soil after 48 d of transplantation. The number of solid grains is negatively correlated with the soil available nitrogen and phosphorus at the time of transplantation, as well as the soil available nitrogen and phosphorus content at 27 and 48 d of transplantation. The spike length is significantly negatively correlated with the soil available nitrogen and phosphorus at the time of transplantation, as well as the soil available phosphorus at 27 d of transplantation and the soil available potassium at 48 d of transplantation. There is a significant negative correlation between grain density and soil-available phosphorus during transplantation, as well as soil available phosphorus and available potassium after 48 d of transplantation. There is a negative correlation between 1000-grain weight and soil's available nitrogen and available phosphorus during transplantation, soil's available phosphorus and available potassium after 27 and 48 d of transplantation, and soil-available potassium after 100 d of transplantation. There is a negative correlation between plant height and soil available phosphorus after 27 and 48 d of transplantation. The biomass of individual straw is significantly positively correlated with the soil available nitrogen content at the time of transplantation and 27 d after transplantation, the soil available phosphorus content from transplantation to 48 d after transplantation, and the available potassium content throughout the growth period.
The grain straw ratio is significantly negatively correlated with the soil's available nitrogen content at transplanting and 27 d after transplanting and the soil available potassium content throughout the growth period, while significantly positively correlated with the soil-available phosphorus content from transplanting to 48 d after transplanting. There is a significant positive correlation between the underground biomass after 100 d of transplantation and the soil available phosphorus content after 16 d of transplantation.
-
Agricultural Jiaosu has been shown to promote crop growth under conditions of low nutrient input[15]. In this study, rice plants treated with three different agricultural Jiaosu did not show significant differences in plant height at 16 and 27 d after transplanting. However, at 48 d after transplanting, the plant height in the fertilizer-treated group was significantly higher than in the JT and JY treatment groups. The plant height in the fertilizer-treated group was higher than in the JA treatment group, although the difference was not significant (Fig. 1a). Additionally, the growth rate of plant height in the JA treatment group was comparable to that in the fertilizer-treated group at each growth stage (Fig. 1b). These results indicate that the combined use of three Jiaosu does not affect the growth of rice plant height under conditions of low N, P, and K fertilizer input. There was no significant correlation between the effective nitrogen content in the soil at each growth stage and plant height. However, throughout the entire growth period, there was a negative correlation between the content of effective nitrogen and available potassium in the soil and plant height (Table 2). Furthermore, plant height at harvest decreased with increasing soil available phosphorus content at 16 and 27 d after transplanting (Table 3). These findings suggest a significant negative correlation between the nitrogen, phosphorus, and potassium content directly utilized by rice at a certain growth stage and plant height. This finding appears inconsistent with the conclusion that there is a significant positive correlation between rice plant height and the application of nitrogen, phosphorus, and potassium fertilizers during the fertilization process[16]. This indicates that the growth of rice plant height after the application of agricultural Jiaosu is not directly correlated with nutrient content. Compared to the plant height 16 d after transplanting, the increase in plant height of rice with fertilizer treatment 27 d after transplanting was lower compared to the three Jiaosu treatments. This trend was also evident in the differences between transplanting periods of 48 to 100 d. Only during the vigorous nutrient growth period, the plant height growth rate of rice with fertilizer treatment was higher compared to the Jiaosu treatments at 27 and 48 d after transplanting (Supplemental Fig. S1a), indicating a differential regulatory mechanism of plant height in rice between agricultural Jiaosu and fertilizers. These differences suggest that in addition to nutrient influences, plant height in rice is also influenced by growth hormones[17]. Liquid agricultural Jiaosu applied to the soil and foliage of rice plants resulted in a faster growth rate of plant height between 16 and 27 d after transplanting compared to conventional fertilizer treatment and JA treatment (Fig. 1b). Additionally, plant height with JT treatment was significantly higher than conventional fertilizer treatment 100 d after transplanting (Fig. 1a). This indicates that in the later stages of growth, the regulation of plant height in rice by liquid agricultural Jiaosu applied to the soil and foliage is superior to fertilizer treatment. However, the differences in plant height growth compared to the full Jiaosu treatment could not be explained in this study. Furthermore, there were no significant differences in aboveground biomass and growth rates at different growth stages from transplanting to 48 d after transplanting (Fig. 2a). However, the growth rate of aboveground biomass with fertilizer treatment between 48 and 100 d after transplanting was much higher than the three Jiaosu treatments (Fig. 2b, Supplemental Fig.S1b), resulting in significantly higher aboveground biomass with fertilizer treatment compared to Jiaosu treatments (Fig. 2a). The aboveground biomass at 27, 48, and 100 d after transplanting was all regulated by soil available potassium, showing a positive correlation. This contradicts the conclusion that increasing potassium application promotes aboveground biomass growth[18,19].
Table 2. Relationship between soil available nitrogen, phosphorus and potassium content and plant growth index.
N P K r P r P r P 16 d after transplanted Plant height −0.132 0.626 0.291 0.274 0.308 0.246 Aboveground biomass 0.207 0.442 0.344 0.192 0.123 0.650 27 d after transplanted Plant height −0.152 0.574 −0.192 0.477 −0.311 0.241 Aboveground biomass −0.382 0.145 −0.491* 0.053 −0.470* 0.066 48 d after transplanted Plant height −0.098 0.775 0.690** 0.019 0.296 0.377 Aboveground biomass −0.309 0.355 −0.348 0.294 −0.760*** 0.007 100 d after transplanted Plant height 0.405 0.120 −0.221 0.411 −0.107 0.693 Aboveground biomass −0.191 0.478 0.300 0.259 0.505** 0.046 Whole growth period Plant height −0.460*** 0.000 0.015 0.911 −0.292** 0.025 Aboveground biomass −0.208 0.115 0.057 0.667 0.065 0.622 '*', '**', and '***' indicate the statistically significant, highly significant, and extremely significant at p < 0.01, p < 0.05, and p < 0.001 level respectively. r in the table is Pearson correlation coefficient, P in the table is p-value in the Pearson correlation analysis. N, P, and K represent soil ammonium nitrogen, available phosphorus, and available potassium, respectively. Table 3. Correlation between soil available nitrogen, phosphorus and potassium at each growth stage and yield and biomass at harvest stage.
6 d after transplanted 16 d after transplanted 27 d after transplanted 48 d after transplanted 100 d after transplanted A.N A.P A.K A.N A.P A.K A.N A.P A.K A.N A.P A.K A.N A.P A.K Yield per plant −0.536* −0.44* Effective panicle 0.711*** 0.705*** 0.517** 0.733*** 0.660*** 0.751*** 0.749*** 0.646** 0.745*** Total grain −0.448* −0.501* −0.442* −0.684** −0.720** Solid grain −0.511** −0.576** −.529** −0.493* −0.536* −0.758*** Panicle length −0.464* −0.469* −0.481* −0.541* Grain density −0.456* −0.666** −0.711** 1000-grain weight −0.434* −0.564** −0.507** −0.435* −0.748*** −0.787*** −0.577** Plant height −0.467* −0.466* Straw weight per plant 0.815*** 0.630** 0.612** 0.446* 0.833*** 0.776*** 0.793*** 0.758** 0.684** 0.681** 0.505** Aboveground biomass 0.680*** 0.489* 0.709*** 0.759*** 0.643*** 0.711** 0.547* Underground biomass 0.706*** Total biomass 0.668*** 0.493* 0.709*** 0.737*** 0.638*** 0.695** 0.556* Total biomass per spike −0.701*** −0.663*** −0.786*** −0.475* −0.474* −0.707*** −0.442* −0.698*** −0.490* −0.794*** −0.624** −0.640*** Head ratio straw −0.723*** −0.666*** −0.769*** −0.475* −0.712*** −0.472* −0.704*** −0.522* −0.777*** −0.624** −0.666*** '*', '**', and '***' indicate the statistically significant, highly significant, and extremely significant at p < 0.01, p < 0.05, and p < 0.001 level respectively. A.N, A.P, and A.K represent soil ammonium nitrogen, available phosphorus, and available potassium, respectively.. In this study, a significant exponential correlation was observed between the plant height and aboveground biomass of rice plants under four different treatments. However, there was no significant difference in the rate of increase in aboveground biomass with plant height, indicating that the application of agricultural Jiaosu did not alter the dry matter accumulation pattern of rice growth (Fig. 4). Nevertheless, the application of agricultural Jiaosu did change the biomass allocation pattern of rice plants at harvest (Fig. 3). Overall, the total biomass was highest for the fertilizer treatment, followed by the full Jiaosu treatment, while the biomass was lowest for the treatments using only solid agricultural Jiaosu or foliar liquid agricultural Jiaosu (Fig. 3a). All three Jiaosu treatments increased the proportion of grains to aboveground biomass and the ratio of grains to straw biomass (Fig. 3c,d), indicating that Jiaosu -treated rice plants allocate a higher proportion of nutrients and energy to the reproductive growth process[20]. Furthermore, the application of liquid agricultural Jiaosu to the soil resulted in a significant increase in underground biomass compared to the foliar application of liquid agricultural Jiaosu (Fig. 3a). Moreover, treatments combining soil liquid agricultural Jiaosu with solid Jiaosu showed an increased root-to-shoot ratio (Fig. 3b), with the underground biomass of the foliar liquid agricultural Jiaosu combined with solid Jiaosu treatment being lower than that of the soil liquid agricultural Jiaosu combined with solid Jiaosu treatment and the full Jiaosu treatment. This study demonstrates that agricultural Jiaosu can promote the reproductive growth of rice plants, further manifested in the 8.51% higher yield per plant in rice plants treated with full Jiaosu compared to the fertilizer treatment, while the yields of rice plants under the JT and JY treatments were comparable to those under the fertilizer treatment (Fig. 5a, Supplemental Fig. S2a). The single plant yield of rice is directly related to several parameters such as effective panicles, number of grains per panicle, seed setting rate, and thousand-grain weight. However, the panicle length, grain density, number of grains per panicle, and thousand-grain weight of the JA, JT, and JY treatments were all higher than those of the fertilizer treatment (Fig. 5, Supplemental Fig. S2), indicating that the application of agricultural Jiaosu promoted panicle development during the reproductive growth stage of rice. The specific molecular biological mechanisms underlying this regulation remain unclear. The increase in the number of grains per panicle, seed setting rate, and thousand-grain weight in the JA treatment offset the decrease in effective panicles, leading to an increase in the yield of the JA treatment (Fig. 5a). Although the seed setting rate of the JT treatment was slightly lower than that of the F treatment, the increase in the number of grains per panicle and thousand-grain weight ensured the yield of the JT treatment. The slight decrease in the single plant yield of the JY treatment may be due to a reduction in the seed setting rate. Throughout the entire growth cycle, the nutrient input of available nitrogen, available phosphorus, and quick-acting potassium in the soil for the three Jiaosu treatments was lower than that of the conventional fertilizer treatment (Table 1, Fig. 7). The relationship between the elements of yield and biomass at the harvest stage and the content of available nitrogen, available phosphorus, and quick-acting potassium in the soil at each growth stage in this study contradicts conventional research conclusions[21,22].
This study demonstrates a positive correlation between soil available nitrogen, available phosphorus, quick-acting potassium, and effective panicle, straw, aboveground biomass, and total biomass at the time of transplanting and 27 d after transplanting. However, panicle length, grain number per panicle, filled grain number, thousand-grain weight, and panicle-to-straw ratio, as well as panicle-to-total biomass ratio, showed a negative correlation with nutrient content at various growth stages (Table 3). Although the application of Jiaosu reduced the number of effective panicles, it promoted the thousand-grain weight, panicle length, grain number per panicle, and filled grain number during the reproductive growth stage[12,23−26], demonstrating the growth-promoting effect of agricultural Jiaosu in the mid to late stages of rice growth[27,28]. Compared to low nitrogen, phosphorus, and potassium supply, rice had more tillers under higher low nitrogen, phosphorus, and potassium supply[16]. This study observed that the onset of tillering in rice treated with Jiaosu was later than fertilizer-treated rice, and the effective tiller number in rice treated with Jiaosu at harvest was lower than in fertilizer-treated rice, possibly due to the lower nutrient supply from agricultural Jiaosu treatment (Fig. 5b, Supplemental Fig. S3). From day 48 to day 100 after transplanting rice, the growth rate of plant height was faster in rice treated with agricultural Jiaosu compared to fertilizer treatment (Fig. 1b), the growth rate of biomass was slower in Jiaosu-treated rice compared to fertilizer treatment (Fig. 2b), but the panicle biomass was higher in Jiaosu-treated rice compared to fertilizer treatment. Agricultural Jiaosu activate the total nitrogen, phosphorus, and potassium in the soil, maintaining the levels of available nitrogen, available phosphorus, and quick-acting potassium relatively stable throughout the entire growth cycle[13,29,30]. In contrast, nitrogen, phosphorus, and potassium from fertilizer treatments is leached due to percolation, resulting in a much smaller difference in the levels of available nitrogen, available phosphorus, and quick-acting potassium between Jiaosu-treated soil and fertilized soil 48 to 100 d after rice transplantation compared to the period from 27 d after transplantation (Fig. 7, Supplemental Fig. S2). This indicates that under conditions of equivalent nutrient supply during the reproductive growth stage of rice, agricultural Jiaosu has the effect of promoting rice reproductive growth, enhancing nutrient utilization efficiency, and further demonstrating that agricultural Jiaosu adjusts the dry matter distribution characteristics of rice.
Agricultural Jiaosu is a type of enzyme that can improve fertilizer efficiency, reduce the use of chemical fertilizers and pesticides, and decrease organic fertilizer pollution in farmland[29]. The agricultural Jiaosu utilized in this study are derived mainly from various plant waste sources such as fruits, grains, and oil crops. Even when the total nitrogen, phosphorus, and potassium input amounts are 28%, 13%, and 15% of the input amounts in conventional fertilizer treatments, they can still achieve the effect of maintaining and increasing crop yields. Therefore, agricultural Jiaosu plays an irreplaceable role in the future development of circular agriculture. It is imperative to investigate the effects of agricultural Jiaosu dosage and application timing on rice production, and to explore the regulatory mechanisms of agricultural Jiaosu on rice physiology and soil nutrient supply by integrating molecular biology and microbiology.
-
The results of this study suggest that agricultural Jiaosu enhances nutrient utilization efficiency in low nutrient input conditions. Plant height increased by 7.01%, 4.18%, and 0.24% in the JA, JT, and JY treatments, respectively, compared to the conventional fertilizer treatment. Additionally, these treatments resulted in higher yields, with the JA treatment showing an 8.51% increase in yield compared to the conventional fertilizer treatment. It was found that Jiaosu adjusted the dry matter distribution characteristics of rice, with the grain harvest index and the grain-straw ratio of Jiaosu-treated being higher than those of conventionally fertilized. Although the total biomass and effective tillering number of rice were reduced after utilization of agricultural Jiaosu, other factors such as the ratio of spike to straw, the rate of spike to above-ground biomass, spike length, grain number, and 1000-grain weight increased, thereby improving rice yield. Furthermore, the agricultural Jiaosu used in this study has great potential for application in the future development of circular agriculture. However, further research is needed to elucidate required to determine the molecular and microbiological mechanisms by which agricultural Jiaosu regulate rice growth and nutrient supply. This will provide a more direct scientific basis and efficient production methods for circular agriculture.
-
The authors confirm contribution to the paper as follows: study conception and design: Zhang R, Zhou W; data collection: Zhang R, Zhou W, Zhang C, Xiong X, Zhou G, Yi Y, Hong SS, Li J, Song Q, Ye F, Liu Y; analysis and interpretation of results: Zhang R, Zhou W; draft manuscript preparation: Zhang R, Zhou W. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed during the current study are not publicly available as the entire research project has not been finalized, and the research findings have not yet been published, but are available from the corresponding author on reasonable request.
This work was supported by National Natural Science Foundation of China (42073080, 32361143516), the Yichun Innovation Driven 5511 Project in 2019, and Yunnan Provincial Department of Scientific and Technology (202205AC160045; 202204BL090014) China. We also thank the postdoctoral project (2021) of the Department of Human Resources and Social Security of Yunnan. We appreciate the staff and technicians of the Climate Change Research Group, XTBG, CAS, and students of the Yichun University, and the staff and technicians of Yichun Academy of Sciences for helping with field and laboratory work. We also thank the Institutional Center for Shared Technologies and Facilities of Xishuangbanna Tropical Botanical Garden, CAS for helping with the soil analyses.
-
The authors declare that they have no conflict of interest.
- Supplemental Table S1 Growth rate of biomass and plant height.
- Supplemental Fig. S1 Growth ratio of plant height and straw biomass in different periods.
- Supplemental Fig. S2 The variation level of yield factors treated with three agricultural JiaoSu compared with that treated with chemical fertilizer.
- Supplemental Fig. S3 Content of available nitrogen, phosphorus and potassium in soil at each growth period of rice.
- Copyright: © 2024 by the author(s). Published by Maximum Academic Press, Fayetteville, GA. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Zhang R, Zhang C, Xiong X, Zhou G, Yi Y, et al. 2024. Improving growth and yield of rice through the use of agricultural Jiaosu in different combinations. Circular Agricultural Systems 4: e012 doi: 10.48130/cas-0024-0011
Improving growth and yield of rice through the use of agricultural Jiaosu in different combinations
- Received: 05 March 2024
- Revised: 05 April 2024
- Accepted: 06 April 2024
- Published online: 04 July 2024
Abstract: Agricultural Jiaosu is rich in nutrients and plant-beneficial microbial communities, and thus has great potential in reducing fertilizer dosage and improving fertilizer use efficiency. This pot experiment was conducted to investigate the effects of agricultural Jiaosu on the growth and yield of rice. The experiment employed three agricultural Jiaosu treatments (i.e., soil solid agricultural Jiaosu, soil liquid agricultural Jiaosu, leaf liquid agricultural Jiaosu) in various combinations, along with one control treatment (chemical fertilizer). Results revealed that plant height of rive agricultural Jiaosu treatments were higher than that of the control treatment, with the ranks as follows: JT (soil solid agricultural Jiaosu + soil liquid agricultural Jiaosu), JY (soil solid agricultural Jiaosu + leaf liquid agricultural Jiaosu), JA (soil solid agricultural Jiaosu + soil liquid agricultural Jiaosu + leaf liquid agricultural Jiaosu) and F (conventional fertilizer) during harvest. The ratio of panicle biomass to total biomass and straw of three types of Jiaosu treatments was significantly higher than that in the chemical fertilizer treatment, indicating that agricultural Jiaosu treatment altered the dry matter distribution characteristics of rice. The yield per plant of JA treatment (49.35 ± 2.43 g) was 8.51% higher than chemical fertilizer treatment. Additionally, the correlation between soil nutrients and plant growth and yield analysis indicated that agricultural Jiaosu has higher nutrient use efficiency with lower input of N, P, and K. The study highlights the potential function of agricultural Jiaosu in promoting crop growth and yields while reducing the need for fertilization.
-
Key words:
- Agricultural Jiaosu /
- Rice /
- Biomass /
- Plant height /
- Yield /
- Nutrient utilization efficiency













