-
Camels are primarily found in Africa and the Middle East, although some populations also exist in other parts of the world. Somalia is believed to have the highest number of camels in Africa, with approximately six million individuals. Other countries in Africa such as Sudan, Mauritania, and Ethiopia also have significant camel populations, with millions of camels living within their borders. Although camels cannot survive in the wild in the United States due to the climate, they can still be observed in zoos across the country[1]. In the Middle East, countries like Saudi Arabia and Iran are also known for their notable camel populations. Additionally, Australia is home to the largest herd of camels in the world, estimated to include around 750,000 individuals[2].
The Camelidae family encompasses various members, including the dromedary (Camelus dromedarius), Bactrian camel (Camelus bactrianus), llama (Lama glama), guanaco (Lama guanicoe), alpaca (Vicugna pacos), and vicuna (Vicugna vicugna). These species have adapted to thrive in diverse environments such as plains, mountains, deserts, and arid regions where water and food scarcity are prevalent[3].
Historically, camels have played an indispensable role in the livelihoods of pastoral communities, providing them with essential resources such as meat, wool, and milk[4]. The nutritional and therapeutic benefits of camel milk, coupled with its unique intrinsic qualities have contributed to its growing popularity[5]. Despite its significance, camel milk constitutes a mere 0.2% of the global milk supply, compared with cow milk accounting for approximately 85%, and buffalo, goat, and sheep milk representing 11.0%, 2.3%, and 1.4%, respectively[6].
In pastoral regions where camel rearing is prevalent, camel milk is typically consumed fresh or allowed to ferment naturally into a sour state. While camel milk shares a similar overall composition with cow milk, it distinguishes itself through variations in molecular structure, distribution, and relative composition of its components[7]. Consequently, processing camel milk and creating dairy products like cheese, yogurt, and butter using techniques designed for bovine milk has proven challenging. When such products are successfully produced, they are often of inferior quality. Nevertheless, scientific research suggests that by optimizing processing parameters, it is possible to convert camel milk into high-quality dairy products[7].
Camel milk is characterized by standard composition components such as lipids, total protein, lactose, dry matter, and ash, with approximate ranges of 3.82 ± 1.08, 3.35 ± 0.62, 4.46 ± 1.03, 12.47 ± 1.53, and 0.79 ± 0.09 (g/100 mL) respectively[8]. It contains various bioactive fractions that contribute to human well-being, including lactoperoxidase, hydrogen peroxide, lactoferrin, lysozyme, immunoglobulin, and free fatty acids[9].
Camel milk has demonstrated notable effects in promoting human health, such as its anti-inflammatory and anti-carcinogenic properties[10], the potential to manage asthma[11], efficacy in addressing milk protein allergies[12], and its role in combating hepatitis C and B[13]. Additionally, camel milk has shown antimicrobial activity[14], neuroprotective and anticonvulsant effects in a strychnine-induced seizure paradigm[15], and antioxidant intervention in autistic children[16].
In addition, camel milk-derived peptides have shown the ability to enhance the transport and absorption of crucial minerals like calcium and magnesium. These peptides possess substantial biological activity, offering anticoagulant effects, oxidative functions, antihypertensive properties, and the potential for managing hypercholesterolemia[17].
The quest for an improved and more efficient oral insulin delivery system is recognized as an ideal strategy for diabetes prevention and management. In this regard, camel milk emerges as a promising solution due to its substantial presence of insulin-like proteins or other small molecular elements that mimic insulin's interaction with its receptor, surpassing other types of ruminant milk[17]. This article aims to comprehensively analyze the physicochemical composition, significant bioactive components, and therapeutic characteristics of camel milk as an alternative dietary strategy for treating various chronic and infectious diseases.
-
Camel milk is a physiologically complex fluid that typically appears opaque white, primarily due to its lower carotene content compared to cow's milk. It possesses a well-homogenized fat content and exhibits a regular milky odor. Regarding flavor, camel milk has a distinctive sweet-salty taste that combines a sharp sweetness, which can vary based on factors such as the animal's feed and the availability of drinking water[18].
In terms of density, camel milk has an average range of 1.01–1.02 g cm−3, slightly lower than the average density of cow milk, which falls within 1.02–1.03 g cm−3. Furthermore, camel milk demonstrates a mean viscosity of 2.2–2.35 cPas, which is higher compared to cow's milk (1.7 cPas), goat's milk (2.12 cPas), and buffalo milk (2.2 cPas), but slightly lower than sheep's milk (2.48 cPas)[5].
Camel milk and sheep milk exhibit similar pH levels, typically ranging from 6.4 to 6.7. However, the pH of camel and sheep milk is slightly lower compared to cow milk[19]. This difference may be attributed to higher levels of vitamin C and certain organic acids present in camel and sheep milk. Additionally, the development of acidity in camel milk occurs at a relatively slower rate compared to cow milk[20].
The freezing point of camel milk typically falls within the range of –0.57 to –0.61 °C, while cow milk has a freezing point range of –0.51 to –0.56 °C. In terms of calorific value, camel milk has a lower value of 665 kcal/L compared to cow milk, which has a calorific value of 701 kcal/L. This lower calorific value in camel milk can be attributed to its lower total solid content, including lactose, fat, and protein content. Additionally, the specific gravity values of camel milk and cow milk are relatively similar, with slight variations. Camel milk has density values ranging from approximately 1,028 to 1,033 kg/m3, while cow milk has a range of 1,026 to 1,034 kg/m3[5].
Shelf life of camel milk
-
Heat treatment is a crucial initial step in milk processing, aimed at extending the shelf life of the final product by reducing the bacterial load and activity of enzymes present in the milk[21].
The shelf life of raw camel milk is greatly influenced by its storage conditions. When stored at 25 °C, raw camel milk can remain suitable for consumption for approximately 24 h, with minimal changes in its composition. Alternatively, refrigerating raw camel milk at 4 °C can extend its usability to about 48 h. However, if the milk is kept at 37 °C, its shelf life is shorter, typically lasting around 8−9 h. Nonetheless, the activation of the camel lactoperoxidase system can prolong this duration to 18−20 h. For optimal preservation, storing raw camel milk at temperatures of 4−6 °C can potentially extend its shelf life up to one week under suitable conditions[22].
The thermal stability of camel milk is influenced by its protein content, which results in poorer stability at high temperatures compared to bovine milk, particularly at temperatures reaching 140 °C[18]. This reduced stability can be attributed to the absence of β-lactoglobulin and κ-casein in camel milk when compared to bovine milk. These proteins play a crucial role in maintaining thermal stability. Furthermore, heat treatment has a significant impact on the antibacterial activity of camel milk, as well as cow and buffalo milk. A study conducted by El-Agamy et al.[12] investigated the effect of heat treatment on lysozyme, lactoferrin, and IgG levels in these milk samples. When cow and buffalo milk samples were subjected to a heat treatment of 65–75 °C for 0.5 h, the protective protein IgG was eliminated after the treatment at 75 °C. However, in camel milk samples, lysozyme, and lactoferrin remained unaffected by this heat treatment, indicating their thermal resistance. In addition, when camel milk and its derivatives were heat-treated for 0.5 h at 70 °C, the defensive activity of the lactoperoxidase enzyme against pathogens was considerably weakened. This highlights the potential use of the lactoperoxidase enzyme as an indicator of the effectiveness of camel milk pasteurization[23].
-
Table 1 compares the chemical compositions of camel and cow's milk. Camel milk's chemical contents are less stable than that of cow milk. Nevertheless, various factors can influence camel milk content, including age, breed, feed type, surrounding topography, and milking season[19].
Table 1. Comparison of chemical compositions of camel and cow milk.
Composition Camel milk Cow's milk Ref. Water content (%) 87–91 85–87 [22,24,56] Total solids (%) 9–13 12.7–13 Milk solids-not-fat (MSNF) (%) 8.45 9.0 Fat (%) 1.2–5 3–5 Protein (%) 2.7–4.5 2.7–4.7 Lactose (%) 2.4–5.8 3.7–5.2 Minerals Higher content of calcium, potassium, phosphorus, magnesium Lower content of calcium, potassium, phosphorus, magnesium Ash (%) 0.6–0.9 0.7–0.8 Vitamins Similar content of vitamins C, B, A, and D Similar content of vitamins C, B, A, and D pH 6.5–6.7 6.6–6.8 Casein Different casein profiles with unique types and ratios (e.g., higher alpha-s1 casein) Different casein profiles with distinct types and ratios Enzyme activity Unique enzymes (e.g., higher lysozyme activity, lactoperoxidase) Varied enzyme activity (e.g., lactase, lipase) Immunoglobulins Higher content of immunoglobulins (e.g., IgG, IgA) Lower content of immunoglobulins Insulin (IU/ml) 40.50–58.67 17.01 Energy (kcal) 66.1–88.9 76.2 * Please note that the values mentioned in the table are approximate ranges and can vary depending on various factors such as breed, age, diet, and other environmental conditions. Additionally, camel milk's composition can vary significantly based on the region and season. Water content
-
The water content is a crucial determinant of milk composition. Cow's milk typically contains approximately 85–87% water. In the case of camel milk, its water content varies depending on environmental factors. During periods of abundant drinking water, camel milk is around 87% water. However, in the summer when temperatures are high, drinking water becomes scarce, and pastures are overgrazed, the water content in camel milk can increase significantly, reaching up to 91%. This variation in water content highlights the adaptive nature of camel milk production in response to challenging environmental conditions. Camels are known for their remarkable ability to conserve water and adapt to arid regions. During the summer, when water resources are limited, camels can maintain milk production by increasing the water content in their milk. This adjustment ensures the hydration and nourishment of their offspring even in harsh conditions. It is important to consider the effect of seasonal variations and environmental factors on milk composition, as they can significantly impact the nutritional profile and quality of camel milk. Understanding these dynamics is crucial for sustainable milk production and the development of effective management strategies in camel farming[22,24].
Fat content
-
The fat content in camel milk typically ranges from 1.8% to 4.3%. However, in situations where camels are experiencing thirst, such as during water scarcity, the fat content can decrease significantly, dropping from 4.3% to 1.1%[19]. Despite these variations, camel milk fat globule membranes possess several health-promoting characteristics that make it a favorable option for individuals allergic to cow's milk. The unique composition of camel milk fat globule membranes contributes to its anti-adhesion and anti-bacterial capabilities, which can provide health benefits. Additionally, the presence of small milk fat globules in camel milk aids in digestion, making it easier for the body to process.
In terms of its lipid profile, camel milk has lower levels of cholesterol and saturated fatty acids compared to cow milk. Conversely, it contains higher quantities of essential fatty acids, resulting in an improved lipid profile. This composition has been associated with lower blood cholesterol levels, which can contribute to better cardiovascular health. Furthermore, camel milk is rich in phospholipids, particularly plasmalogens and sphingomyelin. These components are essential for meeting the daily nutritional needs of both adults and children. The presence of these phospholipids suggests that camel milk fat can provide valuable nutritional benefits[25].
The cholesterol content in camel milk fat is approximately 34.5 mg per 100 g, whereas cow milk fat contains around 25.63 mg per 100 g. In terms of the size of fat globules, camel milk globules are smaller compared to cow's milk globules. The average size of camel milk fat globules is approximately 2.99 μm, which is comparable to the size of goat fat globules (around 3.19 μm). However, camel milk fat globules are significantly smaller than the fat globules found in buffalo milk, which have an average size of 8.7 μm. The smaller size of camel milk fat globules impacts the cream structure of the milk. Due to the smaller size, the cream formed in camel milk is generally lower in volume compared to other farm animals. It is worth noting that the structure of cream in camel milk can differ from that of milk from other animals, which can affect the processing and manufacturing of dairy products. It is important to keep in mind that these differences may vary slightly depending on factors such as breed and diet[8,24].
Khalesi et al.[26] support the finding that camel milk fat globules are the smallest compared to goat, cow, and buffalo milk. The reported average sizes for camel, goat, cow, and buffalo milk fat globules are 3.2–5.6 μm, 3.5–7.0 μm, 4.3–8.4 μm, and 5.6–13.0 μm, respectively. This indicates that camel milk has the smallest fat globules among these milk types.
Camel milk has relatively higher proportions of short-chain fatty acids (C4–C12) compared to those found in other mammals. It is worth noting that camel milk has higher levels of butyric acid (C4:0) and caproic acid (C6:0)[27]. However, long-chain fatty acids account for the majority of fats in camel milk, comprising 92%–99% of the total milk fat content. Camel milk also contains medium-chain fatty acids, including caprylic acid (C8:0), capric acid (C10:0), and lauric acid (C12:0). Palmitic acid (C16:0) makes up about 35% of the total milk fat, followed by myristic acid (C14:0) at 15% and stearic acid (C18:0) at 10%[25]. The fatty acid composition of camel milk demonstrates a good balance between saturated fatty acids (50%–65%) and unsaturated fatty acids (35%–50%), including fatty acids ranging from C18:1 to C18:3[4]. This balance indicates that camel milk contains both saturated and unsaturated fatty acids, which are important for various physiological functions in the body.
Overall, understanding the specific characteristics of camel milk fat opens up avenues for innovation in the dairy industry and provides individuals with alternative milk options that offer distinctive nutritional properties and potential health advantages.
Carbohydrates content
-
The lactose concentration in camel milk typically ranges from 2.4% to 5.8%, with an average of 4.1%. In comparison, cow milk has a lactose concentration ranging from 3.7% to 5.2%, which can vary depending on factors such as the volume of drinking water and the type of plant consumed[8]. Camel milk is considered a good source of oligosaccharides, which are complex carbohydrates. These oligosaccharides play a biological role as prebiotics in the body.
Camel milk contains prebiotics, specifically camel milk oligosaccharides, which promote the growth and activity of beneficial probiotic bacteria and inhibit the adhesion of pathogens to the colon's epithelial cells. This prebiotic effect is crucial for gut health and overall well-being[28]. Additionally, the oligosaccharide content of camel milk makes it a valuable source for producing functional foods that provide specific health benefits. When camel milk is pasteurized, it may contain various polysaccharides like 3'-galactosyl lactose, Lacto-N-neohexaose, 3'-sialyl lactose, 6'-sialyl lactose, sialyllacto-N-novopentaose A, and monosialyllacto-N-neohexaose. The presence of oligosaccharides and polysaccharides in camel milk indicates its potential as a beneficial functional food source[29].
Protein content
-
Casein is the major protein found in milk, including cow milk. However, camel milk differs in its protein composition, containing a wide variety of secondary whey proteins in addition to casein. Camel milk is known to contain secondary whey proteins such as serum albumin, acidic whey protein, peptidoglycan-recognizing protein, and various types of immunoglobulins[28]. These whey proteins contribute to the overall protein content and functional properties of camel milk.
The casein-to-whey protein ratio in camel milk differs from that of cow milk. Camel milk has a higher proportion of whey proteins compared to casein proteins. Typically, cow milk has a casein-to-whey protein ratio of approximately 80:20, whereas camel milk has a lower casein content and a higher proportion of whey proteins, resulting in a casein to whey protein ratio of around 40:60 or even lower. The specific casein-to-whey protein ratio in camel milk may vary depending on factors such as the breed of camel, lactation stage, and individual variations. However, in general, camel milk is known for its relatively higher whey protein content compared to cow's milk[22].
Casein content
-
Casein is the main protein in milk, and it contributes a significant portion of the milk protein content about 65%–70%[24]. The molecular weights of the different casein fractions can vary among different species. In camel milk, the casein fractions are composed of alpha casein, beta casein, and kappa casein, with molecular weights of approximately 35, 32, and 22 kDa, respectively. Comparatively, the molecular weights of alpha-casein and beta-casein in cow's milk are slightly lower, ranging from 22 to 27 kDa, and 24 kDa, respectively. The kappa casein molecular weight in cows is comparable to that of camels, at around 22 kDa. The proportions of casein fractions in camel milk differ from those in cow milk.
Table 2 displays the comparison of casein fractions between camel milk and cow milk. According to the table, camel milk typically contains β-casein about 65%, α-S1-casein 21%, and ҡ-casein 3.5%, while, cow casein consists of 36%, 38%, and 13% respectively[8,24]
Table 2. Comparison of casein fractions of camel and cow milk.
Parameters Camel milk Cow milk Ref. Total caseins % 65−70 80 [8,24] κ- casein (g/L) 0.1−2.4 4.39 β-casein (g/L) 5.5−29.0 32.7−39.0 α casein (g/L) 2.89 12.79 α-S1-casein (g/L) 2.4−10.3 3.80−3.97 α-S2-casein (g/L) 0.3−3.9 10.0−10.3 Micelle size (μm) 3.2−5.6 4.3−8.4 * Please note that the values mentioned in the table are approximate ranges and can vary depending on various factors such as breed, age, diet, and other environmental conditions. Additionally, camel milk's composition can vary significantly based on the region and season. These differences in the proportions of casein fractions between camel milk and cow milk contribute to variations in the protein composition and potential functional properties of the respective milk. Understanding the specific casein composition of camel milk is essential for various applications in the dairy industry and individuals seeking alternative sources of milk protein.
Whey protein content
-
Whey proteins are the second most abundant proteins in milk, comprising approximately 20% to 25% of the overall protein content, which corresponds to around 0.63% to 0.80% of the milk's total content. Camel milk contains a diverse range of whey proteins, including various bioactive functional molecular peptides and whey fractions, as indicated in Table 3[21]. Notable whey proteins found in camel milk include α-lactalbumin, immunoglobulins, peptidoglycan-recognition protein (PGRP), Ig enzymes, N-acetyl-β-glucosaminidase (NAGase), lactoferrin, lactoperoxidase, and lysozyme. Camel milk is particularly rich in lactoferrin and lysozyme, while it lacks β-lactoglobulin, which is abundant in whey proteins of cows, buffalos, and mares and constitutes 50% of their whey protein content[19]. Interestingly, α-lactalbumin, although the second most dominant protein in whey accounts for approximately 40% of the whey protein in camel milk[30].
Table 3. Comparison of bioactive whey fractions of camel and cow milk.
Parameters Camel milk Cow milk Ref. Total whey protein % 9.3–10 7.3–8 [9,
30,
33]Whey acidic protein (mg/L) 15.7–16 – β-lactoalbumin (mg/L) 3.5 1.2 Serum albumin (g/L) 0.46 0.36 Lactoferrin (mg/L) 200–728 80–500 Immunoglobulins (g/L) 1.5 0.5–1.0 Lysozyme (μg/100 mL) 228–500 7–37 Peptidoglycan recognition proteins (PGRPs) (mg/L) 370 – β-lactoglobulin (mg/L) – 1.3–3.5 α-lactalbumin (mg/L)) > 5.000 600–1,700 Immunoglobulins (kDa) Heavy chains Light chains Heavy chains Light chains IgG (whole molecule) 60 29 55 26 IgM 80 27 75 22.5 IgA 55.5 22.5 61 24 * Please note that the values mentioned in the table are approximate ranges and can vary depending on various factors such as breed, age, diet, and other environmental conditions. Additionally, camel milk's composition can vary significantly based on the region and season. Serum albumin is a globular protein that plays a role in maintaining osmotic pressure and transporting various substances in the bloodstream. Another significant whey protein found in camel milk is acidic whey protein, also known as alpha-lactalbumin. Alpha-lactalbumin is particularly rich in essential amino acids and has been extensively studied for its potential health benefits. Research suggests that it exhibits antimicrobial and anticancer properties, making it an interesting bioactive component of camel milk[31].
Peptidoglycan-recognizing protein is another whey protein present in camel milk. This protein plays a crucial role in the immune response, specifically in recognizing and responding to bacterial cell wall components. The presence of peptidoglycan-recognizing protein in camel milk suggests the potential for immune-modulating properties, further enhancing the potential health benefits associated with consuming camel milk[32]. In addition, to these whey proteins, camel milk contains various types of immunoglobulins, including IgG, IgA, and IgM. Immunoglobulins are antibodies that play a critical role in the immune system's defense against pathogens. The presence of immunoglobulins in camel milk indicates its potential as an immune-boosting food source, providing additional support for its beneficial effects on immune health[19].
Overall, the combination of serum albumin, acidic whey protein (alpha-lactalbumin), peptidoglycan-recognizing protein, and immunoglobulins in camel milk contributes to its unique nutritional profile and potential health benefits. These whey proteins highlight the milk's immune-boosting properties, antimicrobial effects, and potential as a functional food source. The diverse range of secondary whey proteins in camel milk provides additional nutritional and functional benefits compared to other types of milk. These proteins contribute to the unique properties of camel milk and may have implications for human health and well-being.
Table 3 presents the bioactive whey fractions of camel and cow milk. It is noted that camel and cow whey proteins exhibit slight differences in their structures, which can impact their functional properties[9,30,33]. However, camel whey protein content is higher than that of buffalo, sheep, goat, and cow milk since it was 0.80%, 0.68%, 0.66%, 0.53%, and 0.47%, respectively. However, the whey protein content in camel milk is still lower than that of human, donkey, and horse milk[34].
Camel milk is a great source of whey protein, with a higher protein content than other animal milk. This indicates its potential to provide better nutritional benefits. Also, the differences in whey protein content and composition between camel and cow's milk highlight variations in their bioactive whey fractions. These bioactive whey fractions contribute to the potential health benefits associated with consuming camel milk, which may differ from those found in cow milk. The unique composition of camel milk, with its higher whey protein content and distinct bioactive whey fractions, sets it apart as a promising source of beneficial proteins. This indicates that camel milk may offer unique capabilities and health-promoting properties compared to cow milk[34].
Researchers can explore the specific bioactive whey fractions and understand the protein content and composition variations to investigate the potential health benefits of consuming camel milk. These findings contribute to the growing recognition of camel milk as a valuable nutritional resource with potential physiological and therapeutic implications[17].
Table 4 compares the amino acid composition of camel and cow milk. Both essential and non-essential amino acids are found in the whey proteins of both camel and cow milk. These amino acids are important for various physiological functions in the body. In particular, branched-chain amino acids (BCAAs) such as leucine, isoleucine, and valine, which are vital for the functional properties of muscles, are present in camel whey proteins. These BCAAs play a crucial role in muscle protein synthesis and maintenance[34].
Table 4. Amino acids content of camel and cow milk.
Parameters (g/100 g) Camel milk Cow milk Ref. Histidine 2.9 1.4 [19,34] Serine 2.2 5.9 Cysteine 0.3 − Arginine 10.6 3.9 Glycine 2.2 2.3 Aspartic acid 10.2 10.7 Glutamic acid 29.2 7.4 Threonine 8 2.7 Alanine 3.1 5.1 Praline 17.8 14 Lysine 10 4.7 Tyrosine 0.9 0.3 Methionine 3.7 1.1 Valine 10.3 8.1 * Please note that the values mentioned in the table are approximate ranges and can vary depending on various factors such as breed, age, diet, and other environmental conditions. Additionally, camel milk's composition can vary significantly based on the region and season. El-Agamy et al.[12] reports that camel milk contains higher levels of amino acids with carbon chains ranging from C14:0 to C18:0. These amino acids are typically associated with longer-chain fatty acids. On the other hand, camel milk has lower levels of amino acids with carbon chains ranging from C4:0 to C12:0, which are associated with shorter-chain fatty acids. The unique amino acid composition of camel milk, with higher levels of certain amino acids and lower levels of others, contributes to its distinct nutritional profile. These variations in amino acid content may have implications for the functional properties and potential health benefits associated with consuming camel milk.
Several reports have investigated the immune properties of whey protein, demonstrating its advantages in enhancing the host's primary immune system. Whey protein has been shown to have immunomodulatory effects, supporting various immune functions such as lymphocyte activation and proliferation, antibody production, phagocytic activity, and cytokine secretion, including IL-1β, IL-6, TNF-α, and NF-κB. Furthermore, it possesses a range of functional properties. Studies have revealed that camel serum proteins can inhibit the production of reactive oxygen species (ROS) in the liver and other tissues of mice by stimulating the production of glutathione (GSH), which is vital for cellular defence[30].
These proteins have also been shown to increase antioxidant activity, which can combat fatigue and inflammation, promote healing, enhance resilience, boost immunity, and alleviate complications of type 2 diabetes. Additionally, camel serum proteins have demonstrated the ability to prevent metabolic syndrome, including hyperglycemia, elevated blood lipids, insulin resistance, and activation of satiety mechanisms[17]. Furthermore, camel serum proteins exhibit immunomodulatory, antiviral, and antibacterial effects. Their bioactive compounds play a role in controlling various immune functions, as well as the secretion of cytokines involved in immune regulation and inflammation[14].
Overall, the immune properties and functional benefits associated with whey protein and camel serum proteins highlight their potential as bioactive components with significant implications for immune health, inflammation control, and various physiological processes. These findings contribute to the growing understanding of the immunomodulatory and protective effects of these proteins, emphasizing their potential as functional ingredients or therapeutic agents.
Minerals content
-
Minerals play a crucial role in the physical condition and stability of milk proteins, primarily through the maintenance of salt balance. Minerals such as calcium (Ca2+), magnesium (Mg2+), phosphate (P3–), and citrate (Cit3–) contribute to this salt balance, which can be influenced by factors such as diet, lactation, and overall animal health. According to Table 5, the proportions of major minerals in camel milk appear to be similar to those in cow milk. This suggests that camel milk can be a valuable source of essential minerals. For instance, consuming one liter of camel milk has been reported to provide 100% of the daily human needs for calcium and phosphorus, 57.6% of potassium, and 40% of iron, copper, zinc, and magnesium. Additionally, it can contribute to 24% of the daily sodium requirements[19,33]. These findings highlight the nutrient richness of camel milk, particularly in terms of its mineral content. The presence of adequate amounts of calcium, phosphorus, potassium, iron, copper, zinc, magnesium, and sodium in camel milk suggests its potential as a valuable source of these essential minerals. Consuming camel milk can contribute to meeting the daily mineral requirements of individuals.
Table 5. The mineral content of camel and cow milk.
Parameters Camel milk Cow milk Ref. Iron (mg/L) 1.3–2.5 0.3–0.8 [19,33] Calcium (mg/100 g) 100–160 100–140 Phosphate (mg/L) 580–1,040 650–1,100 Potassium (mg/L) 600–2,100 1,350–1,550 Zinc (g/L) 4.5–5 3.5–5.5 Copper (g/L) 1.3–1.8 0.1–0.2 Magnesium (mg/L) 75–160 100–150 Manganese (mg/L) 0.08–0.2 0.04–0.2 Sodium (mg/L) 360–620 350–600 * Please note that the values mentioned in the table are approximate ranges and can vary depending on various factors such as breed, age, diet, and other environmental conditions. Additionally, camel milk's composition can vary significantly based on the region and season. The calcium:phosphorus ratio is an important consideration, and it differs among various types of milk. In camel milk, the calcium:phosphorus ratio is approximately 1.5, whereas it is 1.29 for cow's milk and 2.1 for human milk. This ratio is crucial as a formula based on cow milk used for infant feeding often contains high levels of phosphate, which can lead to low serum calcium levels and hyperphosphatemia in infants[35]. The calcium:phosphorus ratio in camel milk is closer to that of human milk, suggesting that it may be a more suitable alternative for infant nutrition.
Camel milk contains an adequate portion of bio-vital microelements, including manganese, iron, copper, and zinc. The concentrations of these microelements in camel milk range from 0.2 to 1.9 mg/L for manganese, 0.0 to 3.7 mg/L for iron, 0.11 to 1.5 mg/L for copper, and 2.8 to 4.4 mg/L for zinc. In comparison, cow's milk typically contains lower levels, with concentrations ranging from 0.04 to 0.20 mg/L for copper, 0.3 to 0.8 mg/L for cobalt, and 0.1 to 0.2 mg/L for molybdenum[19].
These differences in microelement content highlight the potential nutritional advantages of consuming camel milk. The higher concentrations of manganese, iron, copper, and zinc in camel milk suggests that it may provide a more significant contribution to meeting the daily requirements of these essential microelements compared to cow's milk. It's important to note that the specific concentrations of microelements can vary depending on factors such as the camel's diet and environmental conditions. Therefore, it's advisable to refer to scientific research papers or consult with experts in the field to obtain the most accurate and up-to-date information on the microelement content of camel milk.
Iron is a vital mineral that plays a crucial role in various biological processes, including DNA synthesis, oxygen transport, and storage. Adequate iron intake is essential for preventing anemia symptoms and promoting healthy growth in children[8]. Camel milk, which contains higher levels of iron compared to cow's milk, can provide an additional source of this important mineral, potentially contributing to improved iron status and overall health. In contrast to cow's milk, camel milk has higher chloride levels. This difference is attributed to the intake of herbal plants by camels. The higher chloride content in camel milk can be beneficial for certain individuals who require increased chloride intake for various physiological processes. Additionally, the citrate concentration in camel milk is lower (128 mg/dL) compared to cow's milk (160 mg/dL). This lower citrate level in camel milk can have exceptional benefits as it improves the antimicrobial activity of lactoferrin[19].
These findings emphasize the unique composition of camel milk, including its higher iron levels, elevated chloride content, and lower citrate concentration. These differences can have implications for specific biological processes, antimicrobial activity, and potential health benefits. However, it's important to note that individual nutritional requirements may vary, and further research is needed to fully understand the impact of these differences on human health.
Vitamins content
-
The vitamin content of cow milk and camel milk are compared in Table 6. When comparing the two types of milk, it is evident that camel milk is lacking in vitamins A, E, and B1 when compared to cow milk. The levels of these vitamins in camel milk are significantly lower than those found in cow milk. Riboflavin, folic acid, and pantothenic acid content in camel milk are approximately 4, 3, and 4 times less than that of cow's milk, respectively[36]. On the other hand, camel milk exhibits higher levels of certain vitamins when compared to cow's milk. Vitamin C content in camel milk is approximately 2 to 5 times higher than that found in cow milk. Additionally, camel milk contains five times more niacin than cow milk. However, it is worth noting that the β-carotene content in camel milk is insufficient[19,37].
Table 6. The vitamins content of camel and cow milk.
Parameters Camel milk Cow milk Ref. β- Carotene (μg %) − 99.60 [19,37] Vitamin A (mg/L) 0.1–0.15 0.17–0.38 Vitamin D (mg/L) ND 0.5 Vitamin E (mg/L) 0.53 0.2–1.0 Vitamin B1 (mg/L) 0.33–0.6 0.28–0.9 Niacin (mg/L) 4.60 0.5–0.8 Vitamin C (mg/L) 24–36 3–23 Pantothenic acid (mg/L) 0.88 2.6–4.9 Vitamin B12 (mg/L) 0.002 0.002–0.007 Folic acid (mg/L) 0.004 0.01–0.1 Vitamin B2 (mg/L) 0.42–0.8 1.2–2 Vitamin B6 (mg/L) 0.52 0.4–0.63 * Please note that the values mentioned in the table are approximate ranges and can vary depending on various factors such as breed, age, diet, and other environmental conditions. Additionally, camel milk's composition can vary significantly based on the region and season. Consuming 250 mL of camel milk per day can contribute to fulfilling several daily micronutrient and vitamin requirements. Specifically, camel milk provides approximately 15.5% of the recommended daily intake of cobalamin (vitamin B12), 8.25% of riboflavin (vitamin B2), 5.25% of retinol (vitamin A), 10.5% of ascorbic acid (vitamin C), as well as thiamine (vitamin B1) and pyridoxine (vitamin B6). In comparison, consuming the same volume of cow milk (250 mL) covers a different set of daily requirements for consumers. Cow milk can provide approximately 43.5% of the recommended daily intake of cobalamin (vitamin B12), 36% of riboflavin (vitamin B2), 11.5% of pyridoxine (vitamin B6), 3.5% of ascorbic acid (vitamin C), as well as 9% of retinol (vitamin A) and thiamine (vitamin B1). These percentages indicate the proportion of the recommended daily intake that can be met by consuming 250 mL of camel milk or cow milk[38].
-
Bioactive proteins and peptides offer a diverse array of applications, spanning from antimicrobial food additives to functional foods, nutraceuticals, and nutritional supplements. The presence of bioactive proteins in milk provides opportunities for the development of functional products due to their balanced amino acid composition[39]. Camel milk, in particular, contains various protein fractions that contribute to its bioactive properties. These include lactoferrin, lysozyme, lactoperoxidase, and different types of immunoglobulins. Each of these protein fractions supports a wide range of biological functions, further highlighting the potential health benefits of camel milk[40].
The presence of these bioactive proteins and peptides in camel milk opens doors for their utilization in various sectors, such as the development of antimicrobial food additives, functional foods with enhanced health benefits, the production of nutraceuticals, and the formulation of nutritional supplements. Their diverse functionalities and biological activities make them valuable components in promoting human health and well-being.
Lactoperoxidase
-
Lactoperoxidase is an oxidizing enzyme that is commonly found in exocrine secretions such as milk, saliva, and tears. In the case of camel milk, it has been reported to have a lactoperoxidase content of approximately 2.23 units/mL, while cow milk typically contains around 30 mg/L. The variations in lactoperoxidase content in camel milk can be attributed to factors such as feeding, calving, breeding season, environmental conditions, and other physiological factors that affect milk composition[21].
Several studies have shown that the presence of lactoperoxidase in camel milk and colostrum can contribute to the reduction of bacteria such as Listeria monocytogenes and Escherichia coli. When camel milk and colostrum were inoculated with these bacteria and stored at temperatures ranging from 4 to 20 °C for 2 d, the lactoperoxidase/thiocyanate/hydrogen peroxide and lysozyme system exhibited antimicrobial activity, leading to a decrease in bacterial counts. This antimicrobial capability was attributed to the mode of action of the lactoperoxidase system[41]. Furthermore, Al-Baarri et al.[42] demonstrated the anti-tumor effects of lactoperoxidase. These findings highlight the significant role of lactoperoxidase in camel milk, contributing to its antimicrobial properties and suggesting potential health benefits, including anti-tumor activity.
Lysosome
-
Lysozyme, an antimicrobial component, is found in higher concentrations in camel milk compared to cow, buffalo, sheep, and goat milk. However, it is less prevalent in human milk, donkey milk, and mare's milk. The concentration of lysozyme in camel milk ranges from 228 to 500 μg/100 mL, while in cow milk, it ranges from 7 to 37 μg/100 mL. Mare's milk contains approximately 79 μg/100 mL, which is higher than other livestock, and buffalo milk contains approximately 13 μg/100 mL. In contrast, human milk contains about 10 μg/100 mL, and this difference may be attributed to the impact of the lactation period[12].
Immunoassay investigations have revealed that there are no antigen structural similarities between lysozyme formulations in cow milk and camel milk[19]. Understanding the variations in lysozyme content in different types of milk can contribute to the development of functional foods and nutraceuticals with enhanced antimicrobial properties and potential health benefits. Further research on the specific properties and applications of lysozyme in camel milk could provide valuable insights into its functional role and potential uses in various industries.
The higher concentration of lysozyme in camel milk suggests its potential as a natural antimicrobial agent. Lysozyme exhibits antimicrobial activity by disrupting the cell walls of bacteria, thereby inhibiting their growth. The variations in lysozyme concentrations among different types of milk highlight the diverse composition and functional properties of these dairy products[12].
Lactalbumin and lactoglobulin
-
The main whey protein found in camel milk is α-lactalbumin, with an average concentration of 2.2 g/L, which is equivalent to 2.45 g/L in human milk. In human milk, α-lactalbumin accounts for about 36% of whey proteins and approximately 3.5% of total protein content.
The composition of whey proteins in camel milk and cow milk is comparable, but camel whey protein is deficient in β-lactoglobulin and has the highest concentration of α-lactalbumin compared to cow whey proteins[43]. α-lactalbumin in camel milk consists of 123 amino acid residues and has a molecular weight of approximately 14.43 kDa. It contains the same number of cysteine residues at the same positions and the same number of disulfide bonds as other α-lactalbumin proteins (Cys6/Cys120, Cys28/Cys111, Cys61/Cys77, and Cys73/Cys91). However, camel α-lactalbumin is more sensitive to calcium deficiency compared to cow α-lactalbumin[44].
The biological functions of α-lactalbumin include the regulation of lactose synthesis, antioxidant activity due to its abundance of essential amino acids, and its role in improving intestinal health, immune responses, and the absorption of vital trace elements such as iron and zinc[45].
The presence of α-lactalbumin in camel milk suggests its potential as a functional ingredient in various applications, including the development of nutritional supplements, functional foods, and nutraceuticals. Its biological functions and nutritional properties make it a valuable component in promoting intestinal health, supporting immune function, and enhancing nutrient absorption.
Lactoferrin
-
Lactoferrin is an 80 kDa glycoprotein that binds non-heme iron and is found naturally in mammary gland secretions and other exocrine fluids. Its levels in milk can vary due to factors like genetics, environment, and breed[46]. Lactoferrin belongs to the transferrin family and has gained attention in academic, pharmaceutical, and commercial applications. It is consumed as a dietary supplement, particularly in infant nutrition and immune system development[47].
In terms of lactoferrin content, human milk has the highest concentration at approximately 1,700 mg/L. Comparatively, camel milk ranges from 200 to 728 mg/L, cow milk ranges from 80 to 500 mg/L and donkey milk has the lowest lactoferrin content at around 79 mg/L[48]. Compared to other ruminants, camel milk contains higher levels of both lysozyme and lactoferrin. In particular, iron-saturated lactoferrin has been found to impede the multiplication of microbes in the digestive tract. These results reinforce the potential use of camel milk and its components, including lactoferrin, as functional ingredients in various applications, such as the development of antimicrobial agents and food products[49].
Lactoferrin exhibits antimicrobial, anti-inflammatory, and immunomodulatory activities, making it important for immune defense, iron transport, and gut health. Due to its diverse functions and potential health benefits, lactoferrin has garnered interest from researchers and industries[19].
Lactoferrin functions by sequestering iron, which is an essential nutrient for bacterial growth. This action prevents bacterial proliferation. On the other hand, transferrin is responsible for transporting iron in the bloodstream and delivering it to cells that require it for metabolic processes. It has a high affinity for iron and transports it to specific target cells through transferrin receptors. This process helps to maintain iron balance in the body[50].
Camel lactoferrin has been extensively studied for its antifungal properties. A particular study conducted by Liao et al.[51] focused on evaluating the inhibitory effects of camel lactoferrin on fungi known to cause human infections, such as Candida albicans, Aspergillus fumigatus, and Cryptococcus neoformans. The results indicated that camel lactoferrin effectively inhibited the growth of these fungal strains by damaging their cell membranes, leading to cellular damage and hindering fungal proliferation. Overall, lactoferrin's ability to sequester iron and its antifungal properties, especially in camel lactoferrin, provide potential applications in combating bacterial growth and fungal infections.
Camel lactoferrin demonstrates strong antibacterial and bactericidal effects against a wide range of bacteria. It effectively inhibits the growth of various organisms, including Listeria monocytogenes, Salmonella spp., Escherichia coli, Bacillus stearothermophilus, Shigella dysenteriae, Bacillus subtilis, Pseudomonas aeruginosa, Streptococcus aureus, Klebsiella pneumoniae, Clostridium spp., and Helicobacter pylori. These findings highlight the significant antimicrobial properties of camel lactoferrin in preventing the growth and spread of various pathogens[14,41].
Conesa et al.[52] conducted a study to purify lactoferrin from the milk of various animals, including camel, alpaca, sheep, goat, elephant, grey seal, and humans. Lactoferrin was found in all milk samples except for the grey seals. Human lactoferrin exhibited the highest heat resistance, while lactoferrins from other animals showed varying degrees of thermo-resistance, with elephant lactoferrin being the least resistant. The antimicrobial activity of the isolated lactoferrins against Escherichia coli 0157:H7 was also investigated, revealing that camel lactoferrin was the most effective against this bacterium, while alpaca and human lactoferrins were the least effective.
Lactoferrin's antimicrobial properties have led to its application as a natural preservative in various products, including dairy, meat, seafood, beverages, and bakery items. By inhibiting the growth of spoilage and pathogenic bacteria, lactoferrin helps extend the shelf life of these products and ensures their safety for consumption. Its use in bio-preservation can enhance food quality, reduce reliance on synthetic preservatives, and potentially offer health benefits. Furthermore, lactoferrin's antimicrobial activity has implications beyond food preservation. It has been studied for its potential in combating life-threatening diseases such as cancer, hepatitis, respiratory infections, and foodborne illnesses in individuals of all age groups. By inhibiting the growth of pathogenic bacteria, lactoferrin may contribute to the prevention and treatment of these infections[39].
Habib et al.[53] assessed lactoferrin's antioxidant functions against the spread of human colon cancer (HCT-116) and its DNA destruction in vitro. Moreover, it has anti-inflammatory effects and immune functions that modulate various immune cells' maturation, including neutrophils, macrophages, and lymphocytes[50]. Moreover, camel lactoferrin has an anti-arthritic effect, demonstrating anti-inflammatory activity against interleukin-1 and activation of cartilage gaps in human osteoarthritis by limiting kappa B signaling events and nuclear factors. Camel milk proteins were also demonstrated to suppress cyclooxygenase-2 expression and prostaglandin E2 synthesis in activated human osteoarthritis chondrocytes[54]. Lactoferrin has been extensively researched due to its potential as an anticancer agent. Studies have demonstrated its ability to inhibit the growth of cancer cells and induce apoptosis in various types of cancer, including breast, colon, and lung cancer. This suggests that lactoferrin holds promise as a therapeutic intervention in cancer treatment[55].
The therapeutic potential of lysozyme, lactoferrin, and lactoperoxidase found in camel milk has been recognized for their significant role in managing various inflammatory diseases. Studies have shown that camel milk antibodies can regulate the immune system and provide therapeutic effects in the context of inflammatory diseases. Therefore, Kocyigit et al.[56] have highlighted the potential of camel and mare milk and their derived products in treating inflammatory rheumatic disorders. They specifically focused on the anti-inflammatory and immune-regulatory capabilities of these types of milk that do not come from cows. Investigating innovative strategies to regulate and reduce inflammatory conditions using these alternative types of milk can offer potential avenues for future research.
Lactoferrin's antiviral action against a wide range of DNA and RNA viruses, either by direct virus surface attachment or by blocking virus cell receptors. Common viruses targeted by lactoferrin include HSV-1, human papillomavirus, human immunodeficiency virus (HIV), rotavirus[57], hepatitis B, C, and CMV[46,58].
Recently, there has been increased interest in exploring the potential role of lactoferrin, or lactotransferrin, in suppressing the coronavirus family, particularly during the COVID-19 pandemic. Researchers suggest that lactoferrin may enhance the host's immune response and improve the effectiveness of the immune system against coronaviruses.
Lactoferrin interacts with Heparan Sulfate Proteoglycans (HSPGs) receptors on the surface of host cells, which are involved in virus attachment. By binding to HSPGs, lactoferrin can interfere with the attachment of coronaviruses, including SARS-CoV-2, to host cells, thereby preventing viral entry and infection. Additionally, lactoferrin interacts with the angiotensin-converting enzyme 2 (ACE2), the receptor utilized by coronaviruses to enter host cells. Binding to ACE2, lactoferrin competitively inhibits the virus-receptor interaction, reducing viral entry and subsequent infection[59]. Another antiviral mechanism of lactoferrin is its ability to sequester iron. Coronaviruses, including SARS-CoV-2, require iron for replication and survival. By sequestering iron, lactoferrin limits the availability of this crucial nutrient, potentially inhibiting viral replication. Furthermore, lactoferrin plays a role in modulating the immune response by inducing the production of inflammatory chemicals during the cytokine storm. This helps regulate the immune response, preventing excessive inflammation often associated with severe COVID-19 cases[60].
Immunoglobulins and antibodies
-
Immunoglobulins (Igs), often known as antibodies, are polypeptide chains with a large molecular weight that play an important role in safeguarding immune systems against various infections[61]. Both camel and most placental mammals have immunoglobulins that have been grouped into five classes: IgM, IgG, IgA, IgD, and IgE isotypes[62]. Camel milk has the highest immunoglobulin amounts of about 2,000 mg/L, followed by buffalo and cow 460–1,300 and 100–800 mg/L, respectively, while the lowest in human milk was 40–54 mg/L[63].
Members of the Camelidae family, particularly camels, and llamas, generate conventional antibodies that are substantially smaller in size than those produced by humans or cows. When appropriately designed, variable antigen-binding domains formed from these antibodies are referred to as 'nano-bodies,' which have a high specificity and affinity and are more stable than standard antibodies[64].
Camel milk immunoglobulin is quite significant in terms of immunotherapy and has a unique heavy-chain antibody (HCAbs) homodimer that can be skillfully engineered and highly soluble and stable in high temperatures, whereas the light chain (L-chains) is missing[65].
Recently, antibodies have been highly advanced in clinical trials, providing significant benefits in oncology, autoimmune systems, infectious and neurological disorders, and cancer monitoring. Hence, heavy-chain camel immunoglobulin is widely used for immunotherapy in patients with multiple diseases and this research effort will lead to the development of a whole new family of vaccines[66]. Also, HIV/AIDS, or hepatitis C, effective anti-diabetes and prophylactic heart disease[63], stimulates and maintains the gastrointestinal tract's immune system, prevents the proliferation of cancer cells, antihistamines, and reduces tuberculosis symptoms[67].
Immunoglobulins, play a pivotal role for their potential therapeutic applications, including their role in immune modulation and their ability to target specific antigens in diseases such as cancer. Additionally, immunoglobulins, specifically immunoglobulin G (IgG), have been shown to possess anti-inflammatory effects by binding to inflammatory mediators and inhibiting their activity[68].
-
Camel milk and human milk share significant similarities in terms of their composition, particularly when it comes to important antimicrobial proteins like lactoferrin and lysozyme. Additionally, both milks contain a superior immunoglobulin cameloid (Ig) with a spatial structure similar to human immunoglobulin[69].
Shukla et al.[70] reported that camel milk possesses a range of potential health benefits due to the presence of specific essential active peptides. These benefits include antimicrobial, antioxidant, antihypertensive, anti-inflammatory, and immunomodulatory properties. Numerous physical and microbial studies have shown that camel milk ingredients are of considerable nutritional and medicinal value due to their abundance of antibacterial and antiviral compounds. Moreover, camel milk provides essential physiological and therapeutic nutritional benefits that enhance immune function[13]. These findings underscore the potential of camel milk as a valuable source of functional and therapeutic components, offering a wide range of health benefits. Further research in this field can contribute to the development of innovative applications for camel milk and its constituents in promoting human health and well-being.
Alhaj et al.[71] reported, the effects of fermented camel milk using Lactobacillus acidophilus, Lactobacillus helveticus, and Streptococcus thermophiles were compared to unfermented camel milk. The study examined the changes in microbial prevalence after two weeks of storage. The results indicated that unfermented camel milk exhibited a significant reduction in the prevalence of Bacillus cereus, Salmonella Typhimurium, and Staphylococcus aureus when compared to fermented camel milk.
Kumar et al.[72] investigated the activity of hydrolyzed and unhydrolyzed casein on several types of bacteria. Gram-negative bacteria such as Pseudomonas aeruginosa ATCC 15742 and Gram-positive bacteria such as Bacillus cereus ATCC 11778, Listeria innocua LRGIA 01, and Escherichia coli XL1 blue were significantly less common in camel milk or colostrum after being hydrolyzed in the gastrointestinal tract by digestive enzymes containing biologically active antibacterial peptides. Moreover, they suggested that specific protease enzymes, such as chymotrypsin, which may improve polypeptide casein, may be responsible for camel milk's antibacterial effect. Also, they demonstrated that the protein chain size of 2–20 amino acids is optimal for antimicrobial targets since the breakdown of various proteins in camel serum enhances the antibacterial activity.
Camel milk contains immunological compounds known as nano-antibodies, also referred to as single-domain antibodies or VHH antibodies. These nano-antibodies are unique to camelids, including camels and llamas, and possess distinct functional properties compared to conventional antibodies found in other mammals. Antimicrobial activity is affected by the size of the hydrolysis fragment; as tiny immunoglobulins can penetrate the microbial lipid membrane. It also serves to enhance the immune system by attacking only harmful antigens and increasing ion leakage and other cell content, thus disrupting cell activity[73].
Owing to their small size, which permits deep tissue penetration and particular antigens to pass through barriers like the blood-brain barrier and bind intracellular antigens, camel milk antibodies can show a virus-attacking potential to penetrate target areas[74]. Due to their capacity to neutralize enzymes and their efficacy against several types of viruses and bacterial diseases, 'nano-bodies' from camel and llama are much more beneficial than human antibodies, despite being just one-tenth the size of a conventional antibody (12–15 kDa)[75]. In the same context, Homodimeric Heavy Chain (VHH) can be used to diagnose and treat various bacterial zoonotic diseases such as Campylobacterosis, Escherichia coli, Listeriosis, and Anthrax. As well as, parasitic diseases such as Taeniasis, Trypanosomiasis. Also, viral zoonotic diseases such as Influenza A viruses (IAV), H1N1 and, H5N1 viruses, Rabies, and Foot-and-mouth disease (FMD)[65].
Many studies have proven how zoonotic viruses spread, particularly those belonging to the coronavirus family. The family of extremely contagious beta coronaviruses, which includes SARS-CoV-1, SARS-CoV-2, and MERS-CoV, is one of the zoonotic diseases that has contributed to the devastating pandemic in humans[76]. The first molecular strategy for preventing the spread of coronavirus family outbreaks was developed as a consequence of the coating protein that encases positive-sense RNA viruses. Coronavirus Spike (S), a large RNA-covering protein capsule, has been shown to activate host cell receptors and generate a fusion membrane. This research opens the door for the creation of a therapeutic bio-drug to combat these viruses. In addition, the researchers found isolated single-domain antibodies (VHHs) from a well-known 'llama'. These antibodies have the same size as immunoglobulins and can cross cell membranes. It was vaccinated with coronavirus spikes that had been prefusion-stabilized. These VHHs are capable of neutralizing pseudotyped MERS-CoV or SARS-CoV-1 S viruses. On the other hand, VHH crystal structures can disrupt receptor binding. The findings revealed the existence of a cross-reactive VHH as a bivalent human fusion IgG Fc that neutralizes the pseudotyped SARS-CoV-2 S[77].
Because of their accessible biophysical properties, several VHHs have been explored as possible therapies for a variety of respiratory infections, including respiratory syncytial virus (RSV). VHHs should not be regarded as a therapeutic therapy for the current COVID-19 outbreak since further research is needed. Even though VHH has high stability after lengthy storage as a therapeutic option, the inhaler will release it and deliver it directly to the infection site during a pandemic outbreak[78].
For almost two decades, the Hepatitis C virus has been spreading around the world. Although our understanding of the virus has vastly increased, and progress in the creation of medication has been achieved. Nevertheless, no viable vaccination exists to prevent new infections. While camel milk has been certified for medicinal use, traditional and folk medicines are commonly used in rural and nomadic areas. In addition, when compared to human milk, lactoferrin, and IgG camel milk are responsible for avoiding hepatitis HCV infection. Furthermore, camel milk enhances the patient's cellular immune response suppresses viral DNA replication, and promotes the recovery of chronic hepatitis C patients[13].
Briefly, camel milk contains immunological compounds known as nano-antibodies, which have critical functional properties relative to other mammalian varieties, such as the ability to tolerate various malignancies and tumor types. Camelids, particularly camels and llamas have recently demonstrated the ability of nanoparticles to combat a wide range of viruses, including the Hepatic and Coronary virus families.
Hypoallergenic properties
-
In situations where breastfeeding is not feasible or presents difficulties, parents can explore alternative options such as plant-based substitutes like soy extracts or animal alternatives like sheep and goat milk mixed with cow milk proteins. However, it's important to keep in mind that such alternatives may cause hypoallergenic issues in some infants[79]. To counter this problem, researchers have been focusing on finding alternative sources of breast milk that can provide a suitable and hypoallergenic option for infants. In this context, camel milk is emerging as a promising substitute for cow's milk because of the Camelidae family's large evolutionary distance from the Bovidae family, as well as its high nutritional value and therapeutic properties. The protein composition of camel milk differs from that of cow's milk formulas, which increases the likelihood of eliminating allergic reactions associated with cow's milk-based infant formula[80].
Camel milk's unique protein profile, which includes different types of proteins compared to cow's milk, may offer advantages for infants with cow's milk allergies or intolerances. The specific protein composition of camel milk, along with its naturally occurring bioactive compounds, may contribute to its hypoallergenic properties and potential for reducing allergic reactions in susceptible individuals[19]. In this scenario, lower casomorphin synthesis in camel milk plays a key role in improving intestinal motility. Casomorphins are peptide fragments derived from the breakdown of casein proteins, and their presence in cow's milk has been associated with slower intestinal transit time. In contrast, the lower levels of casomorphins in camel milk facilitate faster transit through the intestines, allowing lactose to be more efficiently broken down by the enzyme lactase. This, in turn, reduces the symptoms of lactose intolerance, such as bloating, cramps, and diarrhea. Furthermore, the differences in protein composition between camel milk and cow's milk contribute to reduced allergic responses in individuals with lactose intolerance. β-casein, a major protein in cow's milk, is present in lower concentrations in camel milk. β-lactoglobulin, another protein commonly associated with allergies, is also found in lower levels in camel milk. These diminished levels of allergenic proteins in camel milk decrease the likelihood of triggering allergic reactions and adverse responses in individuals with lactose intolerance. These findings suggest that camel milk offers distinct advantages for individuals with lactose intolerance compared to cow's milk[81].
Anti-autism properties
-
Over the past two decades, the prevalence of autism has increased dramatically and its most serious manifestations include developmental dysfunction. It can also be associated with higher incidences of mental retardation, and autoimmune, and gastrointestinal disorders. Autism has also been linked to immunological, oxidative stress, environmental, neurochemical, and genetic variables[82].
Al-Ayadhi & Elamin[83] revealed that autism symptoms such as sleep disturbances, greater visual and linguistic perception, improved digestive processes, and heightened awareness of surrounding settings were reduced in autistic children who ingested camel milk. Children who have stopped ingesting camel milk may experience autistic symptoms again.
Hamzawy et al.[84] reported that children with autism exhibit elevated levels of plasma malondialdehyde, indicating an increase in oxidative stress. This occurs when the generation of free radicals exceed the cellular antioxidant capacity, leading to harmful effects such as lipid peroxidation, DNA damage, and enzyme inactivation.
However, camel milk has been found to possess therapeutic effectiveness in various conditions, including diabetes, autoimmune disorders like Crohn's disease and multiple sclerosis, and even autism. Its unique chemical composition has been shown to protect against carcinogens and hepatotoxic substances while reducing oxidative stress and inflammatory responses. This is achieved by modulating mitogen-activated protein kinase signaling pathways.
Given the interest in natural products and antioxidants for treating autistic behaviors, camel milk holds promise as a therapeutic option due to its unique composition and beneficial effects on oxidative stress and inflammation. Nonetheless, further research is necessary to explore the specific mechanisms behind camel milk's therapeutic properties and its potential as a complementary treatment for autism and related conditions.
Antidiabetic properties
-
Diabetes, which comprises type 1 and type 2, is an extremely hazardous disease that poses significant global implications for human health. If not managed effectively, it can give rise to serious medical complications. Cardiovascular diseases, renal failure, blindness, and lower-limb amputations are some of the severe consequences that can result from diabetes[85].
Numerous studies have demonstrated that camel milk has potential benefits in managing hyperglycemia in type 1 diabetes mellitus. This is attributed to the presence of insulin-like proteins in camel milk, which exhibit anticoagulant properties on digestive enzymes. As a result, camel milk can be used as an alternative therapy to reduce the required dosage of insulin in type 1 diabetes patients[17].
It's worth noting that the insulin levels in human milk are significantly higher, around 60.23 IU/L, compared to camel milk. However, the presence of clotting enzymes in the stomach prevents the availability of human milk insulin for intestinal absorption[86].
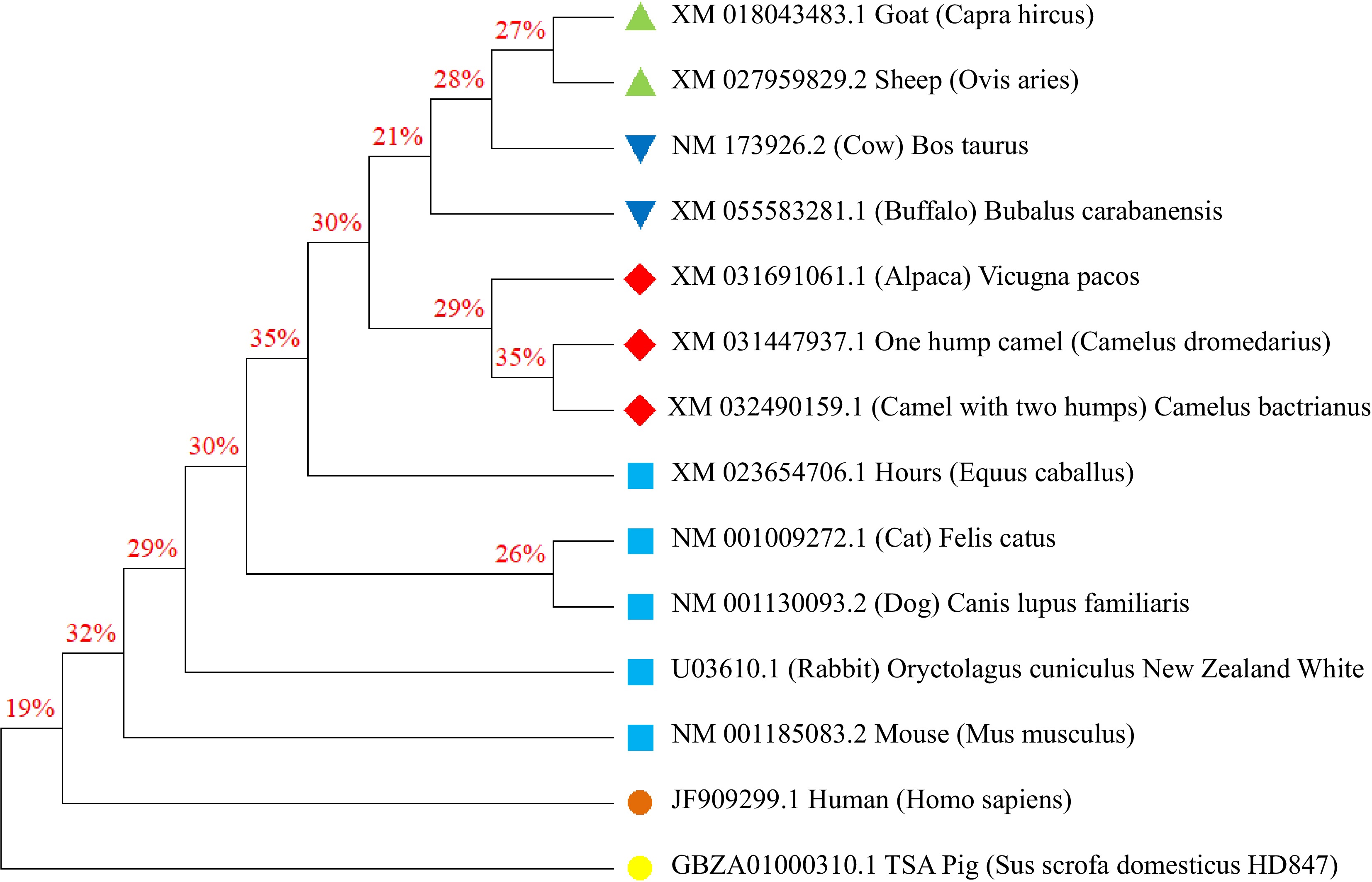
Figure 1 displays an average distance tree that shows the evolutionary relationship between insulin in different mammalian species. The analysis provides interesting insights into the evolutionary connections of insulin, particularly with camels and other animals. Both the one-humped camel (Camelus dromedarius) and the two-humped camel (Camelus bactrianus) exhibit a close affinity in terms of insulin evolution, indicating a strong interrelationship between them. Additionally, these camel species demonstrate a close evolutionary connection with other animals such as alpacas (Vicugna pacos), cows, and buffalo.
The evolutionary analysis further reveals that cattle, buffalo, sheep, and goats share a close evolutionary relationship with the camel group. On the other hand, cats and dogs form a separate and distinct group that is quite distant from the tree, showcasing a distinct evolutionary relationship with the camel group. Moreover, humans, pigs, mice, and rabbits exhibit independent cases that are significantly removed from the camel insulin phylogenetic tree. These findings emphasize the proximity in the composition of camel insulin to that of the bovine family, highlighting the similarities and shared evolutionary history between camels and certain animal species.
Previous studies have identified insulin-like proteins in camel milk that possess a cysteine chain similar to the insulin polypeptide family. Camel milk has a higher concentration of insulin compared to cow's milk, ranging between 40.50 and 58.67 IU/L, which is approximately three times that of cow's milk. The specific insulin concentration in camel milk can vary depending on the breed of the camel and the lactation season. Comparatively, buffalo milk contains 16.2 IU/L. In addition, camel milk is also rich in zinc, which plays a significant role in the functioning of β-cells. This mineral has implications for insulin release and regulation. Furthermore, camel milk insulin has been found to have notable effects on cardiovascular, renal, hepatic enzymes, and baseline proteins. These findings suggest that camel milk insulin may have broader impacts beyond glycemic control[87].
A further study was conducted by Mehaia et al.[88] who reported that camel milk is recognized for its higher content of insulin-like proteins, with approximately 52 micro unit/ml compared to cow milk's 16.32 micro unit/mL. These insulin-like proteins in camel milk exhibit similarities to insulin and can interact with its receptor, mimicking its action. Additionally, camel milk is rich in zinc, which plays a crucial role in the secretory activity of insulin in pancreatic beta cells.
Insulin can be administered through various routes, such as oral and nasal paths. However, these methods have not been successful due to insulin's susceptibility to enzymatic breakdown and poor mucosal absorption. Protein or peptide medications cannot cross the mucosal membrane barrier as they degrade before reaching circulation. The question arises, how does camel milk insulin enter the digestive tract without breaking down? To address this issue, novel tactics are required that focus on insulin production and excipients in the physical structure to preserve stability and increase insulin bioavailability[89].
Despite the closeness in the phylogenetic tree between camels and the bovine family, camels remain superior in the synthesis of insulin itself, which sees insulin protected from the effects of digestive enzymes. There are several theories on how camel milk insulin passes safely through the digestive tract without enzymatic breakdown: i) Camel milk insulin or insulin-like protein is biodegradable in the stomach and can be absorbed into the circulation of the intestine; ii) Camel milk insulin is encased in lipid nanoparticles (i.e. lipid vesicles) that will transfer it silently through the bloodstream; iii) The camel milk insulin hypothesis is identical to insulin at a lower molecular weight and mimics insulin interaction with its receptors[90].
Abdelazez et al.[17] conducted an in vivo study to investigate the effects of raw camel milk on hypoglycemic activity in diabetic mice. The study revealed several significant improvements associated with the consumption of camel milk. Firstly, the mice that received camel milk showed lower postprandial blood glucose levels, indicating improved glycemic control. Secondly, the mice treated with raw camel milk exhibited lower levels of blood hypolipidemia, suggesting potential benefits for maintaining healthier lipid profiles and reducing lipid-related complications in diabetes. Additionally, the mice treated with camel milk showed lower levels of liver enzymes, indicating a potential protective effect on liver health. These findings suggest that camel milk has therapeutic potential in improving hypoglycemia, lipid profiles, and liver function in diabetic mice.
Clinical trials have demonstrated the beneficial effects of raw camel milk on diabetes when administered at a specific dosage of 500 mL per day for three consecutive months. These studies have revealed highly significant reductions in HbA1c (glycated hemoglobin) levels, which indicate improved glycemic control. By lowering HbA1c levels, the risk of diabetes complications in diabetic patients is reduced, and the need for insulin dosage may decrease. Furthermore, research has indicated that camel milk can effectively decrease blood glucose levels, potentially aiding in the management of diabetes. It has also been found to reduce insulin resistance, which is a key factor in the development and progression of diabetes. Additionally, camel milk has shown promise in improving lipid profiles. By positively influencing lipid metabolism, it may contribute to reducing the risk of lipid-related complications associated with diabetes[90].
Agrawal et al.[91] studied two groups of diabetic patients, with all other variables, including dietary intake and daily activities, held constant. The first group, comprised of 12 patients (n = 12), received conventional daily insulin dosages, while the second group, also consisting of 12 patients (n = 12), was administered 500 mL of camel milk daily. Blood glucose levels were regularly monitored in both groups. The results showed that only three patients in the group receiving daily fresh camel milk reported a significant decrease in blood glucose levels from 118.58 to 3.16 mg/dL and a reduction in insulin dosages from 32.50 to 17.50 units/day. However, nine patients did not show such significant improvements.
Another study conducted by Kotb-El-Sayed et al.[92] involved 50 patients with type 1 diabetes. The patients were divided into three groups: the first group received oral insulin, the second group was given 500 mL of camel milk daily, and the third group received both insulin and 500 mL of camel milk. The study found that the group receiving both insulin and camel milk experienced a more substantial reduction in insulin demand, from 59.26 to 20.0 mg/dL, compared to the groups receiving only camel milk or insulin alone. However, it is important to note that fasting blood glucose, postprandial blood glucose and glycated hemoglobin levels increased significantly in all three groups. These studies suggest that camel milk may have the potential to reduce insulin dosages and improve glycemic control in some individuals with diabetes.
Briefly, studies have indicated that camel milk may have beneficial effects for individuals with diabetes. It has been found to lower blood glucose levels, reduce insulin resistance, and improve lipid profiles. Notably, raw camel milk has demonstrated greater effectiveness in managing blood glucose levels in mice and rats compared to other types of milk, including cow, buffalo, pasteurized camel milk, and camel milk products, as observed in both in vivo and clinical investigations. However, it is important to highlight that further research is necessary to gain a deeper understanding of how camel milk impacts different types and stages of diabetes. Additionally, more studies are needed to determine the optimal dosage of camel milk and assess its long-term effects on diabetes management.
Antitumor and anticarcinogenic activity
-
Abdel Gader & Alhaider[67] conducted research that highlights the potential of camel milk in preventing the spread of tumors and malignancies in various forms of cancer, including hepatitis and colon cancer. One of the mechanisms by which camel milk exerts its anti-cancer effects is through the induction of caspase 3 transcription, a process involved in programmed cell death. Moreover, camel milk has been found to possess antithrombotic activity, effectively inhibiting coagulation and fibrin formation. This antithrombotic property plays a role in impeding the proliferation and development of metastatic tumor cells. The presence of lactoperoxidase and peptidoglycan in camel milk is attributed to this beneficial effect. These compounds demonstrate anti-tumor properties that specifically target breast cancer and metastases.
Similarly, Krishnankutty et al.[10] emphasized that camel milk contains high levels of bioactive molecules, including lactoferrin, immunoglobulin (IgM), and iron-bound glycoprotein. These molecules have been shown to possess anti-cancer properties. Notably, they have demonstrated efficacy in destroying cancer cells (HepG2-MCF7) associated with tumor tissues while sparing surrounding healthy tissue.
Antioxidant activity
-
Camel milk is rich in both enzymatic and non-enzymatic antioxidants, such as glutathione and vitamins, which play a crucial role in combating free radicals in the body. These antioxidants help eliminate or prevent disruptions in metabolic pathways and reduce the risk of developing diabetes. Additionally, the high content of vitamin C in camel milk contributes to its antioxidant properties, promoting liver function and enhancing immune response. When camel milk is ingested and hydrolyzed, it produces peptides that serve as natural antioxidants and inhibitors of angiotensin-converting enzyme[9]. Furthermore, camel milk possesses anti-ulcer properties due to its significant amounts of vitamins C, A, B2, and E. These vitamins work together to decrease oxidative stress induced by toxic agents, thereby supporting the health of the gastrointestinal system. Additionally, camel milk is particularly rich in magnesium and zinc, both of which are essential for the absorption and metabolism of vitamin B. These qualities of camel milk, highlights its potential benefits in reducing oxidative stress, supporting gastrointestinal health, and promoting vitamin B absorption and metabolism through its vitamin and mineral content[36].
-
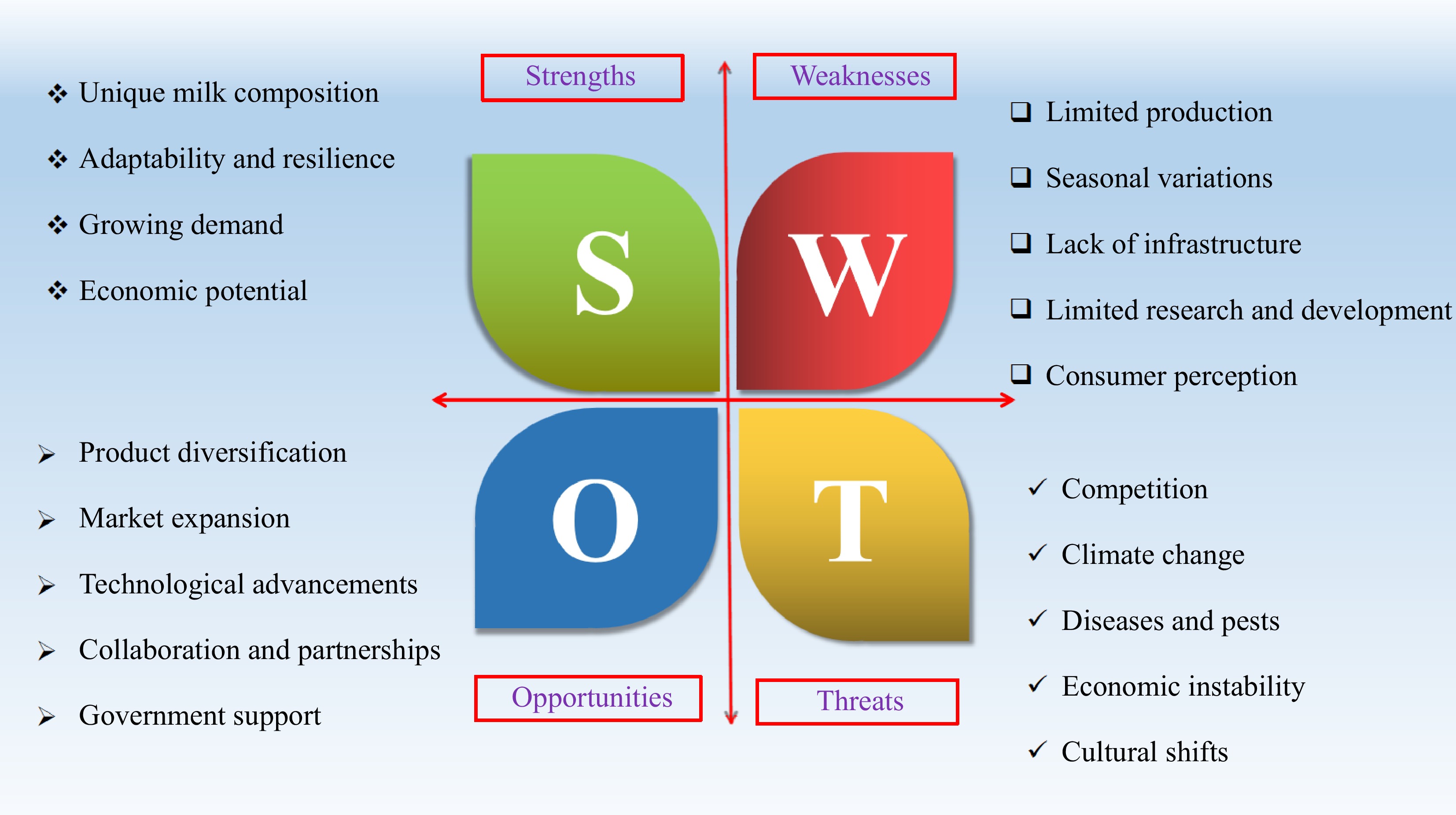
While camel milk holds promise for product development, it's crucial to acknowledge the hurdles linked to its production and market acceptance. These challenges warrant careful examination through strategic analysis, such as the SOAT framework illustrated in Fig. 2, to elucidate several viable strategies that can be pursued.
Strengths
Unique milk composition
-
Camel milk possesses a unique composition, rich in nutrients like vitamins, minerals, and bioactive compounds, offering potential health benefits.
Adaptability and resilience
-
Camels are highly adaptable animals, thriving in harsh environments and resistant to diseases, making them suitable for arid and semi-arid regions.
Growing demand
-
The demand for camel milk and its products is increasing globally, driven by rising health awareness and cultural preferences.
Economic potential
-
Camel production and milk processing can generate income and create employment opportunities in rural areas, contributing to economic development.
Cultural significance
-
Camels hold cultural and religious significance in many societies, fostering a strong connection and potential for sustainable development.
Weaknesses
Limited production
-
Compared to other dairy animals, camels have a lower milk yield, limiting the overall production volume.
Seasonal variations
-
The seasonality that affects camels, including the mating season and variations in weather can impact milk yields, leading to decreased production during hot and dry seasons
Lack of infrastructure
-
The infrastructure and logistics for efficient camel milk production and processing are often lacking in many regions.
Limited research and development
Further research is needed to optimize camel breeding, milk production, and product development.
Consumer perception
-
Camel milk is often perceived as a niche product, and consumers may have limited knowledge or negative perceptions about its taste and properties.
Opportunities
Product diversification
-
Developing a diverse range of camel milk products, including dairy products like yogurt, cheese, ice cream, and non-dairy products such as functional beverages and chocolate, can cater to different consumer preferences and expand market opportunities. Offering a variety of camel milk-based products can help attract a wider consumer base and increase the market demand.
Market expansion
-
Targeting new markets, such as health-conscious consumers and those with specific dietary needs can increase the demand and revenue.
Technological advancements
-
Utilizing advanced technologies for breeding, milk production, and processing can improve efficiency and quality.
Collaboration and partnerships
-
Collaboration among researchers, producers, and entrepreneurs can foster innovation and address challenges in the camel milk industry.
Government support
-
Government policies and initiatives can promote camel production, research, and market development.
Threats
Competition
-
Camel milk faces competition from established dairy products, such as cow milk, which have a wider consumer base and existing distribution networks.
Climate change
-
Can negatively impact camel populations and milk production, particularly in regions already facing water scarcity and extreme weather events.
Diseases and pests
-
Outbreaks of diseases or infestations by pests can significantly impact camel health and productivity.
Economic instability
-
Economic fluctuations and market volatility can affect the profitability of camel production and milk processing.
Cultural shifts
-
Changing cultural preferences and dietary habits may pose challenges for the long-term sustainability of camel production in some regions.
The development of camel and camel milk production presents both opportunities and challenges. By leveraging its strengths, addressing its weaknesses, and capitalizing on opportunities, the camel milk industry can contribute to improved livelihoods, health, and economic development, particularly in arid and semi-arid regions. However, it is crucial to mitigate potential threats through proactive measures and collaborative efforts.
-
Camel milk boasts impressive therapeutic properties, including antimicrobial, antidiabetic, and anti-cancer effects, making it a crucial functional food in pastoral regions due to its unique components and bioactive peptides. However, it often remains underappreciated due to the predominantly arid habitats of camels and limited access to modern agricultural practices among their owners. To maximize the potential of camels and boost milk and meat production, understanding their management is essential. This involves analyzing the strengths, weaknesses, opportunities, and threats related to camel-based projects. By comprehensively grasping factors like nutritional composition and bioactive compounds and promoting innovation through research and breeding programs, we can enhance the value and sustainability of camel-based initiatives, driving progress in this field.
-
The authors confirm contribution to the paper as follows: study conception and design: Abdelazez A, Abd-elmotaal, Abady G; draft manuscript preparation: Abdelazez A; writing- review & editing: Abdelazez A, Abd-elmotaal H; investigation and formal analysis: Abady G; supervision: Abdelazez A, Abd-elmotaal H. All authors reviewed the results and approved the final version of the manuscript.
-
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
-
The authors declare that they have no conflict of interest.
-
# Authors contributed equally: Amro Abdelazez, Heba Abd-elmotaal
- Copyright: © 2024 by the author(s). Published by Maximum Academic Press on behalf of Nanjing Agricultural University. This article is an open access article distributed under Creative Commons Attribution License (CC BY 4.0), visit https://creativecommons.org/licenses/by/4.0/.
-
About this article
Cite this article
Abdelazez A, Abd-elmotaal H, Abady G. 2024. Exploring the potential of camel milk as a functional food: physicochemical characteristics, bioactive components, innovative therapeutic applications, and development opportunities analysis. Food Materials Research 4: e031 doi: 10.48130/fmr-0024-0020
Exploring the potential of camel milk as a functional food: physicochemical characteristics, bioactive components, innovative therapeutic applications, and development opportunities analysis
- Received: 26 June 2024
- Revised: 22 August 2024
- Accepted: 18 September 2024
- Published online: 25 November 2024
Abstract: Cow milk dominates global milk production, accounting for approximately 80%−85% of the total supply. In contrast, milk from buffalo, goats, sheep, and camels collectively contributes to only about 15% of milk production. Among these alternative milk sources, camel milk stands out as a traditional functional food renowned for its bioactive components. These components offer not only nutritional benefits but also potential immunological advantages, owing to the presence of immunoglobulin, insulin, lactoferrin, and other bioactive substances. However, despite its remarkable attributes, camel milk and its derivatives remain underutilized due to limited production primarily restricted to arid regions. In light of this, the present study aims to shed light on the physicochemical characteristics, and key bioactive elements, as well as the strengths, weaknesses, opportunities, and threats associated with camel milk. Moreover, we emphasize the urgent need for enhanced camel milk production and increased research funding to explore the biogenic applications of its constituents.
-
Key words:
- Camel milk /
- Functional foods /
- Immunoglobulin /
- Antidiabetic /
- Lactoferrin